Probing into the crystal plane effect on the reduction of α-Fe2O3 in CO by Operando Raman spectroscopy
-
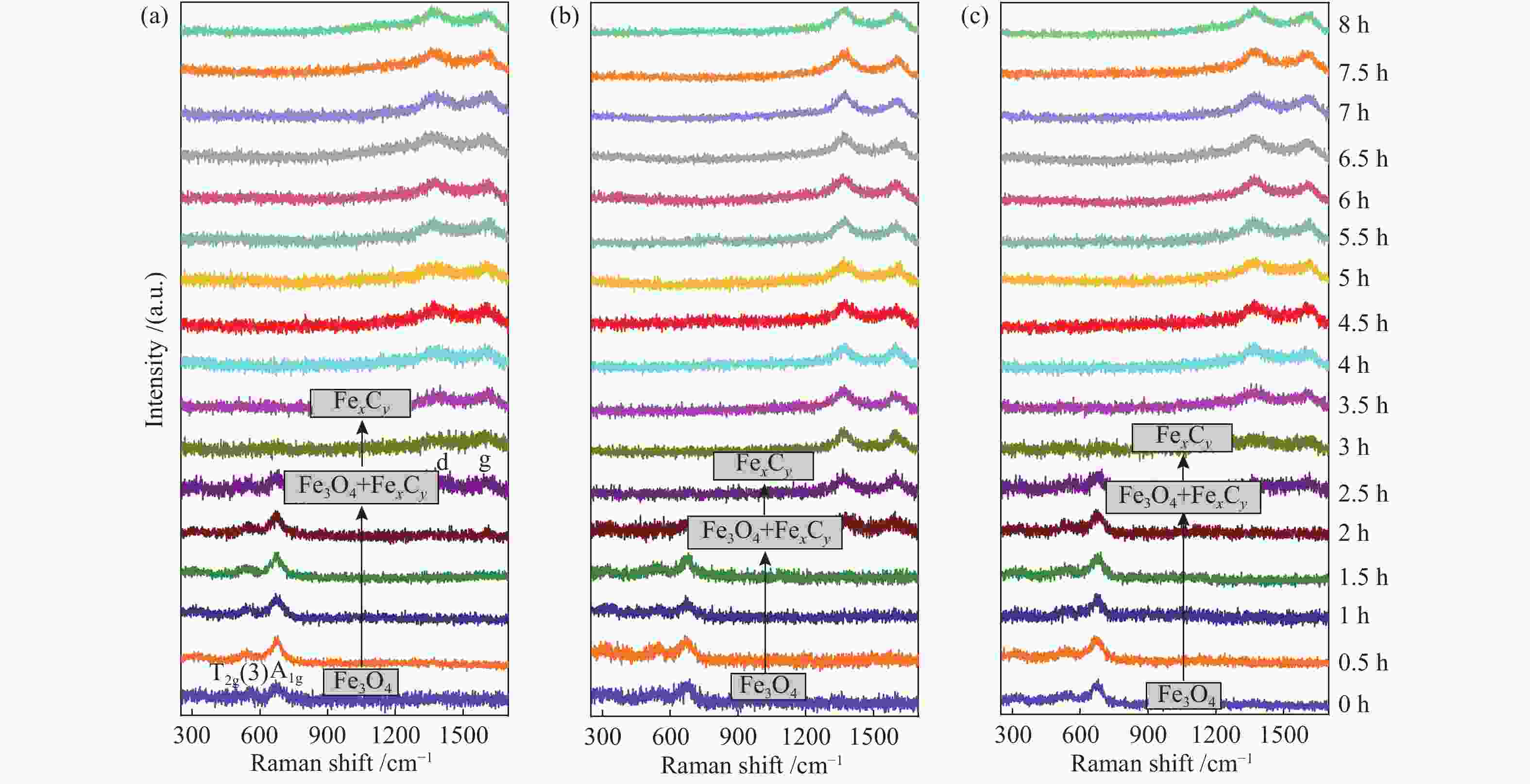
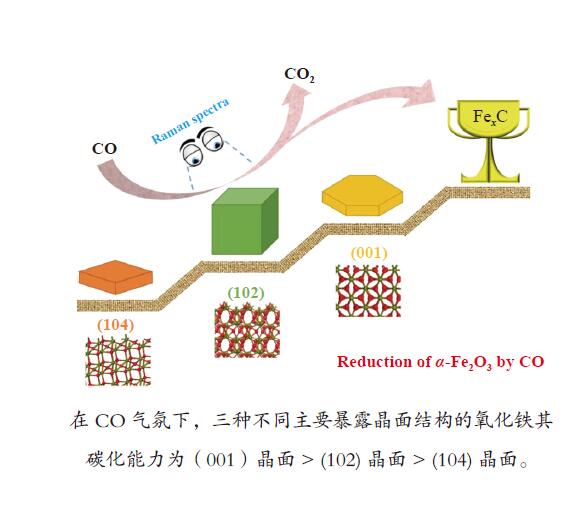
摘要: 氧化铁作为铁基费托合成主催化剂的前驱体,其还原活化过程对催化剂整体性能有着至关重要的影响。α-Fe2O3作为一种晶体材料,其暴露晶面对铁基催化剂的还原和活化过程有重要影响,但目前对此仍然缺乏必要的研究。本研究合成了六角片、立方体和菱面体三种不同形貌的α-Fe2O3单晶纳米颗粒,其对应主暴露面分别为(001)、(102)和(104)晶面,然后利用原位拉曼光谱(ORS)研究了CO还原过程中α-Fe2O3晶体结构的转变过程。结果发现,与(104)和(102)晶面相比,(001)晶面具有更好的还原活性。对于三种晶面上的CO吸附和CO2脱附,SEM、TEM和XPS等表征以及DFT理论计算结果表明,CO2脱附是还原过程的决速步骤;(001)晶面对氧原子的束缚能力较弱,导致了其表面CO2更容易脱附,从而促进了整个还原过程。Abstract: For the Fe-based catalysts in Fischer-Tropsch synthesis, the reduction and activation process of α-Fe2O3 precursor has a significant effect on the catalytic performance. As a crystalline material, the reduction and activation of α-Fe2O3 is assuredly influenced by the exposed crystal plane; however, there is a lack of necessary research in this regard. In this work, α-Fe2O3 nanocrystals of three different morphologies, viz., pseudo-cubic, hexagonal-plate and rhombohedra, were synthesized, which mainly expose the crystal planes of (102), (001) and (104), respectively. The evolution of α-Fe2O3 crystal structure was then investigated in CO atmosphere by using the Operando Raman spectroscopy (ORS). The results show that the α-Fe2O3 (001) plane has a better reductive activity in comparison to the (104) and (102) planes. The SEM, TEM, XPS and XRD characterization and DFT calculation results reveal that CO2 desorption is a decisive step for the reduction of α-Fe2O3; owing to the weak binding ability of (001) crystal plane to oxygen atoms, the desorption of CO2 on the (001) crystal plane is much easier, which can promote the reduction process.
-
Key words:
- α-Fe2O3 /
- crystal planes /
- Operando Raman spectroscopy /
- reduction
-
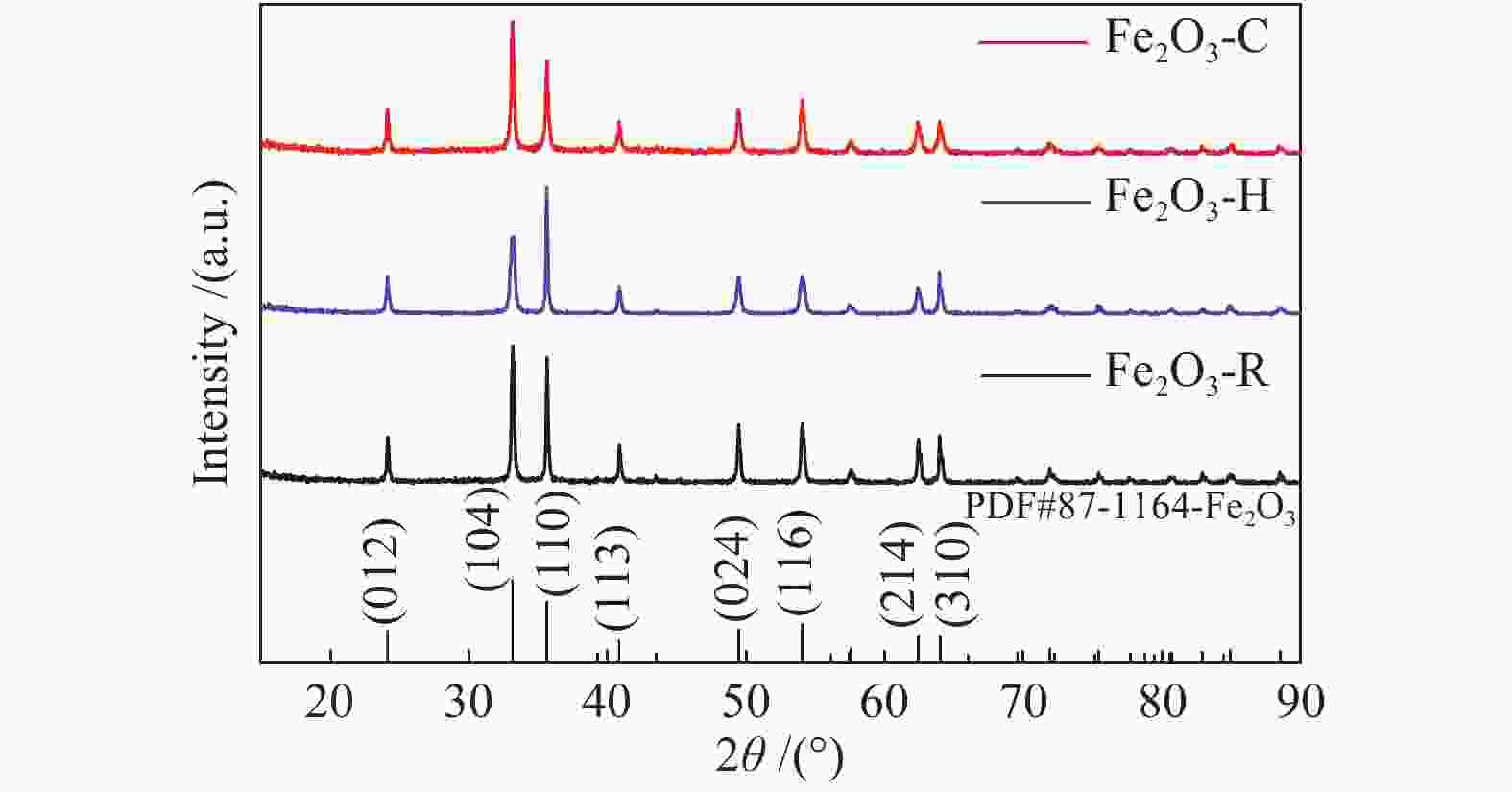
Table 1 Length of O–Fe bond and the adsorption energy of CO adsorption on different planes of α-Fe2O3
Facet O–Fe bond length/Å Adsorption energy/eV 102-1 2.12 4.15 102-2 1.83 4.21 001-1 2.46 3.76 001-1 2.40 3.79 104-1 1.99 1.98 104-1 2.01 0.39 Table 2 Peak position (eV) in the Fe 2p and O 1s XPS spectra and relative contents of various O species (in parentheses) estimated from O 1s XPS spectra for different α-Fe2O3 samples
Sample Fe 2p1/2 Fe 2p3/2 OL OV OC Fe2O3-C 724.1 710.4 529.3 (59.7%) 529.8 (24.6%) 531.2 (15.7%) Fe2O3-H 724.3 710.7 529.4 (56.3%) 529.9 (27.1%) 531.2 (16.6%) Fe2O3-R 724.1 710.7 529.4 (60.7%) 529.7 (24.0%) 531.4 (15.3%) -
[1] SCHLOGL R. Heterogeneous catalysis[J]. Angew Chem Int Ed,2015,54(11):3465−3520. doi: 10.1002/anie.201410738 [2] WANG H, JIN B, WANG H, MA N, LIU W, WENG D, WU X, LIU S. Study of Ag promoted Fe2O3@CeO2 as superior soot oxidation catalysts: the role of Fe2O3 crystal plane and tandem oxygen delivery[J]. Appl Catal B: Environ,2018,237:251−262. doi: 10.1016/j.apcatb.2018.05.093 [3] DING D, HUANG Y, ZHOU C, LIU Z, REN J, ZHANG R, WANG J, ZHANG Y, LEI Z, ZHANG Z, ZHI C. Facet-controlling agents free synthesis of hematite crystals with high-index planes: Excellent photodegradation performance and mechanism insight[J]. ACS Appl Mater Interfaces,2016,8(1):142−151. doi: 10.1021/acsami.5b07843 [4] WU J B, SUN Z P, WEI Z H, QIN Z F, ZHAO Y X. Catalytic performance and mechanistic insights into the synthesisof polyoxymethylene dimethyl ethers from dimethoxymethaneand trioxymethylene over ZSM-5 zeolite[J]. Catal Lett,2021,151:670−684. [5] SUN J, CHEN Y, CHEN J. Morphology effect of one-dimensional iron oxide nanocatalysts on Fischer-Tropsch synthesis[J]. Catal Sci Technol,2016,6(20):7505−7511. doi: 10.1039/C6CY01258B [6] NUMPILAI T, WITOON T, CHANLEK N, LIMPHIRAT W, BONURA G, CHAREONPANICH M, LIMTRAKUL J. Structure-activity relationships of Fe-Co/K-Al2O3 catalysts calcined at different temperatures for CO2 hydrogenation to light olefins[J]. Appl Catal A: Gen,2017,547:219−229. doi: 10.1016/j.apcata.2017.09.006 [7] ZHANG Y, CAO C, ZHANG C, ZHANG Z, LIU X, YANG Z, ZHU M, MENG B, XU J, HAN Y. The study of structure-performance relationship of iron catalyst during a full life cycle for CO2 hydrogenation[J]. J Catal,2019,378:51−62. doi: 10.1016/j.jcat.2019.08.001 [8] XIN Y, ZHANG N, LI Q, ZHANG Z, CAO X, ZHENG L, ZENG Y, ANDERSO J A. Active site identification and modification of electronic states by atomic-scale doping to enhance oxide catalyst innovation[J]. ACS Catal,2018,8(2):1399−1404. doi: 10.1021/acscatal.7b02638 [9] XIANG Q, CHEN G, LAU C. Effects of morphology and exposed facets of α-Fe2O3 nanocrystals on photocatalytic water oxidation[J]. RSC Adv,2015,5(64):52210−52216. doi: 10.1039/C5RA09354F [10] WEI S, WANG W, FU X, LI S, JIA C. The effect of reactants adsorption and products desorption for Au/TiO2 in catalyzing CO oxidation[J]. J Catal,2019,376:134−145. doi: 10.1016/j.jcat.2019.06.038 [11] JIAN Y, YU T, JIANG Z, YU Y, DOUTHWAITE M, LIU J, ALBILALI R, HE C. In-depth understanding of the morphology effect of alpha-Fe2O3 on catalytic ethane destruction[J]. ACS Appl Mater Interfaces,2019,11:11369−11383. doi: 10.1021/acsami.8b21521 [12] QIN C, HOU B, WANG J, WANG Q, WANG G, YU M, CHEN C, JIA L, LI D. Crystal-plane-dependent Fischer-Tropsch performance of cobalt catalysts[J]. ACS Catal,2018,8(10):9447−9455. doi: 10.1021/acscatal.8b01333 [13] LIU Y, LUA F, TANG Y, LIU M, TAO F F, ZHANG Y. Effects of initial crystal structure of Fe2O3 and Mn promoter on effective active phase for syngas to light olefins[J]. Appl Catal B: Environ,2020,261:118219. doi: 10.1016/j.apcatb.2019.118219 [14] LIU Z, HU P. A new insight into Fischer-Tropsch synthesis[J]. J Am Chem Soc,2002,124:11568−11569. doi: 10.1021/ja012759w [15] SMIT E D, WECKHUYSEN B M. The renaissance of iron-based Fischer-Tropsch synthesis: on the multifaceted catalyst deactivation behaviour[J]. Chem Soc Rev,2008,37:2758−2781. doi: 10.1039/b805427d [16] DRY M E. The Fischer-Tropsch process: 1950–2000[J]. Catal Today,2002,71:227−241. doi: 10.1016/S0920-5861(01)00453-9 [17] MARKVOORT A J, VANSANTEN R A, HILBERS P A J, HENSSEN E J M. Kinetics of the Fischer-Tropsch reaction[J]. Angew Chem Int Ed,2012,51(36):9015−9019. doi: 10.1002/anie.201203282 [18] DING M, YANG Y, WU B, WANG T, XIANG H, LI Y. Effect of reducing agents on microstructure and catalytic performance of precipitated iron-manganese catalyst for Fischer-Tropsch synthesis[J]. Fuel Process Technol,2011,92:2353−2359. doi: 10.1016/j.fuproc.2011.08.011 [19] CHEN G, WATERHOUSE G I N, SHI R, ZHAO J, LI Z, WU L, TUNG C, ZHANG T. From solar energy to fuels: Recent advances in light‐driven C1 chemistry[J]. Angew Chem Int Ed,2019,58(49):17528−17551. doi: 10.1002/anie.201814313 [20] DRY M E. High quality diesel via the Fischer-Tropsch process-A review[J]. J Chem Technol Biotechnol,2002,77(1):43−50. doi: 10.1002/jctb.527 [21] MEGANM M, KARAKAYA C, KEE R J, BRIANG T. In situ formation of metal carbide catalysts[J]. ChemCatChem,2017,9:3090−3101. doi: 10.1002/cctc.201700304 [22] WANG D, CHEM B, DUAN X, CHEN D, ZHOU X. Iron-based Fischer-Tropsch synthesis of lower olefins: the nature of χ-Fe5C2 catalyst and why and how to introduce promoters[J]. J Energy Chem,2016,25:911−916. doi: 10.1016/j.jechem.2016.11.002 [23] JANBROERS S, LOUWEN J N, ZANDBERGEN H W, KOOYMAN P J. Insights into the nature of iron-based Fischer-Tropsch catalysts from quasi in situ TEM-EELS and XRD[J]. J Catal,2009,268(2):235−242. doi: 10.1016/j.jcat.2009.09.021 [24] PEREZ S, MONDRAGON F, MORENO A. Iron ore as precursor for preparation of highly active χ-Fe5C2 core-shell catalyst for Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,2019,587:117264. doi: 10.1016/j.apcata.2019.117264 [25] VARANDA L C, JAFELICCI M, TARTAJ P, OGRADY K, GONZALEZ-CARRENO T, MORALES M P, MUNOZ T, SERNA C J. Structural and magnetic transformation of monodispersed iron oxide particles in a reducing atmosphere[J]. J Appl Phys,2002,92(4):2079−2085. doi: 10.1063/1.1496124 [26] FEILMMAYR C, THURNHOFER A, WINTER F, MALI H, SCHENK J. Reduction behavior of hematite to magnetite under fluidized bed conditions[J]. ISIJ Int,2004,44:1125−1133. doi: 10.2355/isijinternational.44.1125 [27] SANTEN R A V, GHOURI M M, SHETTY S, HENSE E M H. Structure sensitivity of the Fischer-Tropsch reaction; molecular kinetics simulations[J]. Catal Sci Technol,2011,1:891−911. doi: 10.1039/c1cy00118c [28] BUKUR D B, KORANNE M, LANG X, RAO K R P M, HUFFMAN G P. Pretreatment effect studies with a precipitated iron Fischer-Tropsch catalyst[J]. Appl Catal A: Gen,1995,126:85−113. doi: 10.1016/0926-860X(95)00020-8 [29] LI S, OBRIEN R J, MEITZNER G D, HAMDEH H, DAVIS B H, IGLESIA E. Structural analysis of unpromoted Fe-based Fischer-Tropsch catalysts using X-ray absorption spectroscopy[J]. Appl Catal A: Gen,2001,219:215−222. doi: 10.1016/S0926-860X(01)00694-9 [30] DING M, YANG Y, WU B, LI Y, WANG T, MA L. Study on reduction and carburization behaviors of iron phases for iron-based Fischer-Tropsch synthesis catalyst[J]. Appl Energy,2014,160:982−989. [31] CANO L A, CAGNOLI M V, BENGOA J F, ALVAREZ A M, MARCHETTI S G. Effect of the activation atmosphere on the activity of Fe catalysts supported on SBA-15 in the Fischer-Tropsch synthesis[J]. J Catal,2011,278:310−320. doi: 10.1016/j.jcat.2010.12.017 [32] ZHANG Y, FU D, LIU X, ZHANG Z, ZHANG C, SHI B, XU J, HAN Y. Operando spectroscopic study of dynamic structure of iron oxide catalysts during CO2 hydrogenation[J]. ChemCatChem,2018,10(6):1272−1276. doi: 10.1002/cctc.201701779 [33] AN H, ZHANG F, GUAN Z, LIU X, FAN F, LI C. Investigating the coke formation mechanism of H-ZSM-5 during methanol dehydration using operando UV-Raman spectroscopy[J]. ACS Catal,2018,8:9207−9215. doi: 10.1021/acscatal.8b00928 [34] GAUR A, SCHUMANN M, RAUN K V, STEHLE M, BEATO P, JENSEN A D, GRUNWALDT J D, HØJ M. Operando XAS/XRD and Raman spectroscopic study of structural changes of the iron Molybdate catalyst during selective oxidation of methanol[J]. ChemCatChem,2019,11:4871−4883. doi: 10.1002/cctc.201901025 [35] BANARES M A. Operando spectroscopy: the knowledge bridge to assessing structure-performance relationships in catalyst nanoparticles[J]. Adv Mater,2011,23:5293−5301. doi: 10.1002/adma.201101803 [36] PATLOLLA A, CARINO E V, EHRLICH S N, STAVITSKI E, FRENKEL A I. Application of operando XAS, XRD, and Raman spectroscopy for phase speciation in water gas shift reaction catalysts[J]. ACS Catal,2012,2:2216−2223. doi: 10.1021/cs300414c [37] ZHANG Y, FU D, XU X, SHENG Y, XU J, HAN Y. Application of operando spectroscopy on catalytic reactions[J]. Curr Opin Chem Eng,2016,12:1−7. doi: 10.1016/j.coche.2016.01.004 [38] FU D, DAI W, XU X, MAO W, SU J, ZHANG Z, SHI B, SMITH J, LI P, XU J, HAN Y. Probing the structure evolution of iron-based Fischer-Tropsch to produce olefins by operando Raman spectroscopy[J]. ChemCatChem,2015,7:752−756. doi: 10.1002/cctc.201402980 [39] SATTLER J J H B, MENS A M, WECKHUYSEN B M. Real-time quantitative operando Raman spectroscopy of a CrOx/Al2O3 propane dehydrogenation catalyst in a pilot-scale reactor[J]. ChemCatChem,2014,6(11):3139−3145. doi: 10.1002/cctc.201402649 [40] KRESSE G, FURTHMULLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set[J]. Phys Rev B,1996,54:11169−11186. doi: 10.1103/PhysRevB.54.11169 [41] KRESSE G, HAFNER J. Norm-conserving and ultrasoft pseudopotentials for first-row and transition elements[J]. J Phys Condens Matter,1994,6:8245−8257. doi: 10.1088/0953-8984/6/40/015 [42] BLOCHL P E. Projector augmented-wave method[J]. Phys Rev B: Condens Matter,1994,50:17953−17979. doi: 10.1103/PhysRevB.50.17953 [43] KRESSE G, JOUBERT D. From ultrasoft pseudopotentials to the projector augmented-wave method[J]. Phys Rev B,1999,59:1758−1775. [44] PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple[J]. Phys Rev Lett,1996,77:3865−3868. doi: 10.1103/PhysRevLett.77.3865 [45] MONKHORST H J, PACK J D. Special points for Brillouin-zone integrations[J]. Phys Rev B,1976,13:5188−5192. doi: 10.1103/PhysRevB.13.5188 [46] COCOCCIONI M, DEGIRONCOLI S. Linear response approach to the calculation of the effective interaction parameters in the LDA + U method[J]. Phys Rev B,2005,71:035105. doi: 10.1103/PhysRevB.71.035105 [47] HANESCH M. Raman spectroscopy of iron oxides and (oxy)hydroxides at low laser power and possible applications in environmental magnetic studies[J]. Geophys J Int,2009,177(3):941−948. doi: 10.1111/j.1365-246X.2009.04122.x [48] SHEBANOVA O N, LAZOR P. Raman spectroscopic study of magnetite (FeFe2O4): A new assignment for the vibrational spectrum[J]. J Solid State Chem,2003,174(2):424−430. doi: 10.1016/S0022-4596(03)00294-9 [49] BELIN T, EPRON F. Characterization methods of carbon nanotubes: a review[J]. Mater Sci Eng B,2005,119:105−118. doi: 10.1016/j.mseb.2005.02.046 [50] DONG C, SHENG S, QIN W, LU Q, ZHAO Y, WANG X, ZHANG J. Density functional theory study on activity of α-Fe2O3 in chemical-looping combustion system[J]. Appl Surf Sci,2011,257(20):8647−8652. doi: 10.1016/j.apsusc.2011.05.042 [51] LIU Y, CHUNG J, JANG Y, MAO S, KIM B M, WANG Y, GUO X. Magnetically recoverable nanoflake-shaped iron oxide/Pt heterogeneous catalysts and their excellent catalytic performance in the hydrogenation reaction[J]. ACS Appl Mater Interfaces,2014,6:1887−1892. doi: 10.1021/am404904p [52] WU Z, LI Z, LI H, SUN M, HAN S, CAI C, SHEN W, FU Y. Ultrafast response/recovery and high selectivity of the H2S gas sensor based on α-Fe2O3 nano-ellipsoids from one-step hydrothermal synthesis[J]. ACS Appl Mater Interfaces,2019,11:12761−12769. doi: 10.1021/acsami.8b22517 [53] SUN L, ZHAN W, LI Y, WANG F, ZHANG X, HAN X. Understanding the facet-dependent catalytic performance of hematite microcrystals in a CO oxidation reaction[J]. Inorg Chem Front,2018,5:2332−2339. [54] LIU X, LIU J, CHANG Z, SUN X, LI Y. Crystal plane effect of Fe2O3 with various morphologies on CO catalytic oxidation[J]. Catal Commun,2011,12:530−534. doi: 10.1016/j.catcom.2010.11.016 -





 下载:
下载: