The effect of NaF amount on solid base catalysts derived from F-Ca-Mg-Al layered double hydroxides and dimethyl carbonate synthesis
-
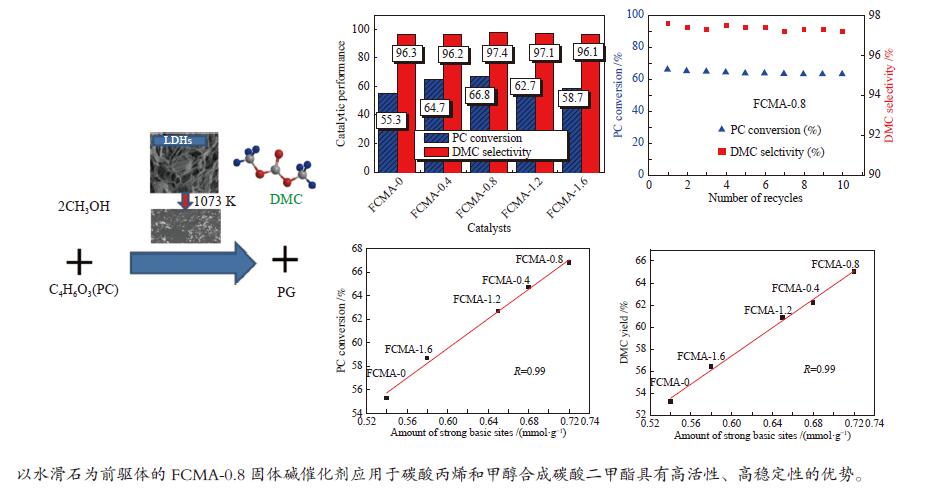
摘要: 以碳酸丙烯(PC)和甲醇为原料,经酯交换反应合成的多功能、环保的碳酸二甲酯(DMC)是一种绿色、节能的合成方法。CaO固体碱催化剂对该反应具有良好的催化性能,但其再生性不理想。以F-Ca-Mg-Al水滑石(LDHs)为原料,制备了一系列不同NaF用量的固体碱催化剂,并对其进行了表征和酯交换反应测试。与不加氟的FCMA-0催化剂相比,经氟改性后的催化剂的比表面积、碱量、催化活性等性能均有明显提高。催化活性由高到低依次为:FCMA-0.8 > FCMA-0.4 > FCMA-1.2 > FCMA-1.6 > FCMA-0,这与总碱位量和强碱位量一致。FCMA-0.8催化剂活性最好,与纯CaO催化剂的相当,PC转化率为66.8%,DMC选择性为97.4%,DMC收率为65.1%。在10次循环使用后,FCMA-0.8催化剂的DMC收率仅下降3.9% (CaO催化剂下降33.2%)。FCMA-0.8在PC与甲醇酯交换制DMC方面具有良好的工业应用前景。Abstract: Versatile and environmentally benign dimethyl carbonate (DMC) synthesized by propylene carbonate (PC) and methanol via transesterification is green and energy efficient. A series of solid base catalysts derived from F-Ca-Mg-Al layered double hydroxides (LDHs) with different NaF amount were prepared, characterized and tested for the transesterification reaction. The properties of the catalysts modified by fluorine have improved obviously. The catalytic activity increases in the order of: FCMA-0.8 > FCMA-0.4 > FCMA-1.2 > FCMA-1.6 > FCMA-0, which is consistent with the total basic sites amount and the strong basic sites amount. FCMA-0.8 has the best catalytic activity as pure CaO catalyst, and the PC conversion, DMC selectivity and DMC yield are 66.8%, 97.4% and 65.1%, respectively. Furthermore, the DMC yield for FCMA-0.8 just decreased 3.9% (33.2% for CaO catalyst) after 10 recycles. FCMA-0.8 has good prospects in the transesterification of PC with methanol to DMC for industrial application.
-
Key words:
- dimethyl carbonate /
- methanol /
- solid base /
- NaF /
- transesterification
-
Figure 6 Correlation between (a): the DMC yield and the amount of strong basic sites; (b): the DMC yield and the amount of total basic sites; (c): the PC conversion and the amount of strong basic sites; (d): the PC conversion and the amount of total basic sites
reaction conditions: n(methanol) / n(PC) = 12, catalyst weight = 2% of total reactants, 333 K, 2 h
Table 1 Structure parameters and basicity of the FCMA-n catalysts
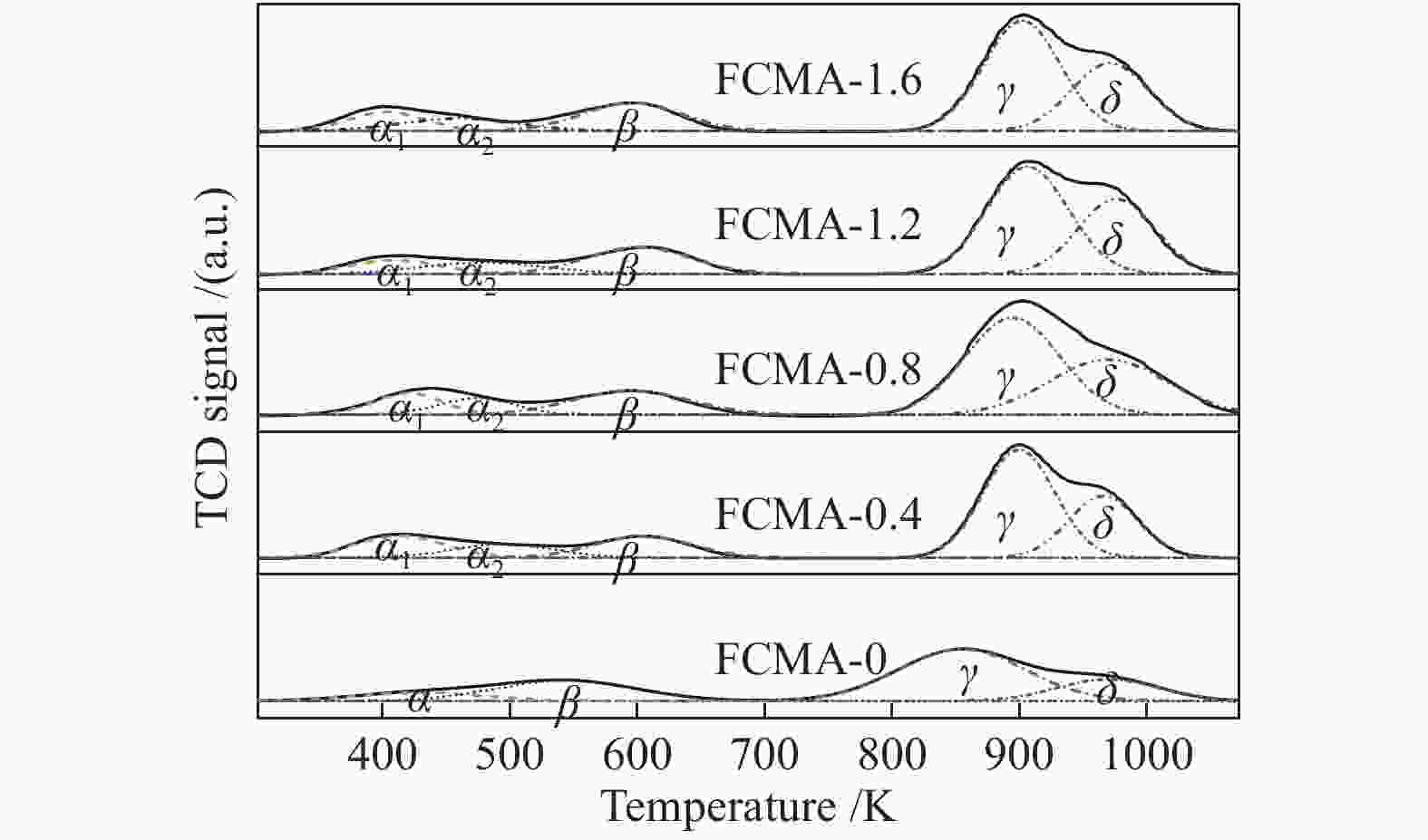
Catalyst SBET/ (m2·g−1) vpore/ (cm3·g−1) CO2 uptake/ (mmol·g−1) Total basic amount/ (mmol·g−1) α β γ δ FCMA-0 42.3 0.30 0.09 0.23 0.54 0.19 1.05 FCMA-0.4 48.2 0.39 0.24 0.24 0.68 0.47 1.63 FCMA-0.8 55.6 0.41 0.23 0.25 0.72 0.53 1.73 FCMA-1.2 45.3 0.38 0.27 0.17 0.65 0.35 1.44 FCMA-1.6 44.1 0.32 0.20 0.21 0.58 0.39 1.38 Table 2 Composition of FCMA-n catalysts
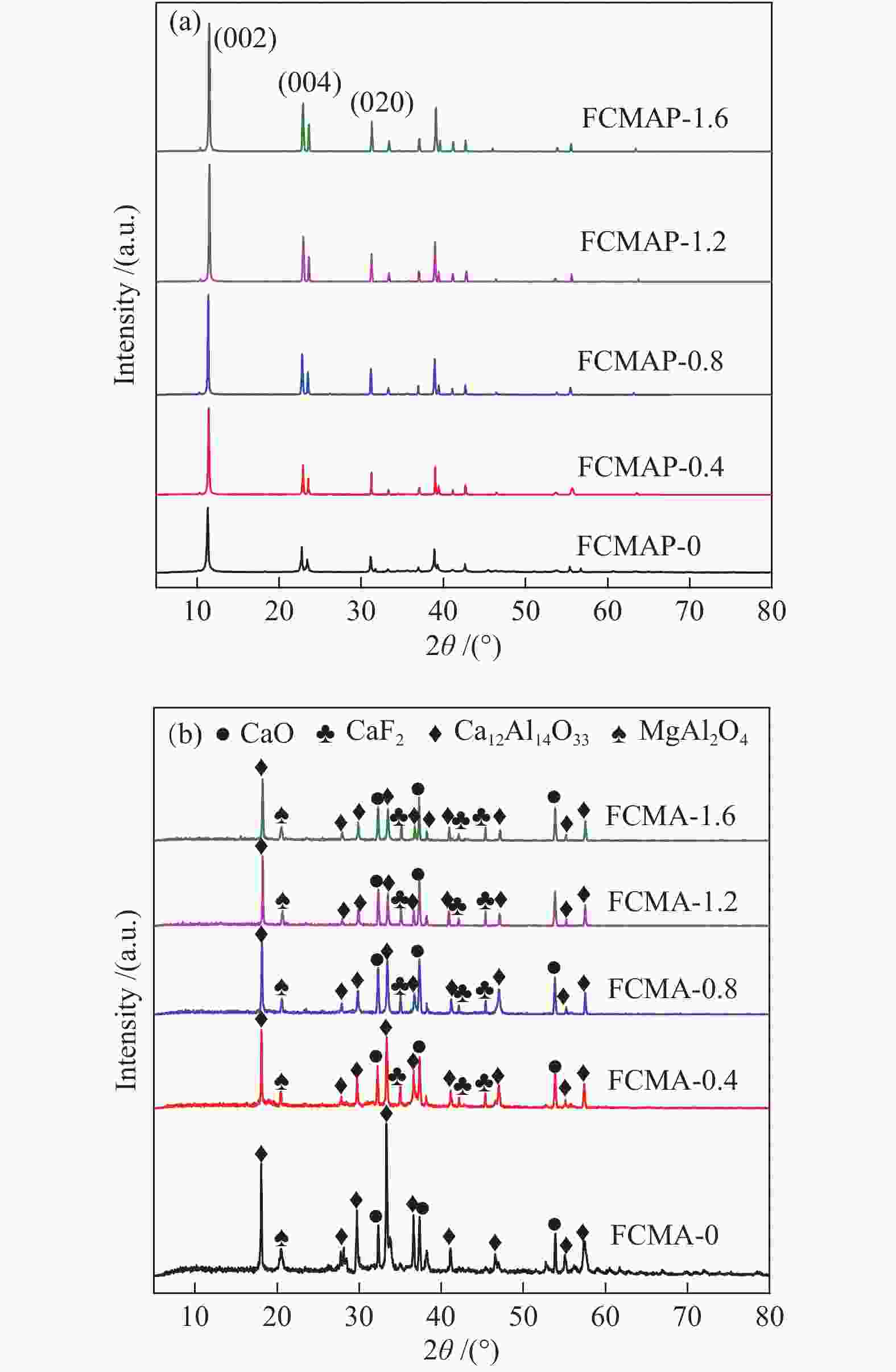
Catalyst Composition/ (mol %) F/Ca (atomic ratio) Ca Mg Al FCMA-0 50.9 (50.5) 16.9 (16.7) 32.2 (32.8) 0 FCMA-0.4 51.5 (50.9) 17.2 (17.1) 31.3 (32.0) 0.38 (0.38) FCMA-0.8 52.8 (51.4) 17.6 (17.5) 29.6 (31.1) 0.75 (0.76) FCMA-1.2 51.4 (50.8) 17.1 (17.0) 31.5 (32.2) 1.14 (1.14) FCMA-1.6 51.3 (50.7) 17.0 (16.9) 31.7 (32.4) 1.55 (1.54) the values outside and inside the parentheses were obtained by XPS and ICP measurements, respectively Table 3 Catalytic activity and recyclability over Ca-based solid basic catalyst
Sample PC conversion/% DMC selectivity/% DMC yield/% DMC yield decrease/%* References CaO 68.8 (35.5) 95.6 65.8 (32.6) 33.2 (10) [17] CA-3 − − 59.9 10.9 (4) [18] CA-2 53.7 (41.9) 92.8 49.8 4.8 (4),
12.6 (10)[16,17] Mg-CA 55.3 (52.6) 96.3 (92.5) 53.3 (48.6) 4.7 (10) [16] CA-F− 65.9(60.1) 95.3 62.8 5.6 (10) [17] FCMA-0.8 66.8 97.4 65.1 3.9 (10) this work reaction conditions: n(methanol) / n(PC) = 12, catalyst weight = 2% of total reactants, 333 K, 2 h
−: data were not reported in the literature,
*: data in the parenthesis stand for the recycle times,
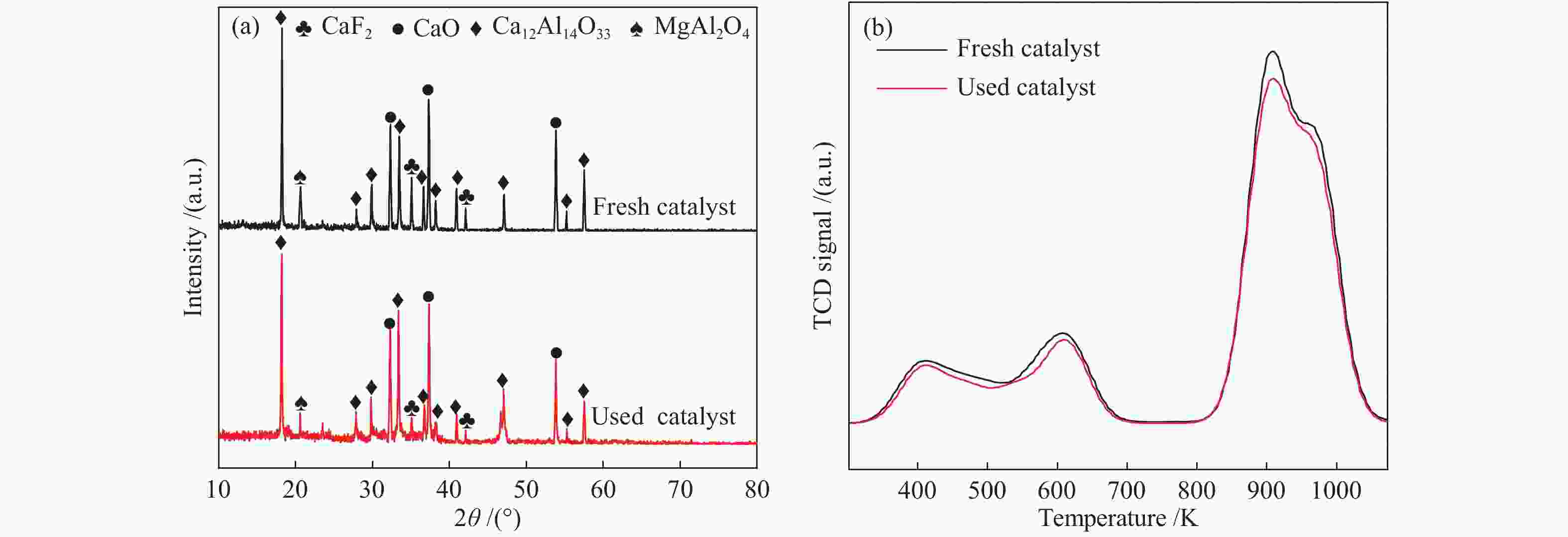
other data in the parenthesis stand for the corresponding values after 10 times recyclesTable 4 Elemental compositions of the fresh and used FCMA-0.8 catalysts
Catalyst Bulk compositions/mol % Ca a Mg a Al a F b Ca∶Mg∶Al F∶Ca Fresh FCMA-0.8 catalyst 36.9 12.6 22.3 28.2 2.93∶1∶1.77 0.76 Used FCMA-0.8 catalyst 35.1 12.0 25.0 27.9 2.92∶1∶2.08 0.79 a: determined by the ICP, b: determined by the ionic chromatography -
[1] TUNDO P, SELVA M. The chemistry of dimethyl carbonate[J]. ACC Chem Res,2002,35(9):706−716. doi: 10.1021/ar010076f [2] CROCELLA V, TABANELLI T, VITILLO J. G, COSTENARO D, BISIO C, CAVANI F, BORDIGA S. A multi-technique approach to disclose the reaction mechanism of dimethyl carbonate synthesis over amino-modified SBA-15 catalysts[J]. Appl Catal B: Environ,2017,211:323−336. doi: 10.1016/j.apcatb.2017.04.013 [3] TUNDO P, MUSOLINO M, ARICO F. The reactions of dimethyl carbonate and its derivatives[J]. Green Chem,2018,20(1):28−85. doi: 10.1039/C7GC01764B [4] WANG D F, ZHANG X L, MA J, YU H W, SHEN J Z, WEI W. La-modified mesoporous Mg-Al mixed oxides: effective and stable base catalysts for the synthesis of dimethyl carbonate from methyl carbamate and methanol[J]. Catal Sci Technol,2016,6(5):1530−1545. doi: 10.1039/C5CY01712B [5] HUANG S Y, YAN B, WANG S P, MA X B. Recent advances in dialkyl carbonates synthesis and applications[J]. Chem Soc Rev,2015,44(10):3079−3116. doi: 10.1039/C4CS00374H [6] SAADA R, KELLICI S, HEILI T, MORGAN D, SAHA B. Greener synthesis of dimethyl carbonate using a novel ceria-zirconia oxide/grapheme nanocomposite catalyst[J]. Appl Catal B: Environ,2015,168−169:353−362. [7] TAMBOLI A H, CHAUGULE A A, KIM H. Catalytic developments in the direct dimethyl carbonate synthesis from carbon dioxide and methanol[J]. Chem Eng J,2017,323:530−544. doi: 10.1016/j.cej.2017.04.112 [8] FIORANI G, PEROSA A, SELVA M. Dimethyl carbonate: A versatile reagent for a sustainable valorization of renewables[J]. Green Chem,2018,20(2):288−322. doi: 10.1039/C7GC02118F [9] SRIVASTAVA R, SRINIVAS D, RATNASAMY P. Fe-Zn double-metal cyanide complexes as novel, solid transesterification catalysts[J]. J Catal,2006,241(1):34−44. doi: 10.1016/j.jcat.2006.04.002 [10] XU J, WU H T, MA C M, XUE B, LI Y X, CAO Y. Ionic liquid immobilized on mesocellular silica foam as an efficient heterogeneous catalyst for the synthesis of dimethyl carbonate via transesterification[J]. Appl Catal A: Gen,2013,464–465:357−363. [11] MURUGAN C, BAJAJ H C, JASRA R V. Transesterification of propylene carbonate by methanol using KF/Al2O3 as an efficient base catalyst[J]. Catal Lett,2010,137:224−231. doi: 10.1007/s10562-010-0348-6 [12] GAO Y, XU C. Synthesis of dimethyl carbonate over waste eggshell catalyst[J]. Catal Today,2012,190(1):107−111. doi: 10.1016/j.cattod.2011.12.004 [13] WANG H, WANG M H, ZHAO N, WEI W, SUN Y H. CaO-ZrO2 solid solution: A highly stable catalyst for the synthesis of dimethyl carbonate from propylene carbonate and methanol[J]. Catal Lett,2005,105:253−257. doi: 10.1007/s10562-005-8699-0 [14] WANG H, WANG M H, LIU S G, ZHAO N, WEI W, SUN Y H. Influence of preparation methods on the structure and performance of CaO-ZrO2 catalyst for the synthesis of dimethyl carbonate via transesterification[J]. J Mol Catal A,2006,258(1-2):308−312. doi: 10.1016/j.molcata.2006.05.050 [15] WEI T, WANG M H, WEI W, SUN Y H, ZHONG B. Synthesis of dimethyl carbonate by transesterification over CaO/carbon composites[J]. Green Chem,2003,5(3):343−346. doi: 10.1039/b210716n [16] LIAO Y H, LI F, DAI X, ZHAO N, XIAO F K. Solid base catalysts derived from Ca-M-Al (M = Mg, La, Ce, Y) layered double hydroxides for dimethyl carbonate synthesis by transesterification of methanol with propylene carbonate[J]. Chin J Catal,2017,38(11):1860−1869. doi: 10.1016/S1872-2067(17)62898-5 [17] LIAO Y H, LI F, PU Y F, WANG F, DAI X, ZHAO N, XIAO F K. Solid base catalysts derived from Ca-Al-X (X = F-, Cl- and Br-) layered double hydroxides for methanolysis of propylene carbonate[J]. RSC Adv,2018,8(2):785−791. doi: 10.1039/C7RA10832J [18] LIAO Y H, LI F, DAI X, ZHAO N, XIAO F K. Dimethyl carbonate synthesis over solid base catalysts derived from Ca-Al layered double hydroxides[J]. Chem Pap,2018,72:1963−1971. doi: 10.1007/s11696-018-0408-8 [19] XU S L, CHEN Z R, ZHANG B W, YU J H, ZHANG F. Z, EVANS D G. Facile preparation of pure CaAl-layered double hydroxides and their application as a hardening accelerator in concrete[J]. Chem Eng J,2009,155(3):881−885. doi: 10.1016/j.cej.2009.08.003 [20] KOCIK J, HAJEK M, TROPPOVA I. The factors influencing stability of Ca-Al mixed oxides as a possible catalyst for biodiesel production[J]. Fuel Process Technol,2015,134:297−302. doi: 10.1016/j.fuproc.2015.02.013 [21] HAN M S, LEE B G, AHN B S, PARK K Y, HONG S I. Kinetics of dimethyl carbonate synthesis from ethylene carbonate and methanol using alkali-metal compounds as catalysts[J]. React Kinet Catal Lett,2001,73:33−38. doi: 10.1023/A:1013904317108 [22] OESTREICHER V, JOBBAGY M. One pot synthesis of Mg2Al(OH)6Cl·1.5H2O layered double hydroxides: the epoxide route[J]. Langmuir,2013,29(39):12104−12109. doi: 10.1021/la402260m [23] ANGELESCU E, PAVEL O D, BIRJEGA R, FLOREA M, ZAVOIANU R. The impact of the “memory effect” on the catalytic activity of Mg/Al; Mg, Zn/Al; Mg/Al, Ga hydrotalcite-like compounds used as catalysts for cycloxene epoxidation[J]. Appl Catal A: Gen,2008,341(1/2):50−57. doi: 10.1016/j.apcata.2007.12.022 [24] BEHRENS M, KASATKIN I, KUHL S, WEINBERG G. Phase-pure Cu, Zn, Al hydrotalcite-like materials as precursors for copper rich Cu/ZnO/Al2O3 catalysts[J]. Chem Mater,2010,22(2):386−397. [25] KUMAR P, SRIVASTAVA V C, MISHRA I M. Dimethyl carbonate synthesis from propylene carbonate with methanol using Cu-Zn-Al catalyst[J]. Energy Fuels,2015,29(4):2664−2675. doi: 10.1021/ef502856z [26] WU G D, WANG X L, WEI W, SUN Y H. Fluorine-modified Mg-Al mixed oxides: A solid base with variable basic sites and tunable basicity[J]. Appl Catal A: Gen,2010,377(1/2):107−113. doi: 10.1016/j.apcata.2010.01.023 [27] WEI T, WANG M H, WEI W, SUN Y H, ZHONG B. Effect of base strength and basicity on catalytic behavior of solid bases for synthesis of dimethyl carbonate from propylene carbonate and methanol[J]. Fuel Process Technol,2003,83(1/3):175−182. doi: 10.1016/S0378-3820(03)00065-1 -




 下载:
下载: