Electronic modulation and synergistic effect on Fe-doped NiSe@NiS composites to improve oxygen evolution performance
-
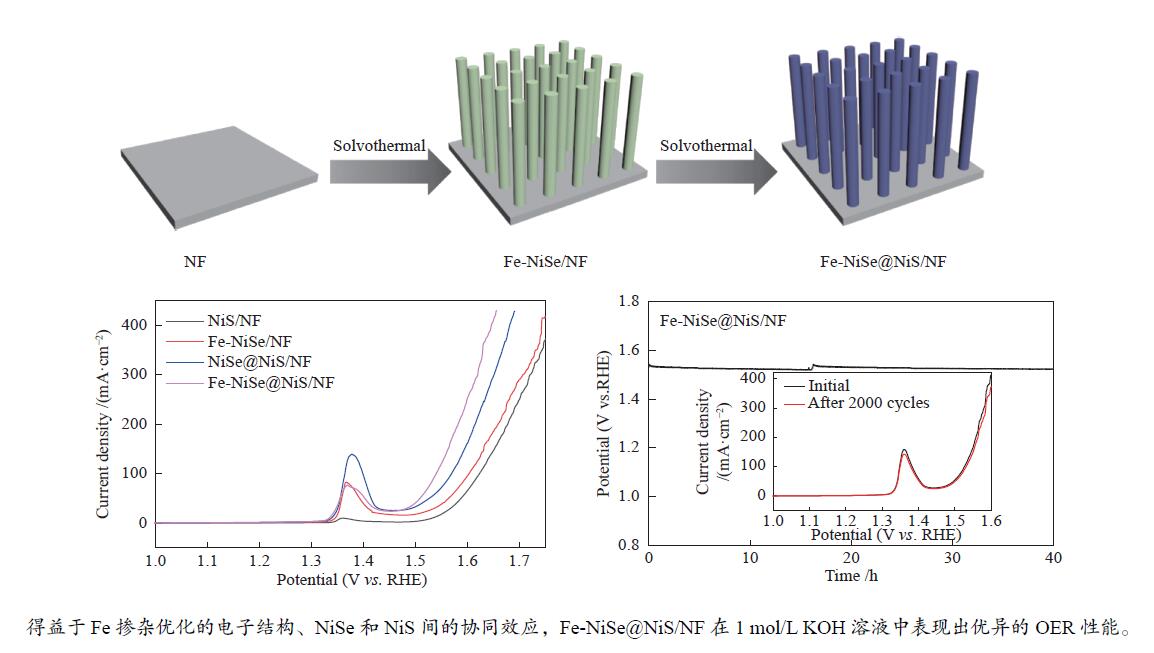
摘要: 本研究采用溶剂热法,在泡沫镍(NF)基底上原位合成出Fe掺杂的硒化镍和硫化镍复合材料(Fe-NiSe@NiS/NF)。得益于Fe掺杂优化的电子结构、NiSe和NiS间的协同效应以及高效的电荷转移速率,Fe-NiSe@NiS/NF在1 mol/L KOH溶液中表现出优异的OER性能。在过电位为330 mV时可实现电流密度150 mA/cm2,且电压在稳定40 h后没有发生显著改变。
-
关键词:
- NiSe@NiS复合材料 /
- Fe掺杂 /
- 协同效应 /
- 电子调制 /
- 析氧反应.
Abstract: In this paper, Fe-doped nickel selenide and nickel sulfide composites was in-situ grown on nickel foam (NF) to prepare Fe-NiSe@NiS/NF by solvothermal method. Benefit from the optimized electron structure by Fe doping, the synergistic effect of NiSe@NiS and faster electron transfer, Fe-NiSe@NiS/NF exhibited excellent OER activity with the overpotential of 330 mV at 150 mA/cm2 in 1 mol/L KOH solution. The voltage barely changed after 40 h of test.-
Key words:
- NiSe@NiS composites /
- Fe doping /
- synergistic effect /
- electronic modulation /
- oxygen evolution reaction
-
图 5 在特定电压范围(0.90224 –1.0224 V vs. RHE)内不同扫描速率下的CV曲线:(a)NiS/NF;(b)Fe-NiSe/NF;(c)NiSe@NiS/NF;(d)Fe-NiSe@NiS/NF;(e)双电层电容;(f)基于ECSA的极化曲线
Figure 5 CV curves at the potential of 0.9224 –1.0224 V vs RHE with different scan rates of (a) NiS/NF, (b) Fe-NiSe/NF, (c) NiSe@NiS/NF, (d) Fe-NiSe@NiS/NF, (e) Measured capacitive currents, (f) Normalized LSV curves based on ECSA for NiS/NF, Fe-NiSe/NF,NiSe@NiS/NF and Fe-NiSe@NiS/NF
图 6 不同铁含量的Fe-NiSe@NiS/NF的(a)极化曲线;(b)奈奎斯特曲线;((c)–(e))在特定电压范围(0.9224 –1.0224 V vs. RHE)内不同扫描速率下的CV曲线;(f)双电层电容
Figure 6 (a) LSV curves, (b) Nyquist plots, ((c)–(e)) CV curves at the potential of 0.9024 –1.0024 V vs. RHE with different scan rates (20–100 mV/s) on Fe-NiSe@NiS/NF with different Fe content, (f) Measured capacitive currents
表 1 ICP测试
Table 1 Results of ICP
Catalyst ICP-MS w/% Ni Fe Fe-NiSe@NiS/NF-1 47.28 1.93 Fe-NiSe@NiS/NF-3 41.35 4.61 Fe-NiSe@NiS/NF-5 39.47 8.03 -
[1] CHU S, MAJUMDAR A. Opportunities and challenges for a sustainable energy future[J]. Nature,2012,448:294−303. [2] JIAO Y, ZHENG Y, JARONIEC M, QIAO S Z. Design of electrocatalysts for oxygen- and hydrogen-involving energy conversion reactions[J]. Chem Soc Rev,2015,44(8):2060−2086. doi: 10.1039/C4CS00470A [3] WU Z P, LU X F, ZANG S Q, LOU X W. Non-noble-metal-based electrocatalysts toward the oxygen evolution reaction[J]. Adv Funct Mater,2020,30(15):1910274. doi: 10.1002/adfm.201910274 [4] LI X, GUAN B Y, GAO S, LOU X W. A general dual-templating approach to biomass-derived hierarchically porous heteroatom-doped carbon materials for enhanced electrocatalytic oxygen reduction[J]. Energy Environ Sci,2019,12(2):648−655. doi: 10.1039/C8EE02779J [5] KASIAN O, GROTE J P, GEIGER S, CHEREVKO S, MAYRHOFER K J J. The common intermediates of oxygen evolution and dissolution reactions during water electrolysis on iridium[J]. Angew Chem Int Ed,2018,57(9):2488−2491. doi: 10.1002/anie.201709652 [6] ZHANG H, LIU Y, CHEN T, ZHANG J, ZHANG J, LOU X W D. Unveiling the activity origin of electrocatalytic oxygen evolution over isolated Ni atoms supported on a N-Doped carbon matrix[J]. Adv Mater,2019,31(48):1904548. doi: 10.1002/adma.201904548 [7] SUEN N T, HUNG S F, QUAN Q, ZHANG N, XU Y J, CHEN H M. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives[J]. Chem Soc Rev,2017,46(2):337−365. doi: 10.1039/C6CS00328A [8] SHI X, KEY J, JI S, LINKOV V, LIU F, WANG H, GAI H, WANG R. Ni(OH)2 nanoflakes supported on 3D Ni3 Se2 nanowire array as highly efficient electrodes for asymmetric supercapacitor and Ni/MH battery[J]. Small,2019,15(29):1802861. doi: 10.1002/smll.201802861 [9] ZHU M, ZHANG D, LU Q, YAN Y, ZHU K, YE K, YAN J, CAO D, WANG Q, HUANG X, WANG G. Hollow hexagonal NiSe-Ni3Se2 anchored onto reduced graphene oxide as efficient electrocatalysts for hydrogen evolution in wide-pH range[J]. Int J Hydrogen Energy,2021,46(39):20524−20533. doi: 10.1016/j.ijhydene.2021.03.170 [10] NIE F, LI Z, DAI X, YIN X, GAN Y, YANG Z, WU B, REN Z, CAO Y, SONG W. Interfacial electronic modulation on heterostructured NiSe@CoFe LDH nanoarrays for enhancing oxygen evolution reaction and water splitting by facilitating the deprotonation of OH to O[J]. Chem Eng J,2022,431:134080. doi: 10.1016/j.cej.2021.134080 [11] CHEN Y, REN Z, FU H, ZHANG X, TIAN G, FU H. NiSe-Ni0.85Se heterostructure nanoflake arrays on carbon paper as efficient electrocatalysts for overall water splitting[J]. Small,2018,14(25):1800763. doi: 10.1002/smll.201800763 [12] GUO K, WANG Y, YANG S, HUANG J, ZOU Z, PAN H, SHINDE P S, PAN S, HUANG J, XU C. Bonding interface boosts the intrinsic activity and durability of NiSe@Fe2O3 heterogeneous electrocatalyst for water oxidation[J]. Sci Bull,2021,66(1):52−61. doi: 10.1016/j.scib.2020.06.003 [13] BAO W, YANG C, AI T, ZHANG J, ZHOU L, LI Y, WEI X, ZOU X, WANG Y. Modulating interfacial charge distribution of NiSe nanoarrays with NiFe-LDH nanosheets for boosting oxygen evolution reaction[J]. Fuel,2023,332:126227. doi: 10.1016/j.fuel.2022.126227 [14] TANG C, ZHANG R, LU W, HE L, JIANG X, ASIRI A M, SUN X. Fe-doped CoP nanoarray: A monolithic multifunctional catalyst for highly efficient hydrogen generation[J]. Adv Mater,2017,29(2):126227. [15] WU Z, ZOU Z, HUANG J, GAO F. Fe-doped NiO mesoporous nanosheets array for highly efficient overall water splitting[J]. J Catal,2018,358:243−252. doi: 10.1016/j.jcat.2017.12.020 [16] TIAN Z, LIU Y, XU Q, SHI Y, MA C, PENG B, LIU G, YANG J, ZHENG W. Fe doped NiSe2 nanoarrays to boost electrocatalytic oxygen evolution reaction[J]. Electrochimica Acta,2022,425:140711. doi: 10.1016/j.electacta.2022.140711 [17] YIN X, DAI X, NIE F, REN Z, YANG Z, GAN Y, WU B, CAO Y, ZHANG X. Electronic modulation and proton transfer by iron and borate co-doping for synergistically triggering the oxygen evolution reaction on a hollow NiO bipyramidal prism[J]. Nanoscale,2021,13(33):14156−14165. doi: 10.1039/D1NR03500B [18] LU C, YAN Y, ZHAI T, FAN Y, ZHOU W. 2-nm-thick NiCo LDH@NiSe single‐crystal nanorods grown on Ni foam as integrated electrode with enhanced areal capacity for supercapacitors[J]. Batteries Supercaps,2020,3:534−540. [19] JEGHAN S M N, KIM D, LEE Y, KIM M, LEE G. Designing a smart heterojunction coupling of cobalt-iron layered double hydroxide on nickel selenide nanosheets for highly efficient overall water splitting kinetics[J]. Appl Catal B: Environ,2022,308:121221. [20] ZOU Z, WANG X, HUANG J, WU Z, GAO F. An Fe-doped nickel selenide nanorod/nanosheet hierarchical array for efficient overall water splitting[J]. J Mater Chem A,2019,7(5):2233−2241. doi: 10.1039/C8TA11072G [21] CHANG K, TRAN D T, WANG J, KIM N H, LEE J H. A 3D hierarchical network derived from 2D Fe-doped NiSe nanosheets/carbon nanotubes with enhanced OER performance for overall water splitting[J]. J Mater Chem A,2022,10(6):3102−3111. doi: 10.1039/D1TA07393A [22] FANG L, LI W, GUAN Y, FENG Y, ZHANG H, WANG S, WANG Y. Tuning unique peapod-like Co(SxSe1−x)2 Nanoparticles for efficient overall water splitting[J]. Adv Funct Mater,2017,27(24):1701008. doi: 10.1002/adfm.201701008 [23] YANG Y, KANG Y, ZHAO H, DAI X, CUI M, LUAN X, ZHANG X, NIE F, REN Z, SONG W. An interfacial electron transfer on tetrahedral NiS2 /NiSe2 heterocages with dual-phase synergy for efficiently triggering the oxygen evolution reaction[J]. Small,2020,16(1):1905083. doi: 10.1002/smll.201905083 [24] MERKI D, FIERRO S, VRUBEL H, HU X. Amorphous molybdenum sulfide films as catalysts for electrochemical hydrogen production in water[J]. Chem Sci,2011,2(7):1262−1267. doi: 10.1039/C1SC00117E [25] LV J, CHENG Y, LIU W, QUAN B, LIANG X, JI G, DU Y. Achieving better impedance matching by a sulfurization method through converting Ni into NiS/Ni3S4 composites[J]. J Mater Chem C,2018,6(7):1822−1828. doi: 10.1039/C7TC05503J [26] LIANG H, GANDI A N, ANJUM D H, WANG X, SCHWINGENSCHLOGL U, ALSHAREEF H N. Plasma-assisted synthesis of NiCoP for efficient overall water splitting[J]. Nano Lett,2016,16(12):7718−7725. doi: 10.1021/acs.nanolett.6b03803 [27] CHEN S, KANG Z, HU X, ZHANG X, WANG H, XIE J, ZHENG X, YAN W, PAN B, XIE Y. Delocalized spin states in 2D atomic layers realizing enhanced electrocatalytic oxygen evolution[J]. Adv Mater,2017,29(30):1701687. doi: 10.1002/adma.201701687 [28] DAI X, LI Z, MA Y, LIU M, DU K, SU H, ZHUO H, YU L, SUN H, ZHANG X. Metallic cobalt encapsulated in bamboo-like and nitrogen-rich carbonitride nanotubes for hydrogen evolution reaction[J]. ACS Appl Mater Interfaces,2016,8(10):6439−6448. doi: 10.1021/acsami.5b11717 [29] WANG P, PU Z, LI W, ZHU J, ZHANG C, ZHAO Y, MU S. Coupling NiSe2-Ni2P heterostructure nanowrinkles for highly efficient overall water splitting[J]. J Catal,2019,377:600−608. doi: 10.1016/j.jcat.2019.08.005 -





 下载:
下载: