Preparation of core-shell ZSM-5@Beta molecular sieve and catalytic alkylation to 2,6-dimethylnaphthalene
-
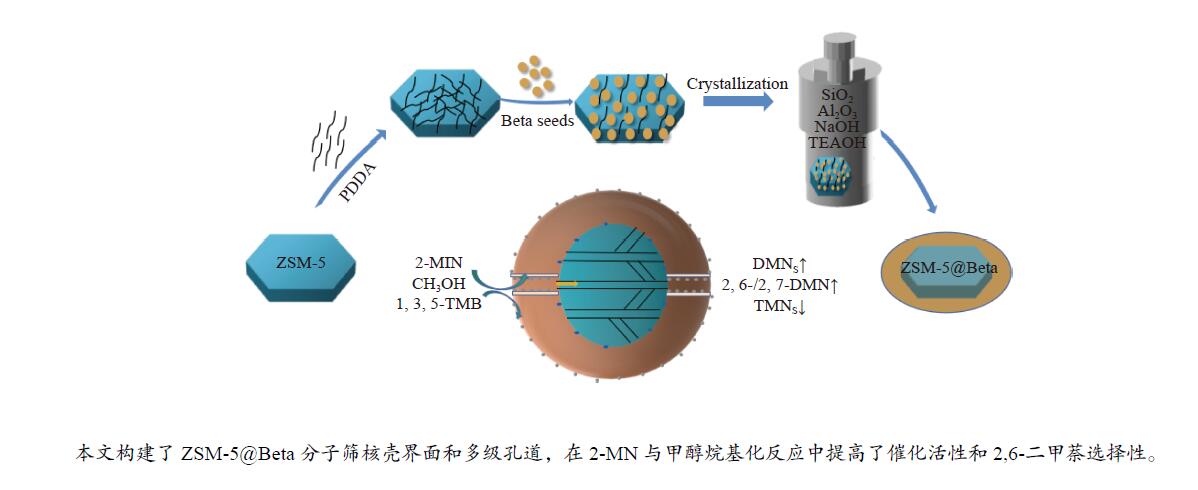
摘要: 以ZSM-5分子筛为核相,通过聚二烯丙基二甲基氯化铵(PDDA)偶连纳米Beta分子筛晶种,经动态水热合成方法制备了ZSM-5@Beta核壳二元复合分子筛。采用XRD、N2吸附-脱附、SEM、TEM、ICP、NH3-TPD及Py-FTIR等手段对复合分子筛的结构和物性进行了表征,考察了复合分子筛催化2-甲萘(2-MN)与甲醇烷基化合成2,6-二甲基萘(2,6-DMN)的催化性能。结果表明,采用该方法制备出粒径约为500 nm的核壳结构ZSM-5@Beta复合分子筛。与机械混合二元分子筛相比,核壳结构材料具有更高的比表面积和外表面积,并降低了酸强度和强酸中心密度。通过限域催化理念、核壳界面和多级孔道的构建,借助12元环壳相Beta分子筛提高催化活性,利用10元环核相ZSM-5的择形催化作用提高催化选择性。在2-MN与甲醇烷基化反应中提高了2-MN的转化率和2,6-DMN的选择性,产物中2,6-/2,7-DMN比达到1.35,2,6-DMN收率达到4.29%。Abstract: ZSM-5@Beta core-shell molecular sieve was prepared by dynamic hydrothermal synthesis method using ZSM-5 adhered Beta seed crystals as the core phase, and polydiallyl dimethyl ammonium chloride (PDDA) was used as a coupling agent to adhere Beta seed crystals on the surface of ZSM-5. The structure and physical properties of composite molecular sieves were characterized by XRD, N2 adsorption-desorption, SEM, TEM, ICP, NH3-TPD and Py-FTIR. The catalytic performance of composite molecular sieves for alkylation of 2-methylnaphthalene (2-MN) with methanol was investigated. The results showed that ZSM-5@Beta composite molecular sieve prepared by this method had a core-shell structure, and the particle size was about 500 nm. Compared with mechanically mixed binary molecular sieve, core-shell molecular sieve had higher specific surface area and external surface area, lower acid strength and stronger acid center density. Through the construction of core-shell interface and hierarchical porous, the catalytic activity was improved by shell phase Beta molecular sieve with 12-membered ring channel, and the catalytic selectivity was improved by core phase ZSM-5 molecular sieve with 10-membered ring channel in the alkylation reaction of 2-MN with methanol. The 2,6-/2,7-DMN ratio in the products reached 1.35, and the yield of 2,6-DMN reached 4.29%.
-
Key words:
- ZSM-5 /
- Beta /
- core-shell structure /
- 2,6-dimethylnaphthalene
-
表 1 样品的孔结构参数
Table 1 Pore structure parameter of the samples
Sample ABET/(m2·g−1) Aext/(m2·g−1) vtotal/(cm3·g−1) vmic/(cm3·g−1) vmes/(cm3·g−1) Ratio(Aext/ABET) HBeta 656.40 142.79 0.51 0.21 0.33 0.28 HZSM-5 297.10 16.89 0.19 0.12 0.07 0.06 HZSM-5@Beta-m 449.36 105.88 0.44 0.14 0.31 0.24 HZSM-5@Beta-cs 633.06 169.11 0.45 0.19 0.27 0.27 表 2 元素组成、NH3-TPD和Py-FTIR谱图量化
Table 2 Element composition, the quantitative results of NH3-TPD profile and Py-FTIR spectrum
Sample SiO2/Al2O3 Desorption peak area of NH3-TPD spectruma Acidityb/(μmol∙g−1) Brønsted/Lewis 100−250 ℃ 250−400 ℃ 400−550 ℃ Total Lewis Brønsted HBeta 30 924 1459 342 2725 202 296 1.47 HZSM-5 24.2 1506 617 767 2890 25 100 4.00 HZSM-5@Beta-m 27.5 909 1101 967 2977 77 254 3.30 HZSM-5@Beta-cs 25.8 1018 959 247 2224 133 222 1.67 a: Peak area Gaussian fitting of NH3-TPD spectrum;
b: Quantitative results of Py-FTIR spectrum表 3 2-MN与甲醇烷基化反应的催化性能a
Table 3 Catalytic performance of 2-MN alkylation with methanol over molecular sievesa
Catalyst HBeta HZSM-5 HZSM-5@Beta-m HZSM-5@Beta-cs Conversion x/% 2-MN 90.94 31.34 55.18 61.62 Distributionb s/% NA 1.84 3.95 8.17 4.47 DMNs 44.37 27.44 43.54 47.15 TMNs 45.46 0.00 0.00 15.96 1-MN 8.33 68.61 48.29 32.42 β,β-DMN/DMNs 30.08 52.69 38.57 34.15 2,6-DMN/DMNs 11.73 16.24 16.46 12.85 2,6-/2,7-DMN 0.90 0.68 0.99 0.89 2,6-DMN selectivityc s/% 2.77 4.52 5.52 6.01 2,6-DMN yieldd w/% 2.52 1.42 3.04 3.70 a: t = 400 ℃, WHSV2-MN=1 h−1, n2-MN/nCH3OH/n1,3,5-TMB = 1∶4∶4, p = 0.2 MPa, TOS = 1 h;
b: Mole of 2-MN derivatives in products × 100/mole of 2-MN in raw materials, including NP(Naphthalene), DMNs(Dimethyl naphthalene) TMNs(Trimethyl naphthalene) and 1-MN(1-methylnaphthalene);
c: Mole of 2,6-DMN in products × 100/(mole of 2-MN in raw materials-mole of 2-MN in products);
d: Mole of 2,6-DMN in products×100/mole of 2-MN in raw materials -
[1] WANG X X, WEN J, ZHANG W, ZHAO L F, WEI W. Synthesis of 2, 6-dimethyl naphthalene over SAPO-11 zeolite[J]. China Pet Process Petrochem Technol,2012,41(11):1282−1287. [2] SANHOOB M A, MURAZA O, TAGO T, TANIGUCHI T, WATAANBE G, MASUDA T. Development of mesoporous ZSM-12 zeolite and its application in alkylation of 2-methylnaphthalene[J]. Res Chem Intermed,2016,42(7):1−12. [3] CHOBSA-ARD A, KRAIKUL N, RANGSUNVIGIT P, KULPRATHIPANJA S. Influences of solvents on the production of high purity 2, 6-dimethylnaphthalene via catalytic isomerization and adsorptive separation[J]. Chem Eng J,2008,139(1):78−83. doi: 10.1016/j.cej.2007.07.086 [4] GULEC F, NIFTALIYEVA A, KARADUMAN A. Selective synthesis of 2, 6-triad dimethylnaphthalene isomers by disproportionation of 2-methylnaphthalene over mesoporous MCM-41[J]. Res Chem Intermed,2018,44:7205−7218. doi: 10.1007/s11164-018-3551-5 [5] LI H J, GONG Q, LIANA H, DING L F, HU Z H, ZHU Z R. Transalkylation of C10 aromatics with 2-methylnaphthalene for 2, 6-dimethylnaphthalene synthesis: High-efficiently shape-selective & synergistic catalysis over a multifunctional SiO2-Mo-HBeta catalyst[J]. J Catal,2019,378:144−152. doi: 10.1016/j.jcat.2019.08.030 [6] WATANABE G, NAKASAKA Y, TANIGUCHI T, YOSHIKAWA T, TAGO T, MASUDA T. Kinetic studies on high-pressure methylation of 2-methylnaphthalene over MTW-type zeolite with different crystal sizes[J]. Chem Eng J,2016,312:288−295. [7] 王亚涛, 张新异, 房承宣, 靳立军, 郭学华, 胡浩权. 中空ZSM-5分子筛的制备及其在2-甲基萘烷基化合成2, 6-二甲基萘中的应用[J]. 石油化工,2012,41(12):1351−1356. doi: 10.3969/j.issn.1000-8144.2012.12.003WANG Ya-tao, ZHANG Xin-yi, FANG Chen-xuan, JIN Li-jun, HU Hao-quan. Preparation of hollow ZSM-5 and its application in methylation of 2-methylnaphthalene to 2, 6-dimethylnaphthalene[J]. Chin Petrochem Technol,2012,41(12):1351−1356. doi: 10.3969/j.issn.1000-8144.2012.12.003 [8] PARK J N, WANG J, HONG S I, LEE C W. Effect of dealumination of zeolite catalysts on methylation of 2–methylnaphthalene in a high-pressure fixed-bed flow reactor[J]. Appl Catal A: Gen,2005,292:68−75. doi: 10.1016/j.apcata.2005.05.039 [9] LUAN S, JIN L J, GUO X, YU Y, WANG Y. Synthesis of mesoporous ZSM-5 zeolite and its application in alkylation of 2-methylnaphthalene with methanol[J]. Acta Pet Sin (Pet Process Sect),2014,30(2):204−210. [10] YE Z, FENG J P, LYU Z J, LI X K. Improved stability and shape selectivity of 2, 6-dimethylnaphthalene by methylation of naphthalene with methanol on modified zeolites[J]. Mod Res Catal,2014,3(2):19−25. doi: 10.4236/mrc.2014.32004 [11] MILLINI R, FRIGERIO F, BELLUSSI G, PAZZUCONI G, PEREGO C, POLLESEL P, ROMANO U. A priori selection of shape-selective zeolite catalysts for the synthesis of 2, 6-dimethylnaphthalene[J]. J Catal,2003,217(2):298−309. doi: 10.1016/S0021-9517(03)00071-X [12] OZERENKO A A, GYULMALIEV A M, GAGARIN S G. Factorial analysis in selecting the zeolite catalyst for 2, 6-dimethylnaphthalene synthesis[J]. Coke Chem,2007,50(7):200−206. doi: 10.3103/S1068364X07070046 [13] SONG C S, SHEN J P, REDDY K M, SUN L, LILLWITZ L D. Shape-selective Fe-MFI catalyst for synthesis of 2, 6-dimethylnaphthalene by methylation with methanol[J]. Stud Surf Sci Catal,2007,170(7):1275−1282. [14] FATIH G, FAROOQ S, ALI K. Catalytic performance of Cu- and Zr-modified beta zeolite catalysts in the methylation of 2-methylnaphthalene[J]. Pet Sci,2019,(1):161−172. [15] 张凤, 高志贤, 吾满江·艾力. Co/Hβ分子筛催化甲基萘烷基化的反应特征[J]. 工业催化,2011,19(9):63−68. doi: 10.3969/j.issn.1008-1143.2011.09.014ZHANG Feng, GAO Zhi-xian, WU Man-jiang·AI Li. Catalytic alkylation of methyl naphthalene by Co/Hβ zeolite[J]. Ind Catal,2011,19(9):63−68. doi: 10.3969/j.issn.1008-1143.2011.09.014 [16] JIN L J, ZHOU X J, HU H Q, MA B. Synthesis of 2, 6-dimethylnaphthalene by methylation of 2-methylnaphthalene on mesoporous ZSM-5 by desilication[J]. Catal Commun,2009,10(3):336−340. [17] LIANG Z. Methylation of 2-methylnaphthalene with methanol over NH4F and Pt modified HZSM-5 catalysts[J]. Chin J Chem Eng,2010,18(5):742−749. doi: 10.1016/S1004-9541(09)60123-3 [18] LI H J, GONG Q, LIANA H, HU Z H, ZHU Z R. New process for 2, 6-dimethylnaphthalene synthesis by using C10 aromatics as solvent and transmethylation-agentia: High-efficiency and peculiar subarea-catalysis over shape-selective ZSM-5/Beta catalyst[J]. Ind Eng Chem Res,2019,58(28):12593−12601. doi: 10.1021/acs.iecr.9b01596 [19] 刘晓芳. 核壳结构分子筛复合材料的合成、表征及应用[D]. 长春: 吉林大学, 2018.LIU XIao-fang. Synthesis、characterization and application of core-shell molecular sieve composites[D]. Changchun: Jilin University, 2018. [20] WANG X X, GUO F, WEI X X, LIU Z M, WANG C Y, GUO S Q, WANG Y Y, YU Y. Retraction Note to: Study on methylation of naphthalene with methanol over ZSM-5(core)/SAPO-11(shell) composite molecular sieve[J]. Russ J Appl Chem,2018,91(6):1002−1008. doi: 10.1134/S1070427218060198 [21] 张佩珊, 马波, 杨卫亚, 凌凤香, 沈智奇, 王少军, 候宇鑫. 核壳结构Beta/MCM-22双微孔复合分子筛的合成与表征[J]. 燃料化学学报,2014,42(10):1240−1245. doi: 10.3969/j.issn.0253-2409.2014.10.013ZHANG Pei-shan, MA Bo, YANG Wei-ya, LING Feng-xiang, SHEN Zhi-qi, WANG Shao-jun, HOU Yu-xin. Synthesis and characterization of core-shell Beta/MCM-22 composite molecular sieves[J]. J Fuel Chem Technol,2014,42(10):1240−1245. doi: 10.3969/j.issn.0253-2409.2014.10.013 [22] 王改, 杨冬花, 薄琼, 李玉鹏, 胡丽, 李晓峰, 窦涛. 具有多级孔MCM-41/Y复合分子筛的合成及表征[J]. 分子催化,2018,32(4):325−333.WANG Gai, YANG Dong-hua, BO Qiong, LI Yu-peng, HU Li, LI Xiao-feng, DOU Tao. Synthesis and characterization of MCM-41/Y composite molecular sieves with Multi-pore structure[J]. J Mol Catal,2018,32(4):325−333. [23] 李君华, 谢锦印, 张丹, 刘琳, 邢锦娟. 碱改性ZSM-5分子筛及其甲醇芳构化性能[J]. 燃料化学学报, 2021, 49(3): 338–345.LI Jun-hua, XIE Jin-yin, ZHANG Dan, LIU Lin, XING Jin-juan. J Fuel Chem Technol, 2021, 49(3): 338–345. [24] VALTCHEV V P, SMAIHI M, FAUST A C, VIDAL L. Equisetum a rvense templating of zeolite beta macrostructures with hierarchical porosity[J]. Chem Mater,2018,16(7):1350−1355. [25] 邵秀丽. 晶种法合成整体式多级孔ZSM-5分子筛[J]. 工业催化,2019,27(6):49−53. doi: 10.3969/j.issn.1008-1143.2019.06.008SHAO Xiu-li. Synthesis of multiporous ZSM-5 zeolite by crystal seed method[J]. Ind Catal,2019,27(6):49−53. doi: 10.3969/j.issn.1008-1143.2019.06.008 [26] 王迎运, 周亚松, 魏强, 杨晓宇. Y/β复合分子筛制备方法及应用研究进展[J]. 化工进展,2016,35(S2):155−159.WANG Ying-yun, ZHOU Ya-song, WEI Qiang, YANG Xiao-yu. Progress in preparation and application of Y/β composite molecular sieves[J]. Chem Ind Eng Prog (China),2016,35(S2):155−159. [27] EMEIS C A. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts[J]. J Chem Inform,2010,24(2):347−354. [28] 刘晓飒, 陈淑俊, 赵铁英, 李德艳, 白小鸽, 留琴. 吡啶-热脱附-红外法确定催化剂酸性[J]. 工业催化,2015,23(10):817−820. doi: 10.3969/j.issn.1008-1143.2015.10.018LIU Xiao-sa, CHEN Shu-jun, ZHAO Tie-ying, LI De-yan, BAI Xiao-ge, LIU Qin. Determination of acidity of catalyst by pyridine thermal desorption and infrared spectroscopy[J]. Ind Catal,2015,23(10):817−820. doi: 10.3969/j.issn.1008-1143.2015.10.018 [29] 薛彦峰, 牛宇岚, 郑洪岩, 崔晓静, 马庆国, 唐建可, 丁莉峰. ZSM-5分子筛水蒸气选择性脱铝及其对乙醇转化制丙烯的影响[J]. 燃料化学学报,2021,49(8):1111−1121. doi: 10.1016/S1872-5813(21)60064-6XUE Yan-feng, NIU Yu-lan, ZHENG Hong-yan, CUI Xiao-jing, MA Qing-guo, TANG Jian-ke, DING Li-feng. Water vapor selective dealuminization of ZSM-5 zeolite and its effect on ethanol conversion to propylene[J]. J Fuel Chem Technol,2021,49(8):1111−1121. doi: 10.1016/S1872-5813(21)60064-6 [30] 靳立军, 刘斯宝, 胡浩权. 纳米晶堆积结构ZSM-5微球的合成及其在2-甲基萘甲基化中的应用[J]. 石油学报(石油加工),2015,31(3):705−710.JIN Li-jun, LIU Si-bao, HU Hao-quan. Synthesis of nanocrystalline stacked ZSM-5 microspheres and their application in methylation of 2-methylnaphthalene[J]. Acta Pet Sin (Pet Process Sec),2015,31(3):705−710. -





 下载:
下载: