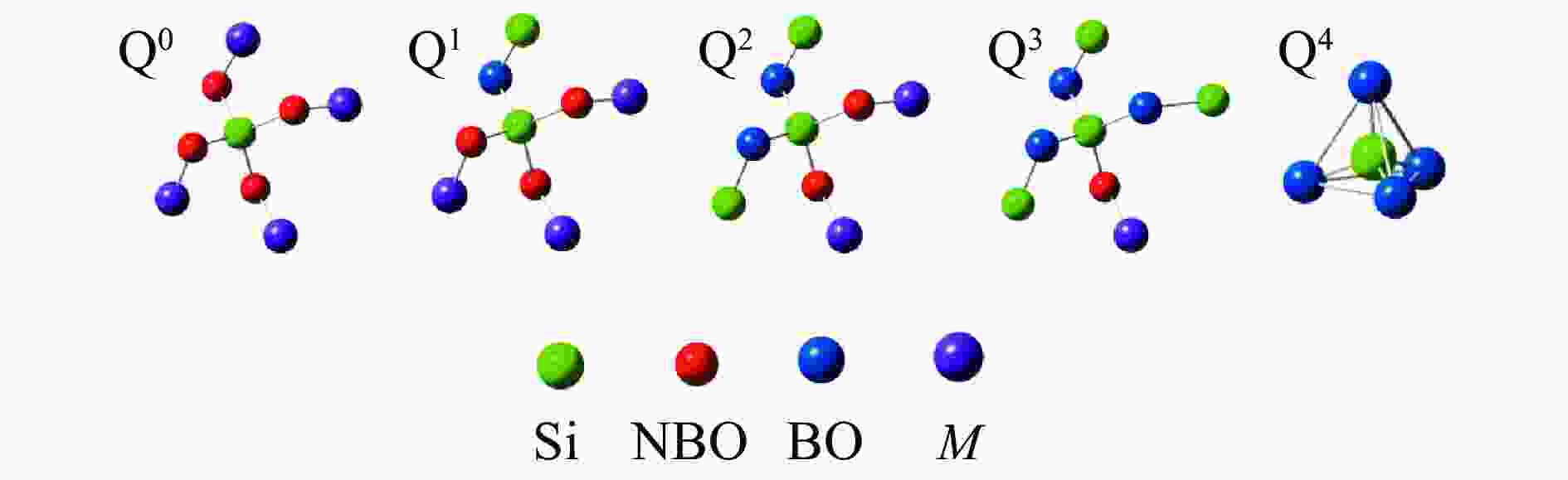
Modification of ash fusion behavior of high ash fusion temperature (AFT) coal by textile dyeing sludge addition and its mechanism
-
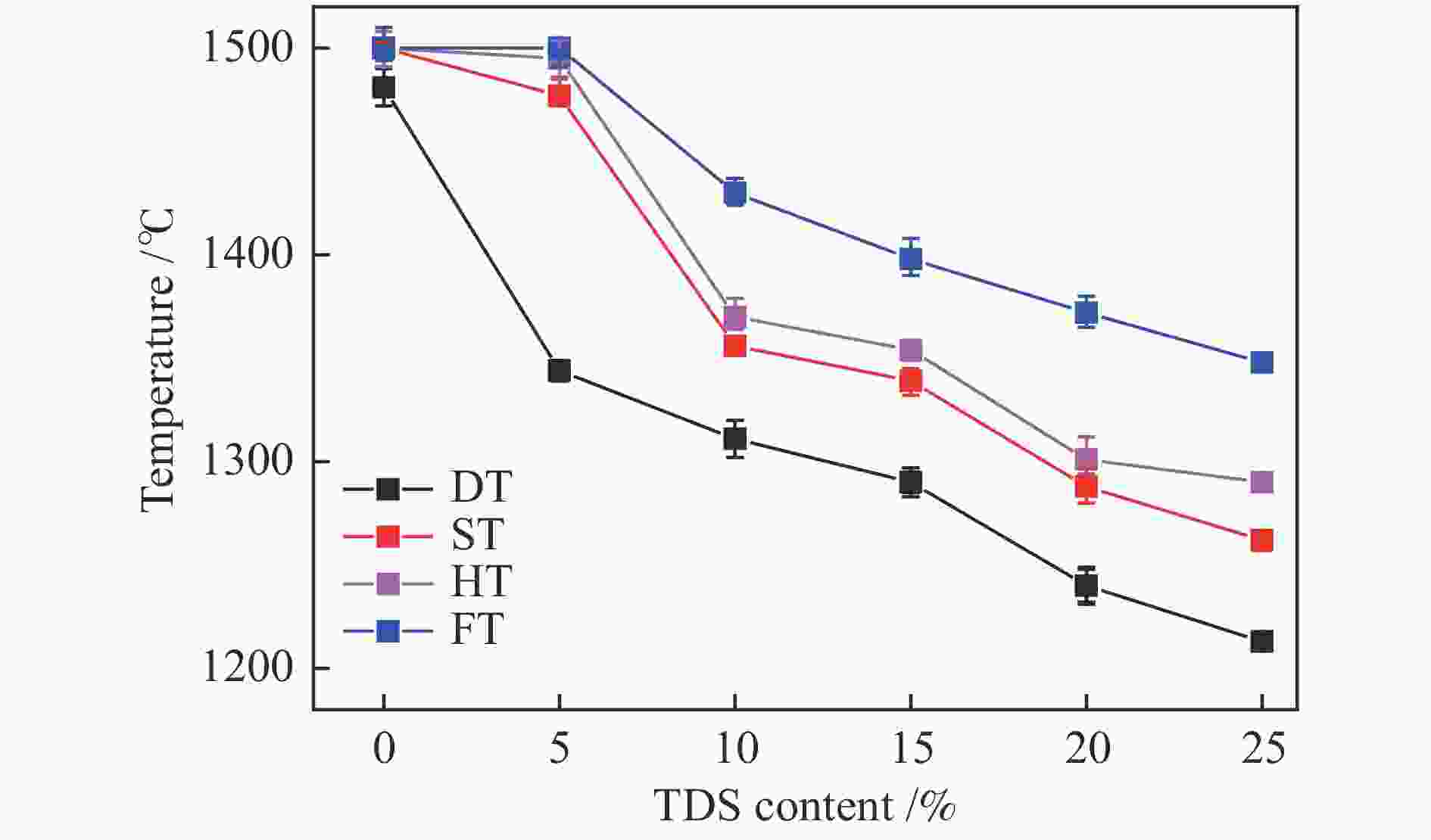
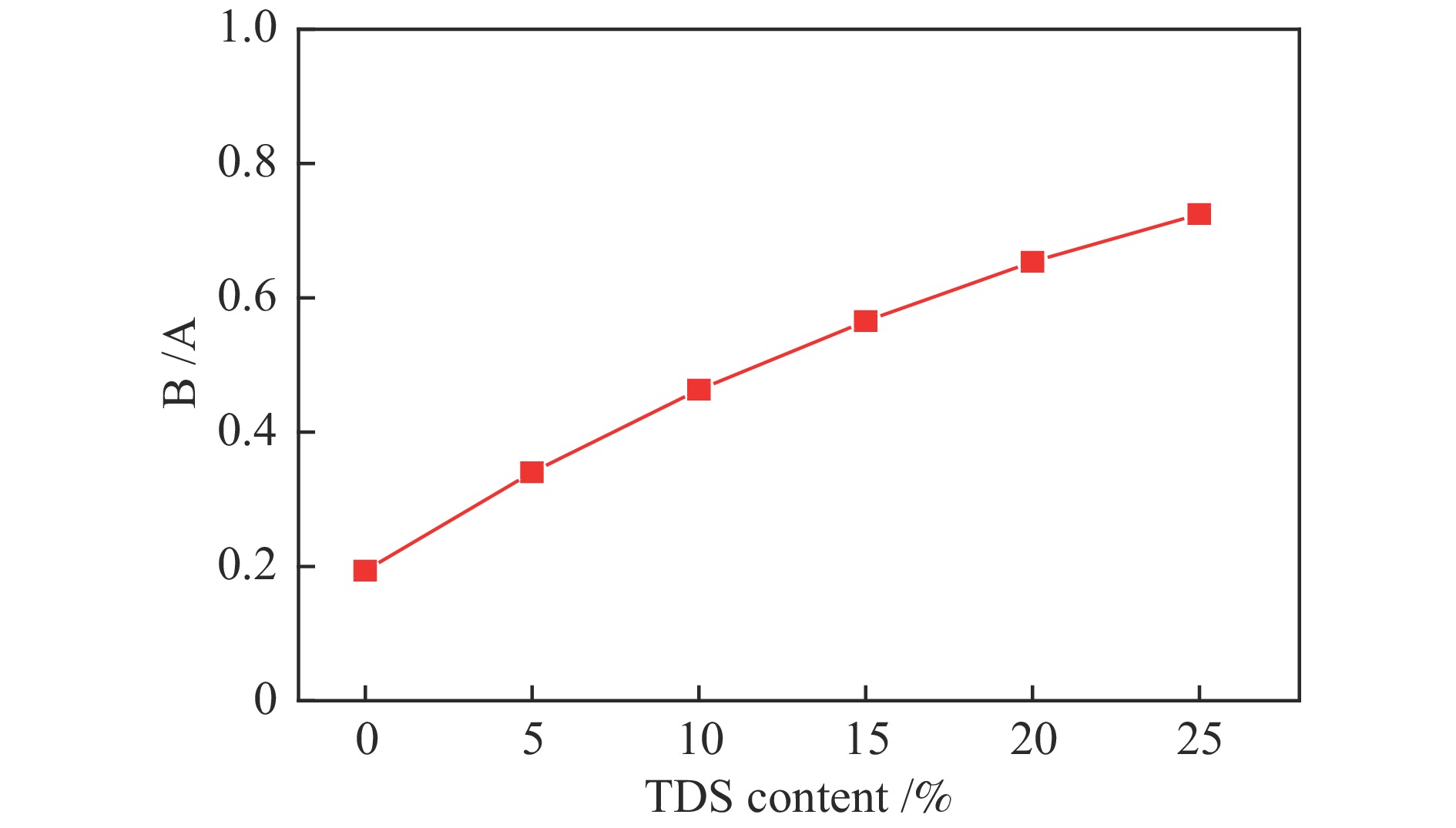
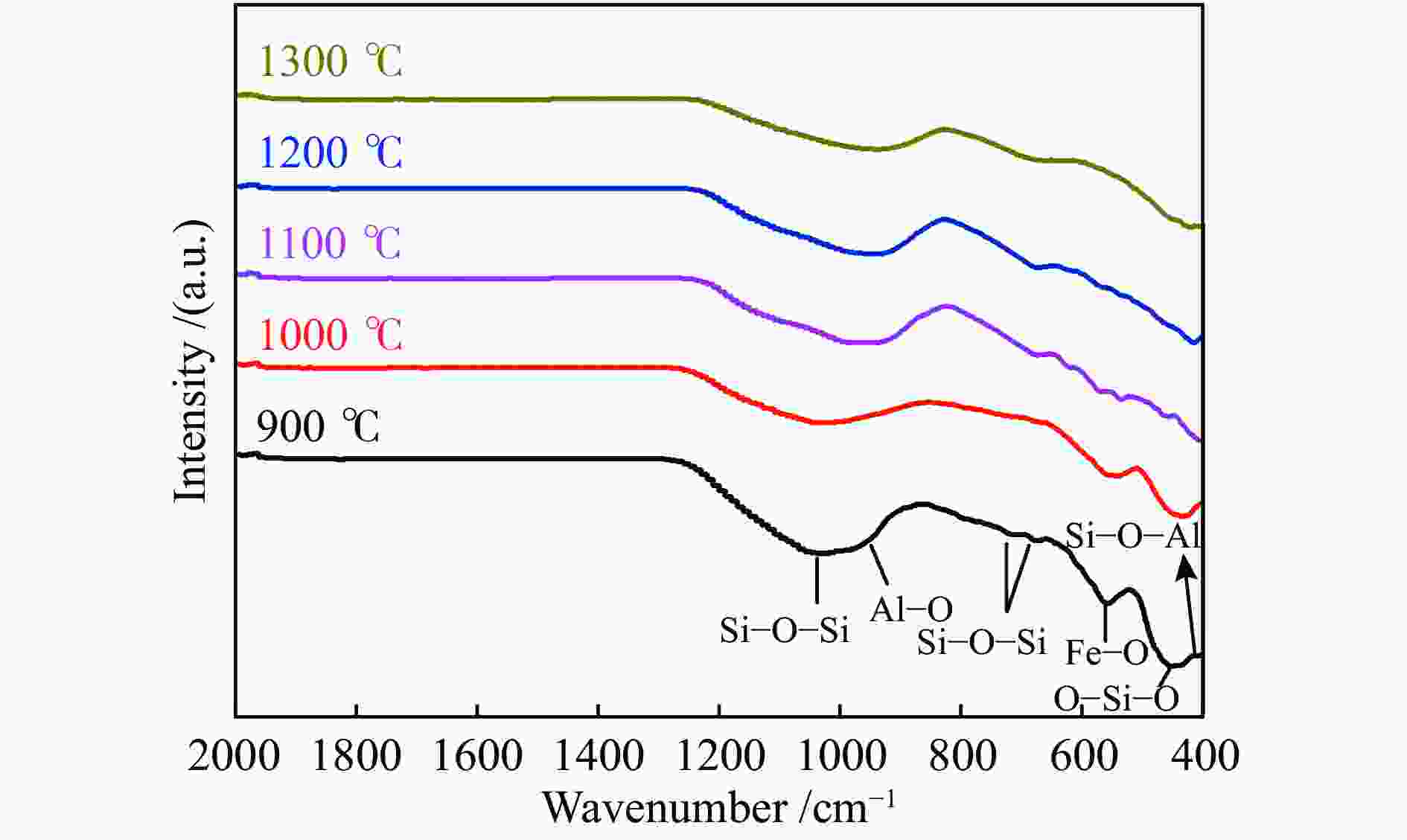
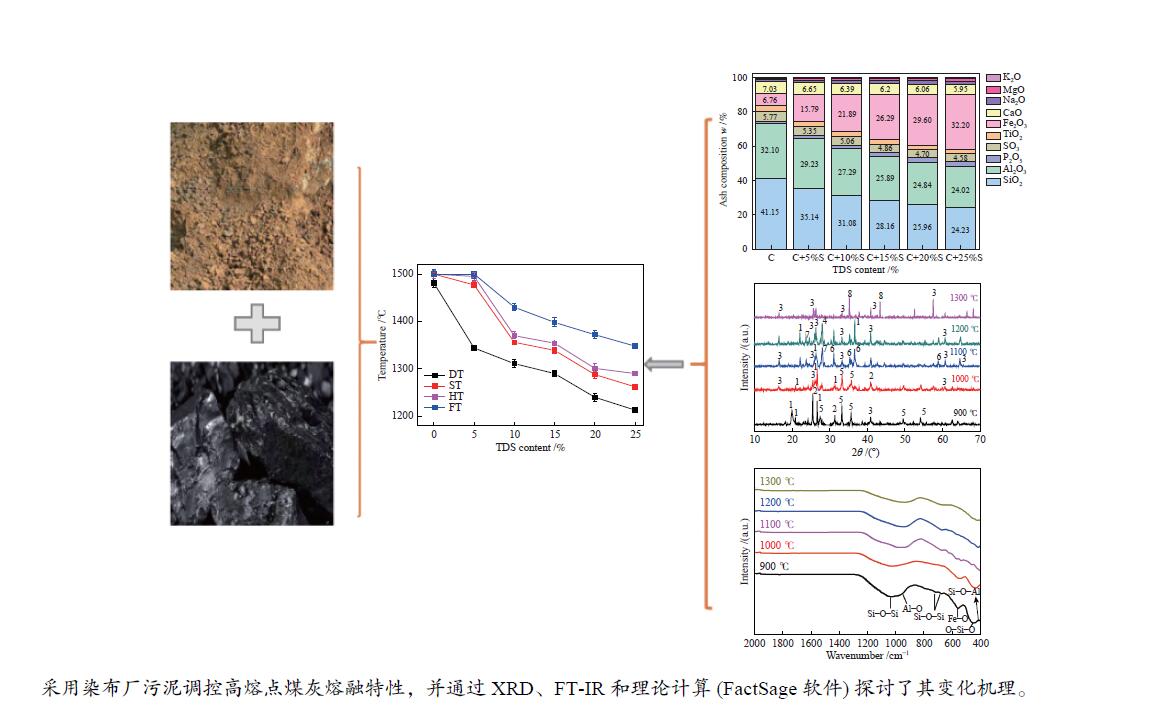
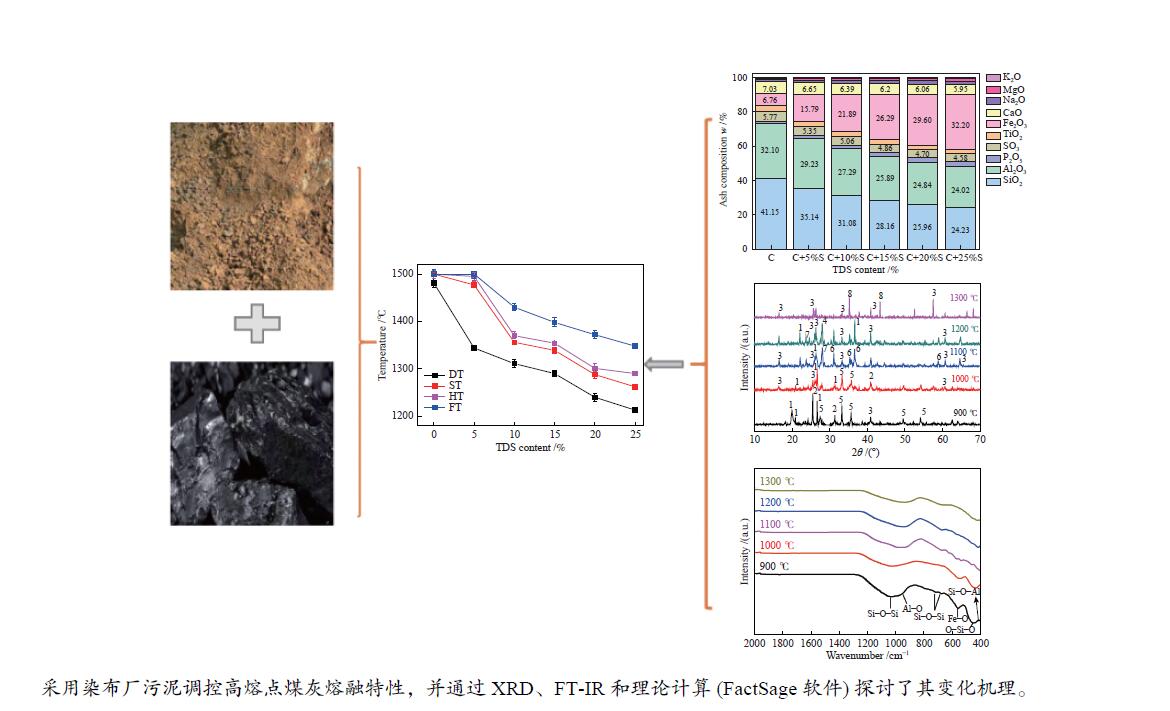
摘要: 为解决煤气流床(EFB)气化过程中的结渣问题,在还原气氛下研究了染布厂污泥(TDS)对高灰熔融温度(AFT)煤的熔融特性的影响。通过X射线衍射、傅里叶变换红外光谱(FT-IR)和FactSage计算研究其变化机理。结果表明,当TDS含量添加20%−25%时,流动温度降至1380 ℃以下,满足EFB气化的液态排渣要求。随着TDS含量的增加,低熔点矿物(如铁尖晶石、钙长石和钠长石)的形成导致AFT降低。网络结构的桥氧键被金属离子(如Fe2+、Ca2+、Na+)破坏,大量的非桥氧(NBO)键生成,导致硅酸盐网络结构疏松,AFT降低。Si−O−Si键和Si−O−Al键的峰值强度逐渐降低,Fe−O键和Si−O−M(M:Ca2+或Na+)键的振动增强被FT-IR证实了NBO的形成。FactSage计算的结果与实验中灰熔融行为具有一致性。Abstract: To address the slagging problem during coal entrained-flow bed (EFB) gasification, the influences of textile dyeing sludge (TDS) addition on the fusing characteristics of high ash fusion temperature (AFT) coal were explored under a reducing atmosphere. And the change mechanisms were investigated by X-ray diffraction, Fourier Transform Infrared Spectroscopy (FT-IR) and FactSage calculation. The results showed that the flow temperature of high ash fusion temperature (AFT) coal decreased below 1380 °C when the TDS addition reached 20%−25%, which met the requirements of liquid-slag removal for EFB gasification. With the content of TDS increasing, the formations of low-melting minerals (e.g., hercynite, anorthite, and albite) decreased AFT. The bridging oxygen bonds of the network structure were destroyed by metal ions (e.g., Fe2+, Ca2+, Na+), formation of much non-bridged oxygen (NBO) bonds relaxed the silicate network, thus decreasing the AFT. The formations of NBO bonds were confirmed by gradual decreases in the peak strengths of Si−O−Si and Si−O−Al bonds and intensified the vibration of Fe−O and Si−O−M ( M: Ca2+ or Na+) bonds. FactSage calculation results were in good agreement with the experimental ash fusion behavior.
-
Key words:
- ash fusion behaviors /
- textile dyeing sludge /
- high AFT coal /
- modification mechanisms
-
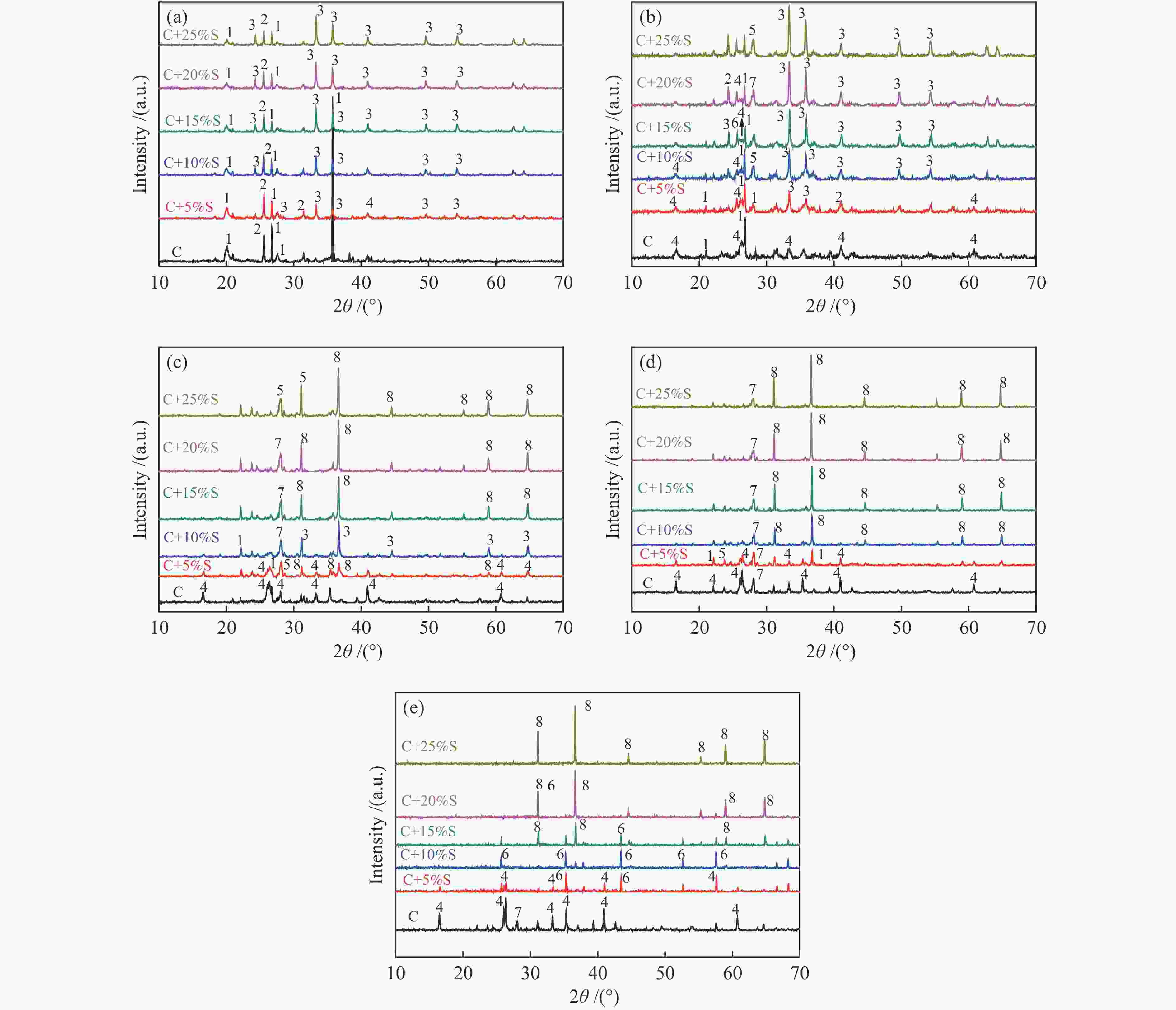
Figure 5 XRD patterns of coal ash and mixture ashes with different TDS ash mass ratios at different temperatures (a): C; (b): C+5%; (c): C+10%S; (d): C+15%S; (e): C+20%S; (f): C+25%S; 1: Quartz (SiO2); 2: Anhydrite (CaSO4); 3: Mullite (Al6Si2O13); 4: Anorthite (CaAl2Si2O8); 5: Hematite (Fe2O3); 6: Hercynite (FeAl2O4); 7: Albite (NaAlSi3O8); 8: Alumina (Al2O3)
Figure 6 XRD patterns of mixed ashes with different TDS ash mass ratios at 900 to 1300 °C (a): 900 °C; (b): 1000 °C; (c): 1100 °C; (d): 1200 °C; (e): 1300 °C 1: Quartz (SiO2); 2: Anhydrite (CaSO4); 3: Hematite (Fe2O3); 4: Mullite (Al6Si2O13); 5: Albite (NaAlSi3O8); 6: Alumina (Al2O3); 7: Anorthite (CaAl2Si2O8); 8: Hercynite (FeAl2O4)
Figure 9 Phase assemblage-temperature curves of coal ashes and mixture ash (a): C; (b): C+5%S; (c): C+10%S; (d): C+15%; (e): C+20%; (f): C+25% 1: Mullite (Al6Si2O13); 2: Albite (NaAlSi3O8); 3: Anorthite (CaAl2Si2O8); 4: Potassium Feldspar (KAlSi3O8); 5: Hercynite (FeAl2O4); 6: Calcium Phosphate (Ca5HO13P3); 7: Cordierite (Al4Fe2Si5O18); 8: Ferrous Sulfide (FeS); 9: Quartz (SiO2); 10: Titania (TiO2); 11: Alumina (Al2O3); 12: Ilmenite (FeTiO3); 13: Protopyroxene (FeAl2SiO6); 14: Titania Spinel (Fe2TiO4); 15: Fayalite (Fe2SiO4)
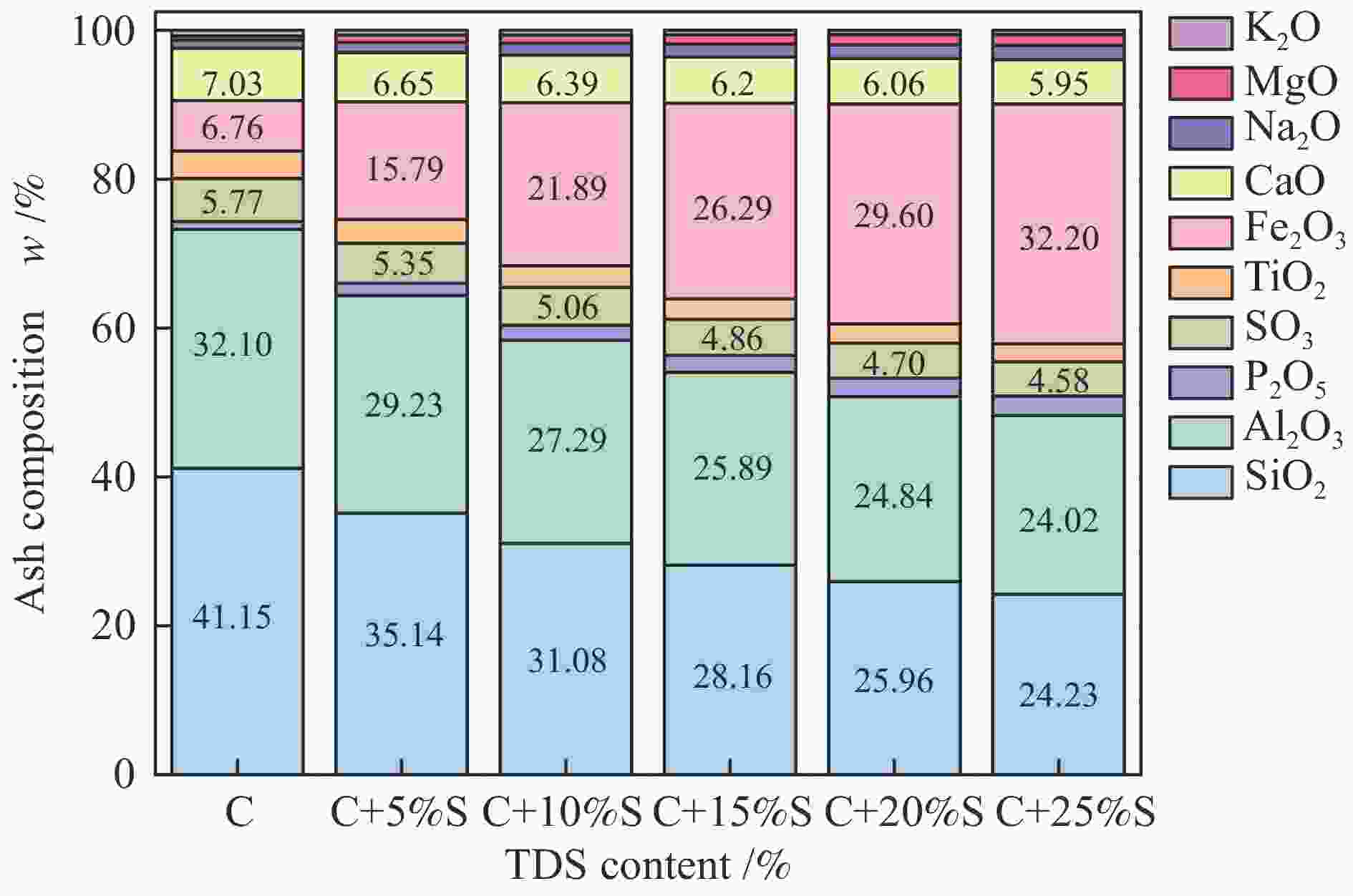
Table 1 Proximate and ultimate analyses of coal and TDS samples
Sample Proximate analysis wad/% Ultimate analysis wdaf/% Mad Vad Aad FCad C H O N S Coal 6.60 12.86 10.15 70.39 92.62 3.99 1.65 1.42 0.32 TDS 14.25 28.37 58.63 1.25 21.16 2.66 1.10 9.32 2.49 Chemical composition of the ash w/% SiO2 Al2O3 CaO Na2O Fe2O3 MgO P2O5 SO3 TiO2 K2O Coal 41.15 32.10 7.03 1.07 6.76 0.60 1.10 5.77 3.68 0.74 TDS 15.37 19.65 5.33 2.38 44.90 1.86 3.40 3.93 1.78 0.47 ad-air dry basis; daf- dry ash free basis; M-moisture; A-ash; V-volatile matter; FC-fixed carbon Table 2 AFTs of the coal and TDS
Sample Temperature/℃ DT ST HT FT Coal 1481 > 1500 > 1500 > 1500 TDS 1188 1235 1267 1321 -
[1] CAO X, KONG L X, BAI J, ZHAO H, GE Z F, LI H Z, BAI Z Q, LI W. Effect of water vapor on coal ash slag viscosity under gasification condition[J]. Fuel,2019,227:18−27. [2] DUAN W J, YU Q, LIU J X, HOU L M, XIE H Q, WANG X K, QIN Q. Characterizations of the hot blast furnace slag on coal gasification reaction[J]. Appl Therm Eng,2016,98:936−943. doi: 10.1016/j.applthermaleng.2015.12.029 [3] LI X M, ZHI L F, SHI W J, KONG L X, BAI J, YU J L, MARKUS R, STEFAN G, BERND M, LI W. Effect of K2O/Na2O on fusion behavior of coal ash with high silicon and aluminum level[J]. Fuel,2020,265:116964. doi: 10.1016/j.fuel.2019.116964 [4] WAN D C, LIANG Q F, GONG X, LIU H F, LIU X. Influence of coal blending on ash fusion property and viscosity[J]. Fuel,2017,189:15−22. doi: 10.1016/j.fuel.2016.10.050 [5] FAN G X, ZHANG M Y, PENG W J, ZHOU G L, DENG L J, CHAMG L P, CAO Y J, LI P. Clean products from coal gasification waste by flotation using waste engine oil as collector: Synergetic cleaner disposal of wastes[J]. J Clean Prod,2021,286:124943. doi: 10.1016/j.jclepro.2020.124943 [6] LI F H, FAN H L, WANG X C, WANG T, FANG Y T. Influences of phosphorus on ash fusion characteristics of coal and its regulation mechanism[J]. Fuel,2019,239:1338−1350. doi: 10.1016/j.fuel.2018.11.004 [7] MA X W, LI F H, MA M J, HUANG S X, JI S H, FANG Y T. Regulation of ash fusibility characteristics for high-ash-fusion-temperature coal by bean straw addition[J]. Energy Fuels,2018,32(6):6678−6688. doi: 10.1021/acs.energyfuels.8b01013 [8] LI F H, LI Y, ZHAO C Y, FAN H L, XU M L, GUO Q Q, GUO M X, WANG Z Q, HUANG J J, FANG Y T. Investigation on ash-fusion characteristics of livestock manure and low-rank coals[J]. Energy Fuels,2020,34:5804−5812. doi: 10.1021/acs.energyfuels.0c00587 [9] HE C, ILYUSHECHKIN A, BAI J, HLA S S, KONG L X, LI W. Viscosity and crystallisation behaviour of coal ash slag from the primary phase of anorthite[J]. Fuel Process Technol,2021,213:106680. doi: 10.1016/j.fuproc.2020.106680 [10] JAKUB L, MAREK B, MARTIN L, HANA L, PAVEL M, PATRIK E. An overview of slagging and fouling indicators and their applicability to biomass fuels[J]. Fuel Process Technol,2021,217:106804. doi: 10.1016/j.fuproc.2021.106804 [11] XU J, SONG X D, YU G S, DU C H. Investigating the effect of flux on ash fusibility of high-calcium coal[J]. ACS Omega,2020,5(20):11361−11368. doi: 10.1021/acsomega.0c00320 [12] WANG H, CHENG L H, PU J L, ZHAO J G. Melting characteristics of coal ash and properties of fly ash to understand the slag formation in the shell gasifier[J]. Energy Fuels,2021,6(24):16066−16075. [13] MA X W, LI F H, MA M J, FANG Y T. Fusion characteristics of blended ash from Changzhi coal and biomass[J]. J Fuel Chem Technol,2018,46(02):129−137. doi: 10.1016/S1872-5813(18)30007-0 [14] YAO X W, ZHENG Y, ZHOU H D, XU K, XU Q W, LI L. Effects of biomass blending, ashing temperature and potassium addition on ash sintering behaviour during co-firing of pine sawdust with a Chinese anthracite[J]. Renew Energy,2020,147:2309−2320. doi: 10.1016/j.renene.2019.10.047 [15] XIE L C, LI F H, XUE Z M, XU L, MA X X. Influence of coal blending on ash fusion characteristics for coal with high ash fusion temperature[J]. J Fuel Chem Technol,2016,44(12):1430−1439. [16] WANG Y B, LI L Y, AN Q W, WANG M, TAN H Z, LI P, PENG J H. Effect of Ca3(PO4)2 additive on the slagging behavior during the cofiring of high-sodium coal and iron-rich coal[J]. Fuel Process Technol,2021,222:106965. doi: 10.1016/j.fuproc.2021.106965 [17] DAI X, BAI J, HUANG Q, LIU Z, BAI X J, LIN C T, LI W, GUO W, WEN X D, DU S Y. Coal ash fusion properties from molecular dynamics simulation: the role of calcium oxide[J]. Fuel,2018,216:760−767. doi: 10.1016/j.fuel.2017.12.048 [18] WANG J J, GUO Q H, WEI J T, LIU X, WANG X J, YU G S. Understanding the influence of iron on fluidity and crystallization characteristics of synthetic coal slags[J]. Fuel Process Technol,2020,209:106532. doi: 10.1016/j.fuproc.2020.106532 [19] HU S H, ZHANG J K, CEN K F, ZHOU H. Ash deposition characteristics during co-combustion of coal and sawdust[J]. J Fuel Chem Technol,2020,48(9):1055−1062. [20] XIAO H X, LI F H, LIU Q R, JI S H, FAN H L, XU M L, GUO Q Q, MA M J, MA X W. Modification of ash fusion behavior of coal with high ash fusion temperature by red mud addition[J]. Fuel,2017,192:121−127. doi: 10.1016/j.fuel.2016.12.012 [21] MA X W, LI F H, MA M J, FANG Y T. Investigation on blended ash fusibility characteristics of biomass and coal with high silica-alumina[J]. Energy Fuels,2017,31:7941−7951. doi: 10.1021/acs.energyfuels.7b01070 [22] ISAM J, IDOWU A, SHABBAR R, CHAOUKI G. A review of recent developments and future prospects in gasification systems and their modeling[J]. Renewable Sustainable Energy Rev,2021,138:110505. doi: 10.1016/j.rser.2020.110505 [23] RAN C M, LIU Y, SIDDIQUI A R, SIYAL A A, MAO X, KANG Q H, FU J, AO W Y, DAI J J. Pyrolysis of textile dyeing sludge in fluidized bed: Analysis of products, and migration and distribution of heavy metals[J]. J Clean Prod,2019,241:118308. doi: 10.1016/j.jclepro.2019.118308 [24] ZHUO Z X, LIU J Y, SUN S Y, SUN J, KUO J H, CHANG K L, FU J W, Wang Y J. Thermogravimetric characteristics of textile dyeing sludge, coal and their blend in N2/O2 and CO2/O2 atmospheres[J]. Appl Therm Eng,2017,111:87−94. doi: 10.1016/j.applthermaleng.2016.09.089 [25] LIU Y, RAN C M, SIYAL A A, SONG Y M, JIANG Z H, DAI J J, CHTAEVA P, FU J, AO W Y, Deng Z Y, ZHANG T H. Comparative study for fluidized bed pyrolysis of textile dyeing sludge and municipal sewage sludge[J]. J Hazard Mater,2020,396:122619. doi: 10.1016/j.jhazmat.2020.122619 [26] ZHANG J H, ZOU H H, LIU J Y, EVRENDILEK F, XIE W M, HE Y, BUYUKADA M. Comparative (co-)pyrolytic performances and by-products of textile dyeing sludge and cattle manure: Deeper insights from Py-GC/MS, TG-FTIR, 2D-COS and PCA analyses[J]. J Hazard Mater,2021,401:23276. [27] XIE C D, LIU J Y, BUYUKADA M, EVRENDILEK F, SAMAKSAMAN U, KUO J H, OZYURT O. Parametric assessment of stochastic variability in co-combustion of textile dyeing sludge and shaddock peel[J]. Waste Manage,2019,96:128−135. doi: 10.1016/j.wasman.2019.07.010 [28] XIE W H, HUANG J L, LIU J Y, ZHAO Y J, CHANG K L, KUO J H, HE Y, SUN J, ZHENG L, XIE W M, SUN S Y, BUYUKADA M, EVRENDILEK F. Assessing thermal behaviors and kinetics of (co-)combustion of textile dyeing sludge and sugarcane bagasse[J]. Appl Therm Eng,2018,131:874−883. doi: 10.1016/j.applthermaleng.2017.11.025 [29] SANSANIWAL S K, PAL K, ROSEN M A, TYAGI S. K. Recent advances in the development of biomass gasification technology: A comprehensive review[J]. Renewable Sustainable Energy Rev,2017,72:363−384. doi: 10.1016/j.rser.2017.01.038 [30] WANG M L, MAO M M, ZHANG M, WEN G D, YANG Q W, SU B G, REN Q L. Highly efficient treatment of textile dyeing sludge by CO2 thermal plasma gasification[J]. Waste Manage,2019,90:29−36. doi: 10.1016/j.wasman.2019.04.025 [31] FOLGUERAS M B, ALONSO M, FOLGUERAS J R. Effect of sludge addition to coal on Na, K and S volatilisation in ashing process and ash fusibility[J]. Fuel Process Technol,2015,138:714−723. doi: 10.1016/j.fuproc.2015.07.013 [32] FOLGUERAS M B, ALONSO M, FOLGUERAS J R. Modification of lignite ash fusion temperatures by the addition of different types of sewage sludge[J]. Fuel Process Technol,2015,131:348−355. doi: 10.1016/j.fuproc.2014.12.002 [33] SCHWITALLA D, REINMÖLLER M, FORMAN C, WOLFERSDORF C, GOOTZ M, BAI J, GUHLA S, NEUROTHC M, MEYER B. Ash and slag properties for co-gasification of sewage sludge and coal: An experimentally validated modeling approach[J]. Fuel Process Technol,2018,175:1−9. doi: 10.1016/j.fuproc.2018.02.026 [34] WANG T, FU T M, CHEN K, CHENG R S, CHEN S, LIU J X, MEI M, LI J P, XUE Y J. Co-combustion behavior of dyeing sludge and rice husk by using TG-MS: Thermal conversion, gas evolution, and kinetic analyses[J]. Bioresour Technol,2020,311:123527. doi: 10.1016/j.biortech.2020.123527 [35] SONG Y Y, HU J W, LIU J Y, BUYUKADA M, EVRENDILEK F. CO2-assisted co-pyrolysis of textile dyeing sludge and hyperaccumulator biomass: Dynamic and comparative analyses of evolved gases, bio-oils, biochars, and reaction mechanisms[J]. J Hazard Mater,2020,400:123190. doi: 10.1016/j.jhazmat.2020.123190 [36] DUCHESNE M A, HUGHES R W, LU D Y, MCCALDEN DJ, ANTHONY E J, MACCHI A. Fate of inorganic matter in entrained-flow slagging gasifiers: Pilot plant testing[J]. Fuel Process Technol,2014,125:18−33. [37] ZHANG L M, WANG J F, SONG X D, BAI Y H, YAO M, YU G S. Influence of biomass ash additive on fusion characteristics of high-silicon aluminum coal ash[J]. Fuel,2020,282:118876. doi: 10.1016/j.fuel.2020.118876 [38] PETER Y H. Viscous deformation as a measure of heat work during coal ash fusibility testing[J]. Fuel,2020,281:118723. doi: 10.1016/j.fuel.2020.118723 [39] LI M, LI F H, LIU Q R, FANG Y T, XIAO H X. Regulation of ash fusibility for high ash-fusion-temperature (AFT) coal by industrial sludge addition[J]. Fuel,2019,244:91−103. doi: 10.1016/j.fuel.2019.01.161 [40] FAN H L, LI F H, GUO Q Q, GUO M X. Effect of biomass ash on initial sintering and fusion characteristics of high melting coal ash[J]. J Energy Inst,2021,94:129−138. doi: 10.1016/j.joei.2020.11.008 [41] YAN T G, BAI J, KONG L X, BAI Z Q, LI W, XU J. Effect of SiO2/Al2O3 on fusion behavior of coal ash at high temperature[J]. Fuel,2017,193(1):275−283. [42] REINMÖLLER M, SCHREINER M, GUHL S, NEUROTH M, MEYER B. Formation and transformation of mineral phases in various fuels studied by different ashing methods[J]. Fuel,2020,202:641−649. [43] ZHOU H, MA W C. An experimental study on the effects of adding biomass ashes on ash sintering behavior of Zhundong coal[J]. Appl Therm Eng,2017,126:689−701. [44] LI F H, FAN H L, FANG Y T. Investigation on the regulation mechanism of ash fusion characteristics in coal blending[J]. Energy Fuels,2017,31:379−386. doi: 10.1021/acs.energyfuels.6b02539 [45] GE Z F, KONG L X, BAI J, Chen X D, HE C, LI H Z, BAI Z Q, LI P, LI W. Effect of CaO/Na2O on slag viscosity behavior under entrained flow gasification conditions[J]. Fuel Process Technol,2018,181:352−360. doi: 10.1016/j.fuproc.2018.10.002 [46] GE Z, KONG L X, BAI J, ZHAO H L, CAO X, LI H Z, BAI Z Q, MEYER B, GUHLD S, LI P, LI W. Effect of CaO/Fe2O3 ratio on slag viscosity behavior under entrained flow gasification conditions[J]. Fuel,2019,258:116129. doi: 10.1016/j.fuel.2019.116129 [47] JIANG J Y, ZHANG S, LONGHURST P, YANG W H, ZHENG S J. Molecular structure characterization of bituminous coal in Northern China via XRD, Raman and FTIR spectroscopy[J]. Spectrochim Acta Part A: Mol Biomol Spectrosc,2021,255:119724. doi: 10.1016/j.saa.2021.119724 [48] POPOV V K, POSOKHOV Y M, RACHEV I L. IR spectroscopy of coal 2. Analysis of ash content from the difference spectra of minerals[J]. Coke Chem,2009,52(12):519−522. doi: 10.3103/S1068364X09120035 [49] MOZGAWA W, KRÓL M, DYCZEK J, DEJA J. Investigation of the coal fly ashes using IR spectroscopy[J]. Spectrochim Acta Part A:Mol Biomol Spectrosc,2014,132:889−894. doi: 10.1016/j.saa.2014.05.052 [50] BAI J, LI W, LI B Q. Characterization of low-temperature coal ash behaviors at high temperatures under reducing atmosphere[J]. Fuel,2008,87:583−591. doi: 10.1016/j.fuel.2007.02.010 [51] LI Y, LI F H, MA M J, YU B, ZHAO C Y, FANG Y T. Prediction of ash flow temperature based on liquid phase mass fraction by FactSage[J]. J Energy Inst,2020,93(6):2228−2231. doi: 10.1016/j.joei.2020.06.004 [52] SCHWITALLA D H, GUHL S, LAABS M, REINMÖLLER M, BAI J, MEYER B. Thermochemical and analytical approach to describe secondary slag phase formation and local process conditions in a full-scale BGL gasifier[J]. Fuel Process Technol,2021,217:106833. doi: 10.1016/j.fuproc.2021.106833 [53] FAN H L, LI F H. Ash fusion temperature regulation mechanism of Xiangyang coal by coal blending[J]. J Therm Anal Calorim,2020,139(3):2055−2066. doi: 10.1007/s10973-019-08602-0 [54] KONG L X, BAI J, LI W, WEN X D, LI X M, BAI Z Q, GUO Z X, LI H Z. The internal and external factor on coal ash slag viscosity at high temperatures, Part 3: Effect of CaO on the pattern of viscosity-temperature curves of slag[J]. Fuel,2016,179:10−16. doi: 10.1016/j.fuel.2016.03.063 [55] LI F, LI M, ZHAO H M, FANG Y T. Experimental investigation of ash deposition behaviour modification of straws by lignite addition[J]. Appl Therm Eng,2017,125:134−144. doi: 10.1016/j.applthermaleng.2017.06.144 -




 下载:
下载: