Enhanced photocatalysis using metal-organic framework MIL-101(Fe) for crude oil degradation in oil-polluted water
-
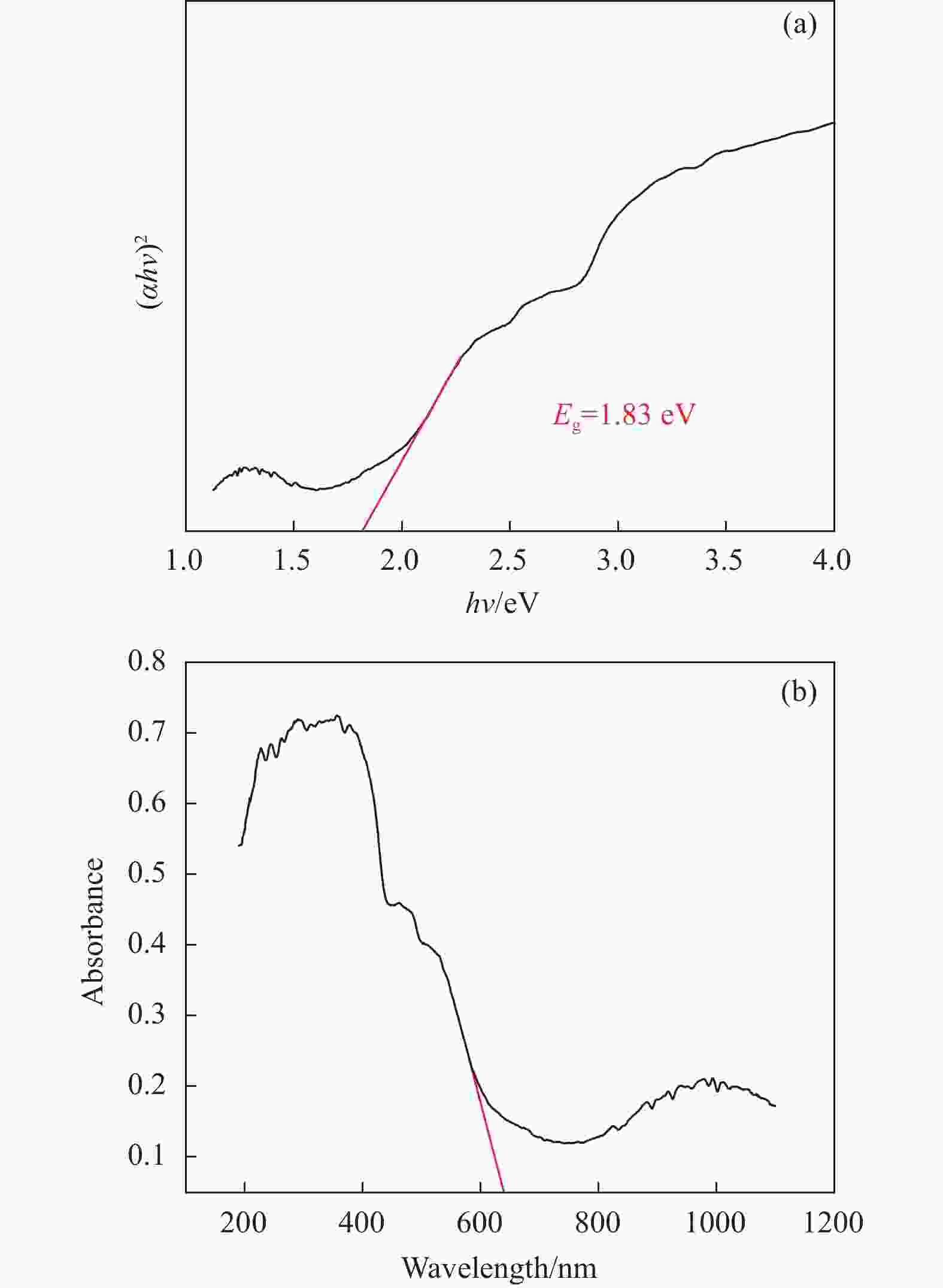
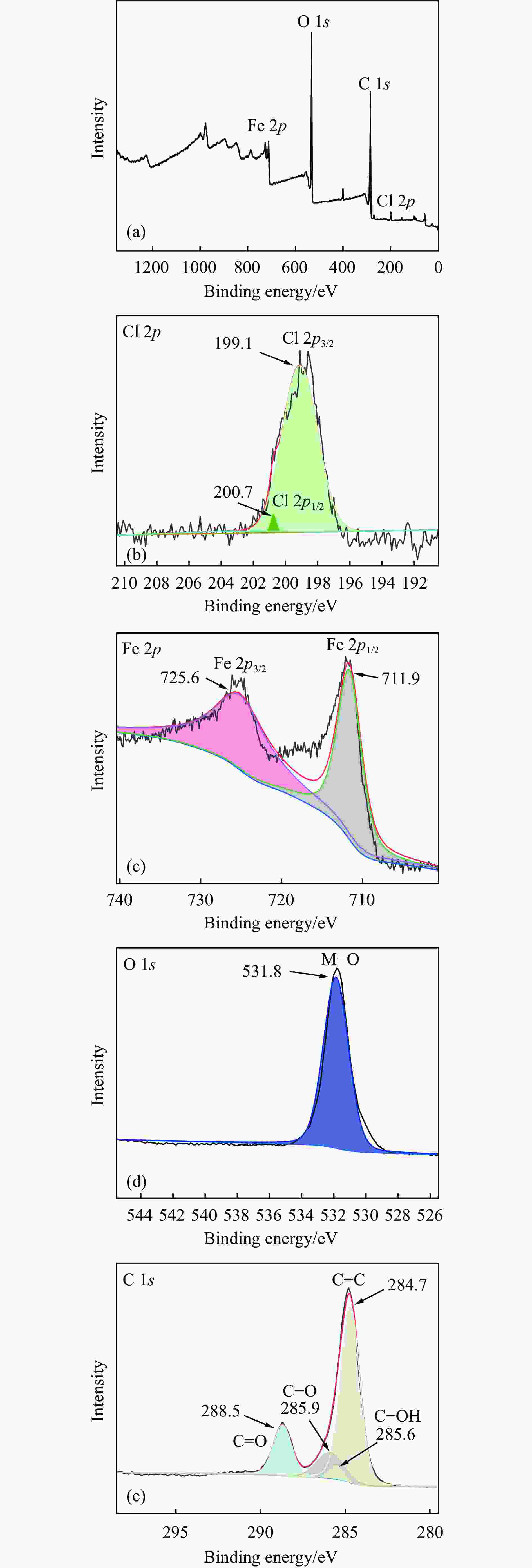
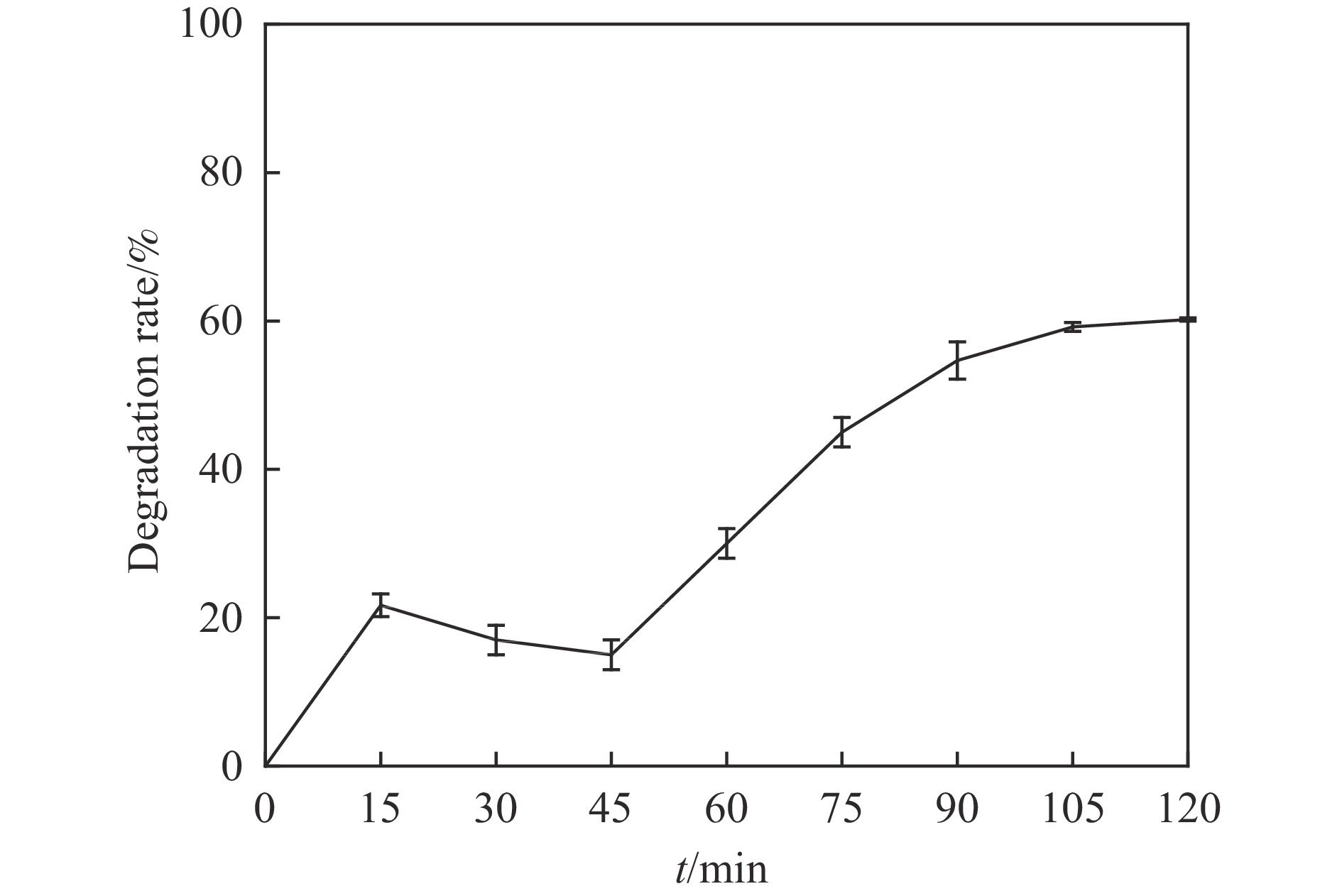
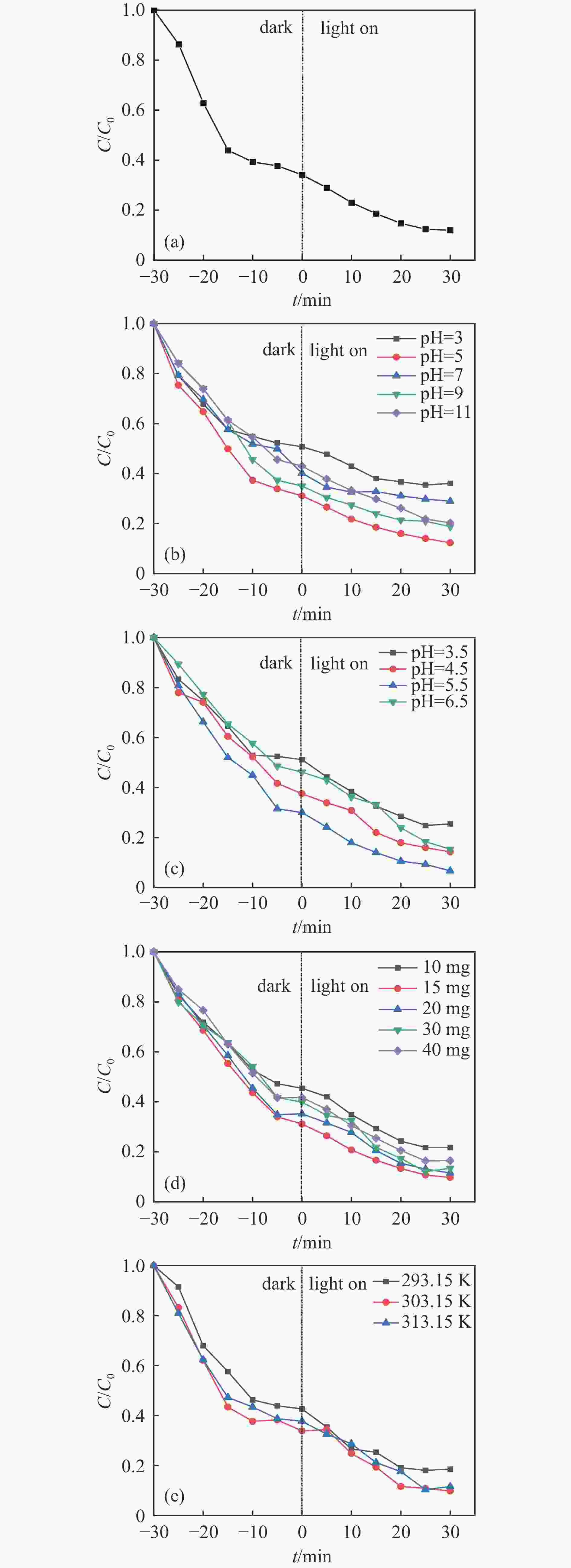
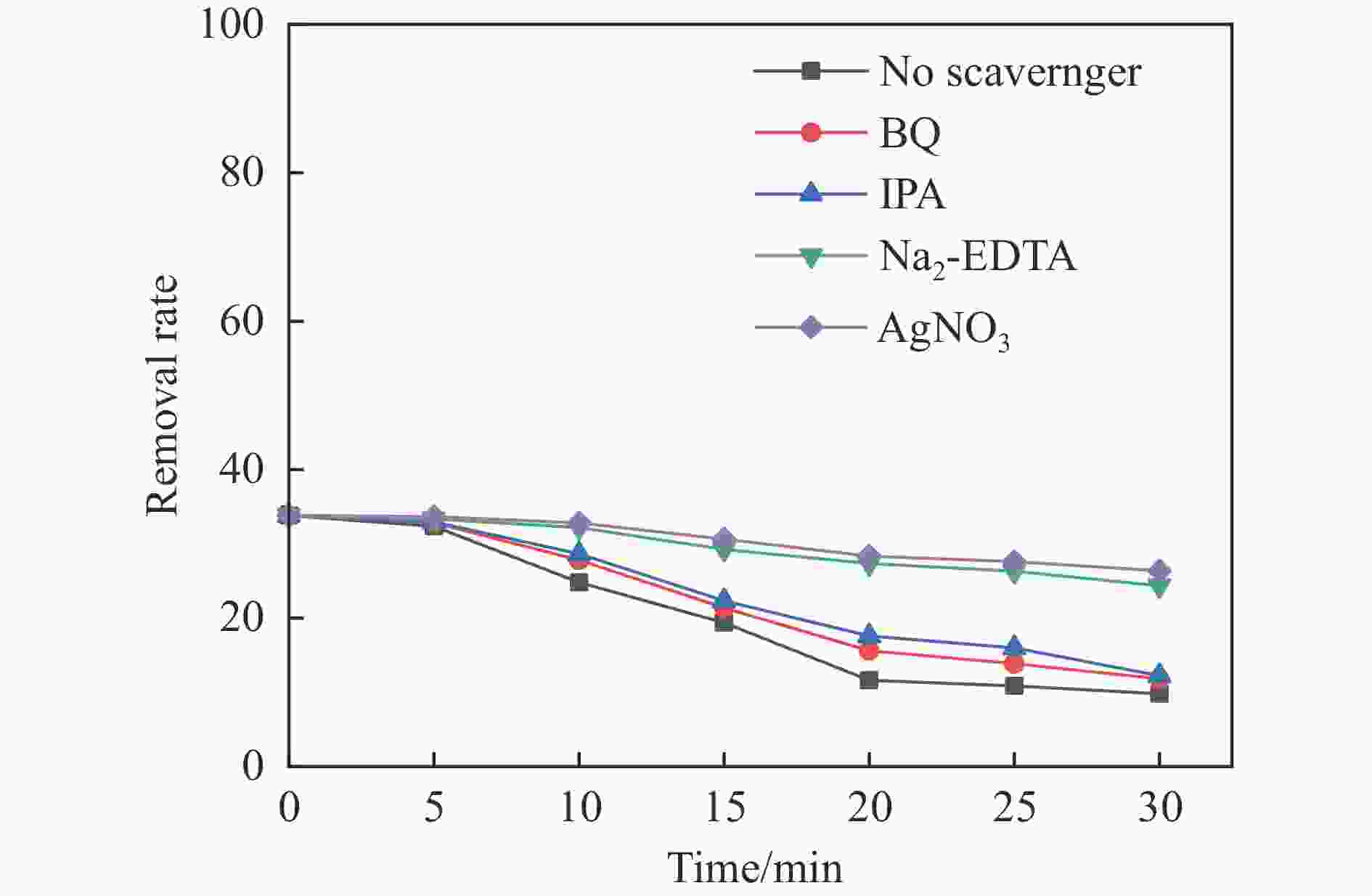
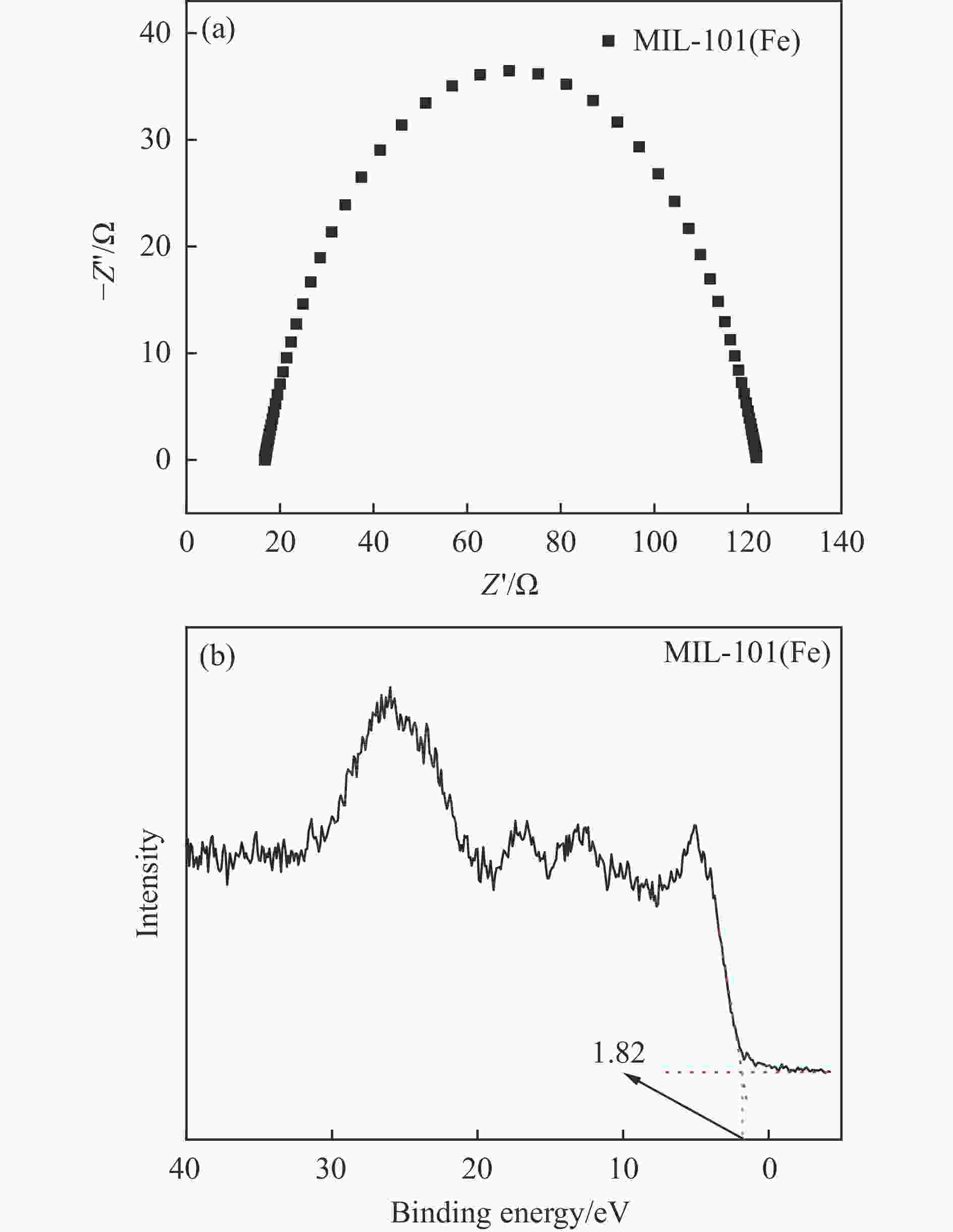
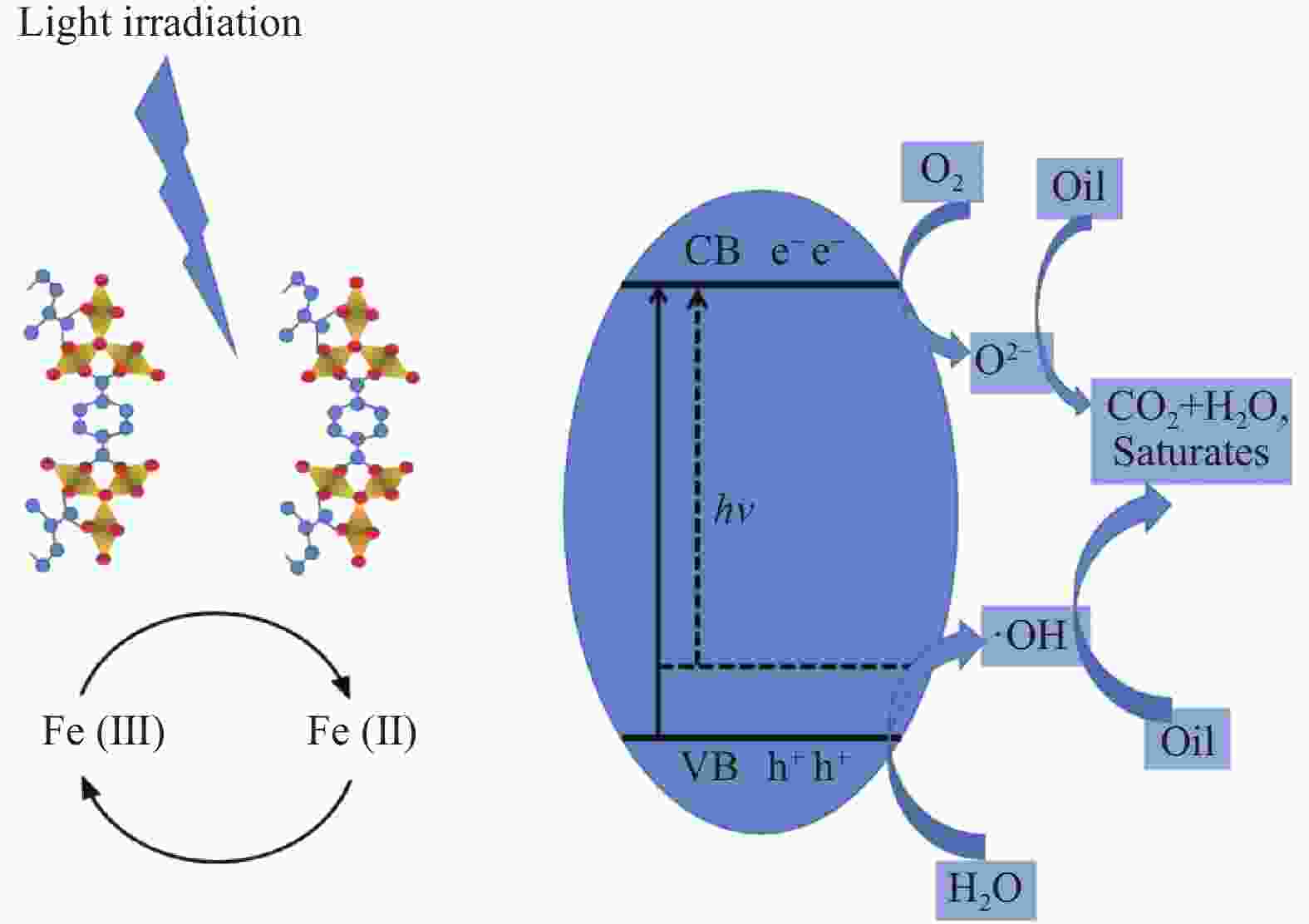
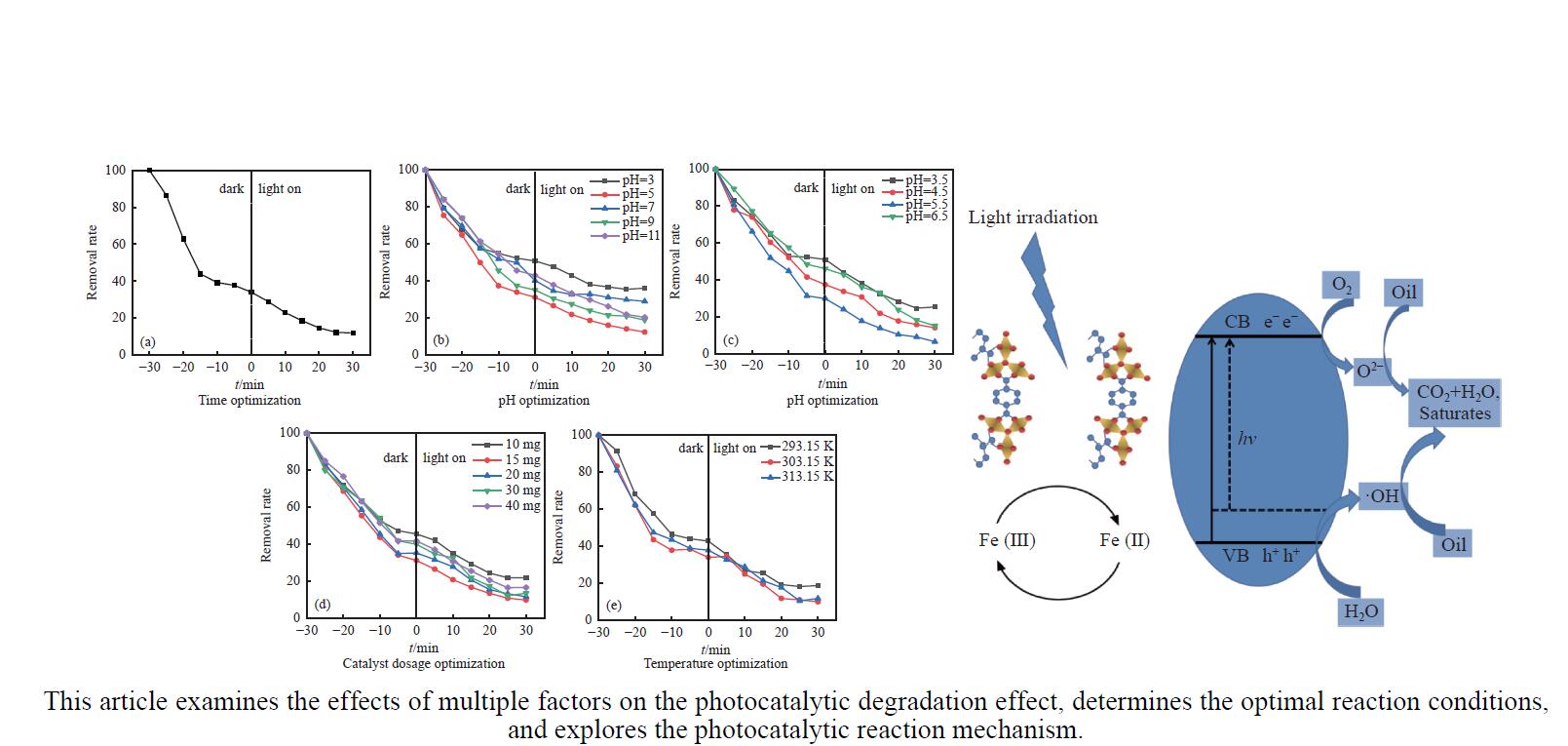
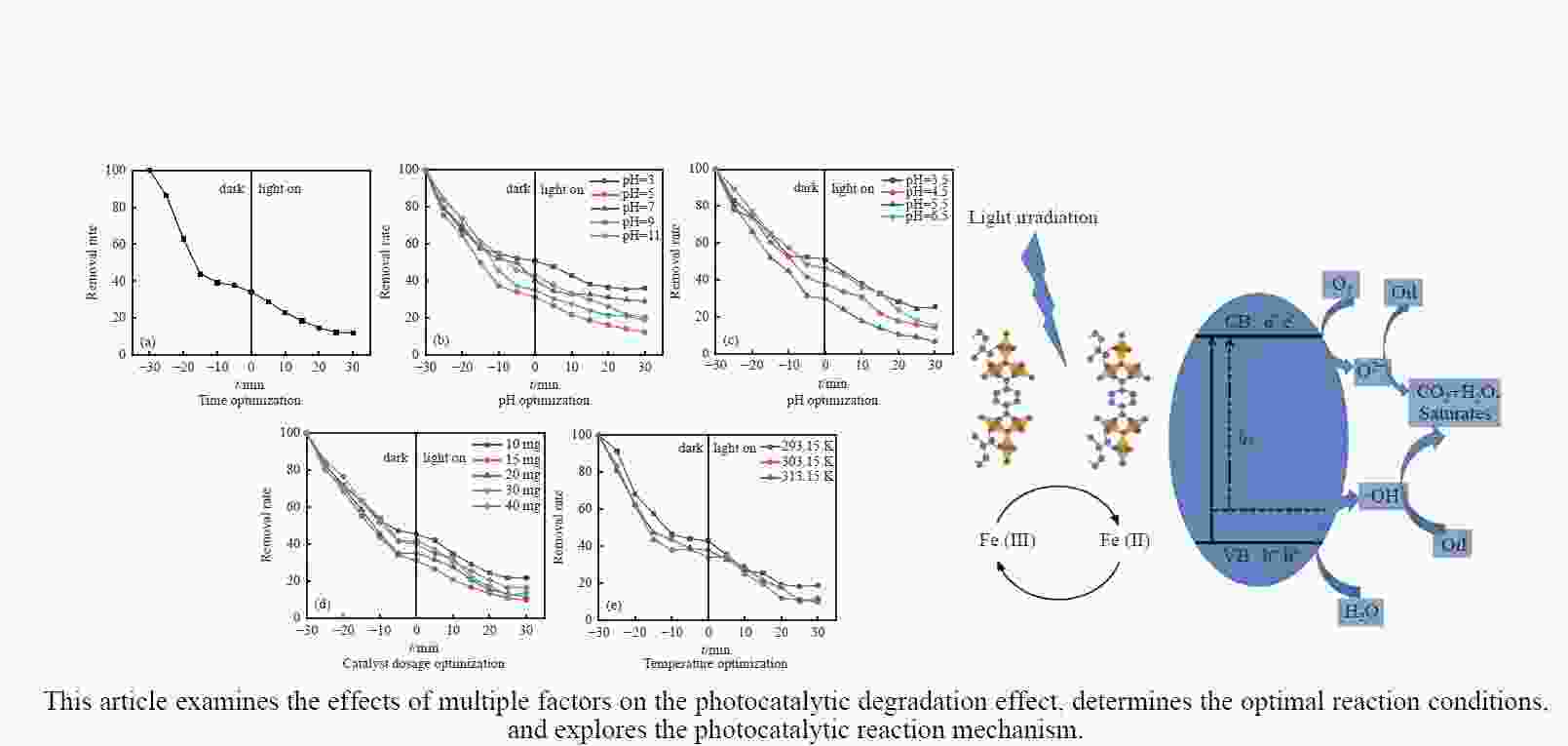
摘要: 利用溶剂热法合成了一种稳定的金属有机框架(MOF)MIL-101(Fe),并作为一种新型光催化剂提高了油田废水中原油的降解性能。通过对反应条件的优化,确定了以下最佳参数:暗反应时间为30 min,光反应时间为30 min,pH值为5.5,催化剂量为150 mg/L,反应温度为303.15 K。在这些反应条件下,去除率达到了94.73%。本研究是铁基MOFs在油田废水光催化降解中的应用。MIL-101(Fe)在温和的酸性条件下表现出良好的稳定性,并且可以有效地循环利用。这些发现为利用MIL-101(Fe)作为一种很有前途的工业应用材料,通过光催化降解从受油污染的水中去除原油提供了有价值的见解。Abstract: A stable metal-organic framework (MOF), MIL-101(Fe), was successfully synthesised using a solvothermal method and employed as a novel photocatalyst for degrading crude oil in oilfield wastewater. Through optimisation of reaction conditions, the following optimal parameters were determined: a dark reaction time of 30 min, a light reaction time of 30 min, a pH of 5.5, a catalyst amount of 150 mg/L, and a reaction temperature of 303.15 K. Under these reaction conditions, an impressive removal of 94.73% was achieved. This study represents the first application of Fe-based MOFs in the photocatalytic degradation of oilfield wastewater. MIL-101(Fe) notably demonstrated excellent stability under mild acid conditions and can be efficiently recycled. These findings offer valuable insights into using MIL-101(Fe) as a promising material for industrial applications in removing crude oil from oil-polluted water through photocatalytic degradation.
-
Key words:
- MIL-101(Fe) /
- MOF /
- photocatalysis /
- solvothermal /
- oilfield wastewater /
- degradation
-
Table 1 Summary of research on photocatalytic degradation of oily wastewater by various catalysts
Photocatalyst Activator
amount/gPollutant Concentration Dose of
pollutant/mLSource of
irradiationTime/
minMaximum
degradation/
adsorptionRef. MIL-101(Fe) 0.015 OPW 5.0×10−4 60 UV 30 94.73 this paper TiO2-SiO2 0.16 OPW 1.0×10−4 400 UV 30 95 [19] Ce/Bi2O3 10.51 OPW 5.0×10−5 2000 Visible 30 90 [20] Fe- TiO2 0.10 OPW 2.05×10−4 100 UV 60 98.1 [21] Go/ZnIn2S4 0.10 OPW 1.0×10−4
(COD)100 Visible 60 72 [22] MoS2/ZIS 0.10 OPW 1.2×10−4 100 Visible 80 92 [23] TiO2
(Aerogel)0.16 OPW 1.0×10−4 400 UV 90 91 [24] MoS2/P-C3N4 0.10 OPW 1.6×10−4
(COD)100 Visible 100 94 [25] γ-Fe2O3 1 toluene 5.0×10−2 100 Visible 120 90 [26] γ-Fe2O3 1 toluene 1.0×10−1 100 Visible 120 86 [26] γ-Fe2O3 1 toluene 1.5×10−1 100 Visible 120 78 [26] Ag@ZnO/
Zn2Ti3O80.10 OPW 1.5×10−4 100 UV 300 89 [27] AgTiZn
(MW)0.10 OPW 1.5×10−4 100 UV 300 90.15 [28] CuTiZn 0.10 OPW 1.5×10−4 100 UV 300 87.02 [28] AgMgZn
(MW)0.10 OPW 1.5×10−4 100 UV 300 93.35 [28] AgMnZn
(MW)0.10 OPW 1.5×10−4 100 UV 300 88.95 [28] Pt/TiO2 0.30 POME (COD) 4.0×10−2−1.0×10−1 300 UV& Visible 480 90 [29] Ag/TiO2 0.30 POME (COD) 4.0×10−2−1.0×10−1 300 UV& Visible 480 90 [30] PVDF/TiO2 15 cm2 membrane OIL 1.0×10−3 100 UV 480 60+ [31] g-C3N4-2AC 0.025 OPW 1.0×10−3 50 Visible 480 97.2 [32] GCN 0.20 OPW 1.0×10−3 200 UV 540 96.6 [33] GCN 0.20 OPW 1.0×10−3 200 Visible 540 85.4 [33] EG-ZnO 0.10 OPW 1.0×10−3 100 UV 4320 35 [34] N/TiO2/rGO 0.025 OPW 5.0×10−1 200 UV 40320 54.80 [35] -
[1] HASSANI S S, DARAEE M, SOBAT Z. Advanced development in upstream of petroleum industry using nanotechnology[J]. Chin J Chem Eng,2020,28(6):1483−1491. doi: 10.1016/j.cjche.2020.02.030 [2] SHEN Y, LI D W, WANG L L, et al. Superelastic polyimide nanofiber-based aerogels modified with silicone nano filaments for ultrafast oil/water separation[J]. ACS Appl Mater Interfaces,2021,13(17):20489−20500. doi: 10.1021/acsami.1c01136 [3] LIU B C, LI H S, LIU N, et al. Experimental study on filtration effect of oilfield sewage based on new polyurethane modified materials[J]. Water Sci Technol,2020,82(10):2039−2050. doi: 10.2166/wst.2020.462 [4] ZHU X L, RAN Y L, GUO W J, et al. Optimization of reinjection treatment technology for oilfield wastewater in Longdong area[J]. E3S Web of Conferences,2020,194:04046. doi: 10.1051/e3sconf/202019404046 [5] HOFFMANN M R, MARTIN S T, CHOI W. Environmental applications of semiconductor photocatalysis[J]. Chem Rev,1995,95(1):69−96. doi: 10.1021/cr00033a004 [6] SINGH M, PAKSHIRAJAN K, TRIVEDI V. A study on combined effect of methylene blue and sodium anthraquinone-2-sulphonate on inactivation efficiency of escherichia coli and enterococcus hirae[J]. Appl Catal B: Environ,2019,88(3):283−291. [7] SIEDL N, BAUMANN S O, ELSER M J, et al. Particle networks from powder mixtures: generation of Tio2-Sno2 heterojunctions via surface charge-induced heteroaggregation[J]. J Phys Chem C,2019,116(43):22967−22973. [8] MA S L, ZHAN S H, XIA Y G, et al. Enhanced photocatalytic bactericidal performance and mechanism with novel Ag/ZnO/g-C3N4 composite under visible light[J]. Catal Today,2019,330:179−188. doi: 10.1016/j.cattod.2018.04.014 [9] ZHU T T, Ye X J, ZHANG Q Q, et al. Efficient utilisation of photogenerated electrons and holes for photocatalytic redox reactions using visible light-driven Au/ZnIn2S4 hybrid[J]. J Hazard Mater,2019,367:277−285. doi: 10.1016/j.jhazmat.2018.12.093 [10] XIA Q, WANG H, HUANG B B, et al. State‐of‐the‐art advances and challenges of iron-based metal organic frameworks from attractive features, synthesis to multifunctional applications[J]. Small,2019,15(2):1803088. doi: 10.1002/smll.201803088 [11] RUI K, ZHAO G, CHEN Y, et al. Hybrid 2D dual-metal-organic frameworks for enhanced water oxidation catalysis[J]. Adv Funct Mater,2018,28(26):1801554. doi: 10.1002/adfm.201801554 [12] ZHAO F, LIU Y, HAMMOUDA S B, et al. MIL-101(Fe)/g-C3N4 for enhanced visible-light-driven photocatalysis toward simultaneous reduction of Cr(VI) and oxidation of bisphenol A in aqueous media[J]. Appl Catal B: Environ,2020,272:119033. doi: 10.1016/j.apcatb.2020.119033 [13] HAN H, ZHANG H X, CHEN Y, et al. Enhanced photocatalysis degradation of organophosphorus flame retardant using MIL-101(Fe)/persulfate: effect of irradiation wavelength and real water matrixes[J]. Chem Eng J,2019,368:273−284. doi: 10.1016/j.cej.2019.02.190 [14] ZHANG Y, XIONG M Y, SUN A R, et al. MIL-101(Fe) nanodot-induced improvement of adsorption and photocatalytic activity of carbon fiber/TiO2-based weavable photocatalyst for removing pharmaceutical pollutants[J]. J Clean Prod,2021,290:125782. doi: 10.1016/j.jclepro.2021.125782 [15] MILLANGE F, GUILLOU N, MEDINA M E. Selective sorption of organic molecules by the flexible porous hybrid metal-organic framework MIL-53(Fe) controlled by various host-guest interactions[J]. J Mater Chem A , 2010, 22, 14: 4237–4245. [16] KIM P J, YOU Y W, PARK H, et al. Separation of SF6 from SF6/N2 mixture using metal–organic framework MIL-100(Fe) granule[J]. Chem Eng J,2015,262:683−690. doi: 10.1016/j.cej.2014.09.123 [17] KIM B C, HONG W G, LEE S M, et al. Enhancement of hydrogen storage capacity in polyaniline-vanadium pentoxide nanocomposites[J]. Int J Hydrogen Energ,2010,35:1300−1304. [18] AI L H, ZHANG C H, LI L L, et al. Iron terephthalate metal–organic framework: Revealing the effective activation of hydrogen peroxide for the degradation of organic dye under visible light irradiation[J]. Appl Catal B: Environ, 2014, 148–149: 191−200. [19] LI X W, ZHAO H L, LU P P, et al. Photocatalytic activity of monolithic TiO2-SiO2 composite aerogels obtained by ambient drying for degrading oily wastewater[J]. J Univ Sci Technol Beijing,2013,35(5):651−658. [20] ZHOU G H. Degradation of oily wastewater by coated photocatalyst with visible light response[D]. Dalian: Dalian Maritime University, 2014. [21] GAO Y H, CHEN Y, WANG Z X, et al. Treatment of oily wastewater by photocatalytic degradation using nano Fe-TiO2[J]. Adv Mater Res,2013,781−784(2606):2241−2244. [22] ZHANG Y, ZHAO L. Ozone-assisted photocatalytic degradation of oil-polluted water on graphene/ZnIn2S4 composites[J]. Chem Res Appl,2022,34(8):1719−1726. [23] XIA D G, DU Y L. Preparation of MoS2/ZnIn2S4 and photocatalytic degradation of oily wastewater[J]. Ind Water Wastewater,2022,53(5):51−56. [24] LI X W, LÜ P P, YAO K F, et al. Preparation of monolithic TiO2 aerogel via ambient drying and photocatalytic degradation of oily wastewater[J]. J Inorg Mater,2012,27(11):1153−1158. [25] RUAN Y. Study on the performance of P-C3N4 coated with MoS2 for the degradation of oily wastewater[J]. Energy Chem Ind,2022,43(5):64−68. [26] ROUSHERNAS P, YUSOP Z, MAJIDNIA Z. Photocatalytic degradation of spilled oil in sea water using maghemite nanoparticles[J]. Desalin Water Treat,2016,57(13):5837−5841. doi: 10.1080/19443994.2015.1005149 [27] CHU Y H, WU X, LU D Q, et al. Preparation of nano-Ag@ZnO/Zn2Ti3O8 and its application in photocatalytic degradation of oilfield wastewater[J]. Contemp Chem Ind,2022,51(2):350−353. [28] SUN B. Study on microwave synthesis, characterisation and properties of nanocomposites[D]. Fushun: Liaoning Petrochemical University, 2020. [29] CHENG C K, DERAHMAN M R, KHAN M R. Evaluation of the photocatalytic degradation of pre-treated palm oil mill effluent (POME) over Pt-loaded titania[J]. J Environ Chem Eng,2015,1(3):261−270. [30] CHENG C K, DERAHMAN M R, NG K H, et al. Preparation of titania doped argentum photocatalyst and its photoactivity towards palm oil mill effluent degradation[J]. J Clean Prod,2016,112(1):1128−1135. [31] RUSLI U N, ALIAS N H, SHAHRUDDIN M Z, et al. Photocatalytic Degradation of Oil using Polyvinylidene Fluoride/Titanium Dioxide Composite Membrane for Oily Wastewater Treatment. Munich: MATEC Web of Conferences, 2016, 69: 05003. [32] RAN L T. Study on modification of g-C3N4 photocatalyst and its degradation of oily wastewater[D]. Xi'an: Xi'an Shiyou University, 2022. [33] ALIAS N H, JAAFAR J, SAMITSU S, et al. Photocatalytic degradation of oilfield produced water using graphitic carbon nitride embedded in electrospun polyacrylonitrile nanofibers[J]. Chemosphere,2018,204(AUG.):79−86. [34] WANG W Y, WANG L Q, ZHANG R J. Preparation of expanded graphite-ZnO composite and its photocatalytic degradation of crude oil[J]. Environ Prot Chem Ind,2007,27(4):367−370. [35] LI Y H, ZHANG Q Q, JIANG J X, et al. Long-acting photocatalytic degradation of crude oil in seawater via combination of TiO2 and N-doped TiO2/ reduced graphene oxide[J]. Environ Technol,2021,42(5/8):860−870. -




 下载:
下载: