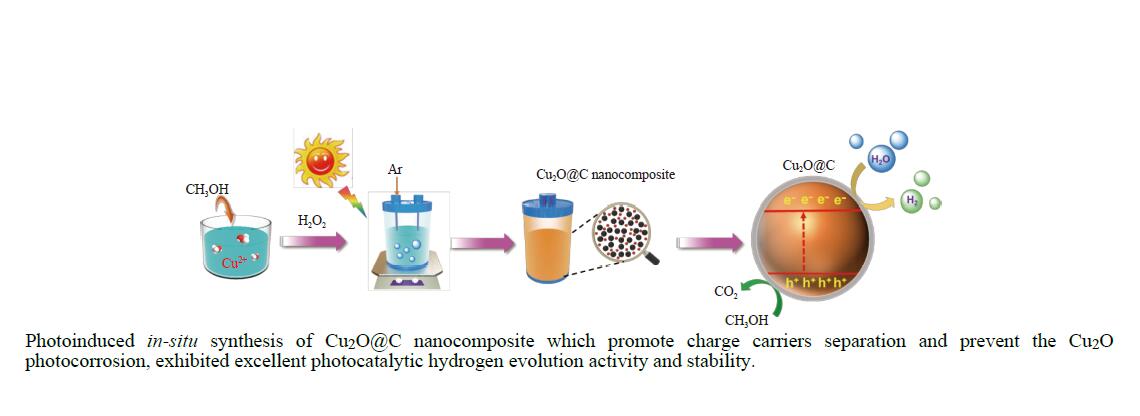
Photo-induced in-situ synthesis of Cu2O@C nanocomposite for efficient photocatalytic evolution of hydrogen
-
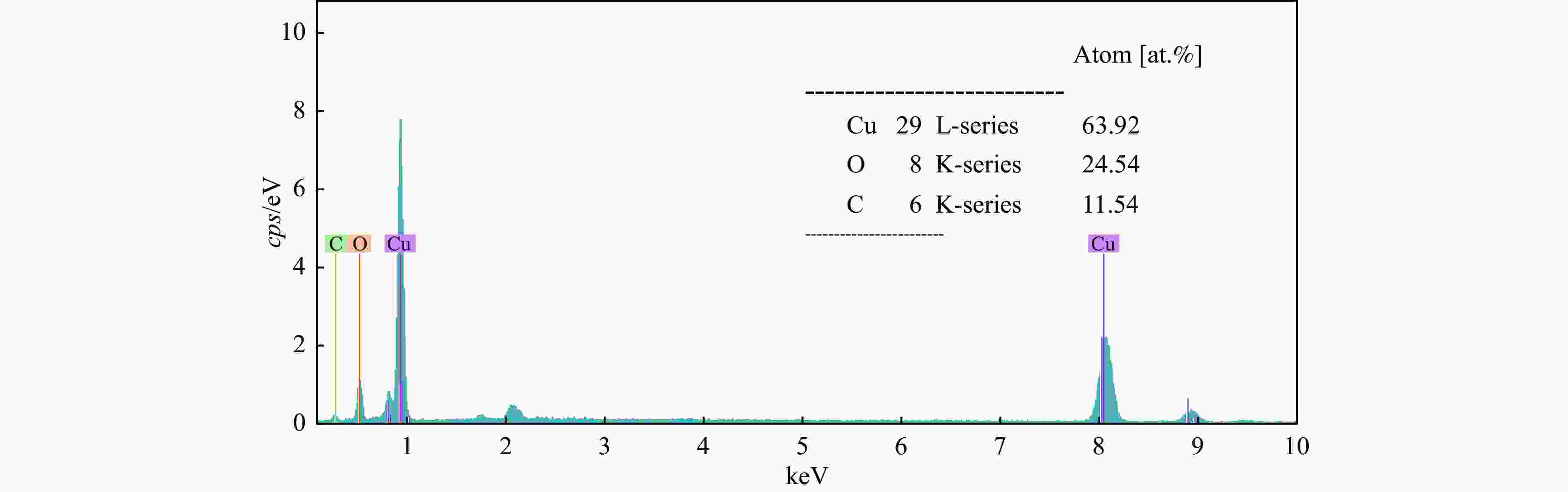
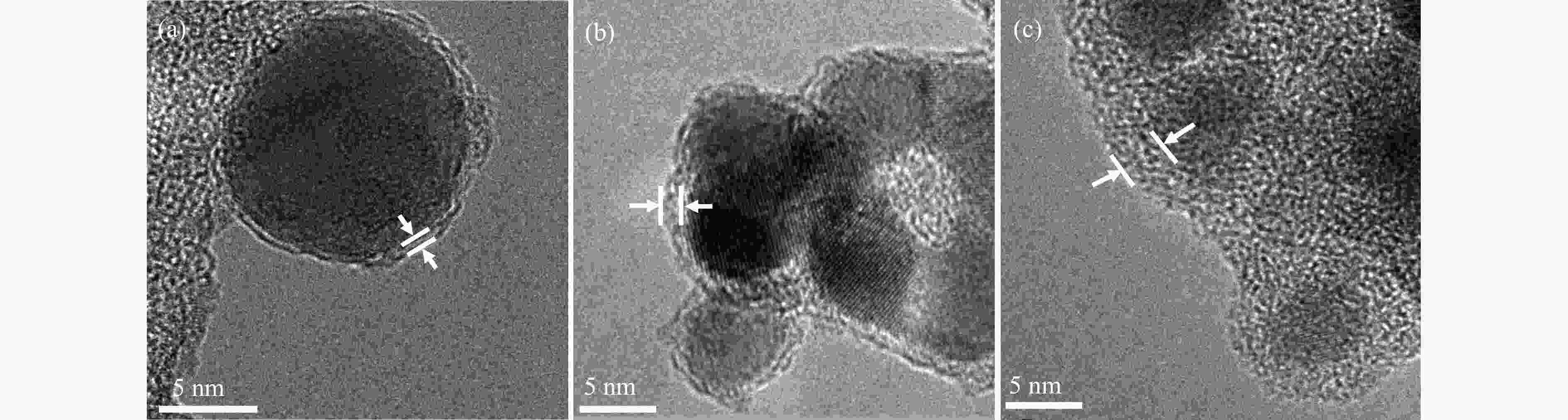
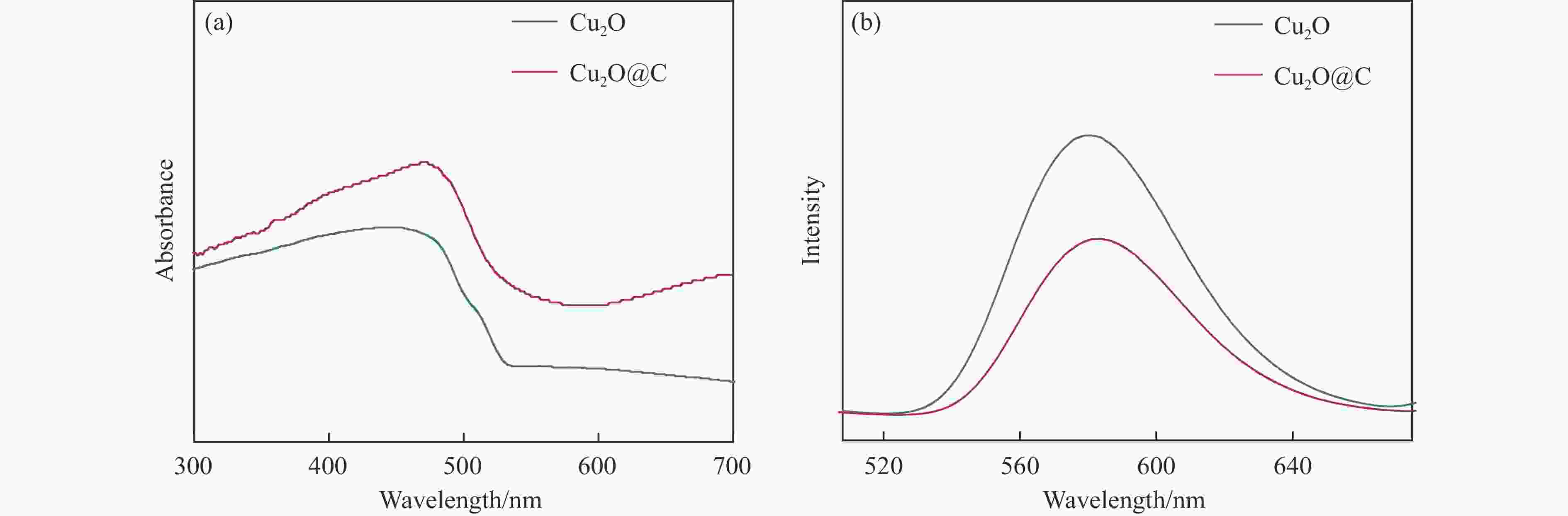
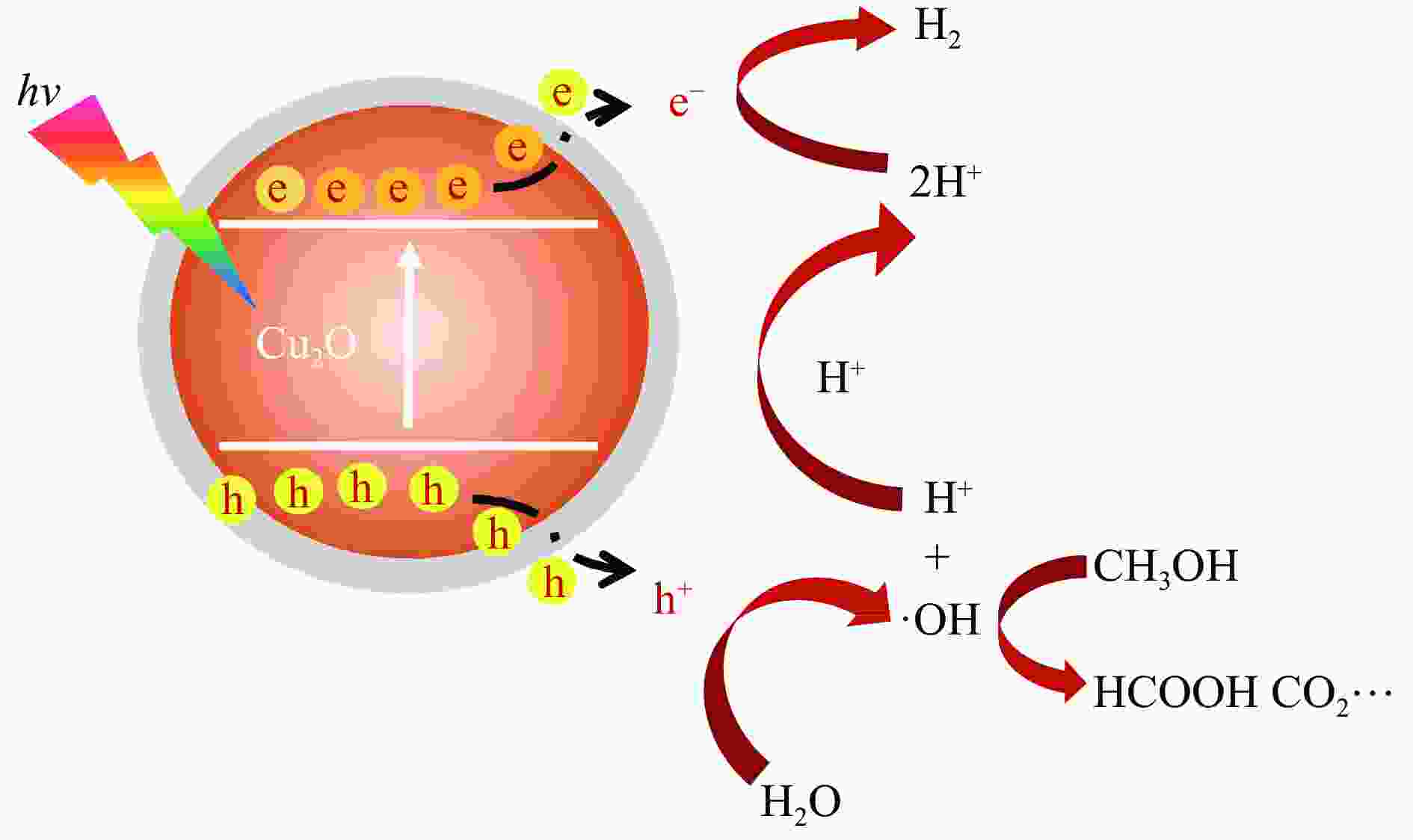
摘要: Cu2O具有禁带窄、环境友好和储量丰富等优点,是一种理想的可见光催化剂,然而其光生载流子复合率高和稳定性差等问题限制了Cu2O在光催化领域的实际应用。为此,本文采用光诱导原位技术,以甲醇为碳源、硫酸铜为铜源,一步成功制备了超薄炭壳层包覆的Cu2O复合纳米材料(Cu2O@C)。结果显示,与常规炭包覆方法相比,光诱导原位技术避免了苛刻的反应条件及繁琐的合成步骤对Cu2O半导体结构的破坏,有效保留了Cu2O本征电子结构,使其具有优异的光催化活性及稳定性。同时,Cu2O@C的核壳结构不仅可以钝化半导体表面缺陷和促进光生载流子的分离,而且炭壳层的包覆还可以避免Cu2O纳米颗粒与溶液的直接接触,有效抑制高活性反应中间体对催化剂结构的破坏。与单独的Cu2O纳米颗粒相比,Cu2O@C复合纳米材料在可见光下的光解水产氢活性和稳定性得到显著提高,产氢速率可达1.28 mmol/(g·h),且在连续五次循环稳定性测试中,氢气生成速率无明显变化。Abstract: Cuprous oxide (Cu2O) is an ideal visible light catalyst owing to its narrow band gap, environmental benignity and abundant storage; however, the fast recombination of photogenerated charge carriers and poor stability of Cu2O has impeded its application in photocatalysis. Herein, we demonstrate that Cu2O@C nanocomposite can spontaneously evolve from a methanol aqueous solution containing cupric ions under the induction of irradiation. Compared with the traditional carbon coating method, the Cu2O@C nanocomposite obtained by the photo-induced in-situ synthesis can reserve superior original characteristics of the semiconductor under mild reaction conditions, promote the charge transfer and enhance the separation efficiency of charge carriers; in addition, the carbon shells can also effectively prevent Cu2O from photo-corrosion. As a result, the Cu2O@C nanocomposite exhibits excellent photocatalytic activity in the hydrogen evolution in comparison with the Cu2O particles; the H2 evolution rate over the Cu2O@C nanocomposite reaches 1.28 mmol/(g·h) under visible light, compared with the value of 0.065 mmol/(g·h) over Cu2O. Moreover, the Cu2O@C nanocomposite displays good cycle stability, viz., without any deactivation in the catalytic activity after five cycles.
-
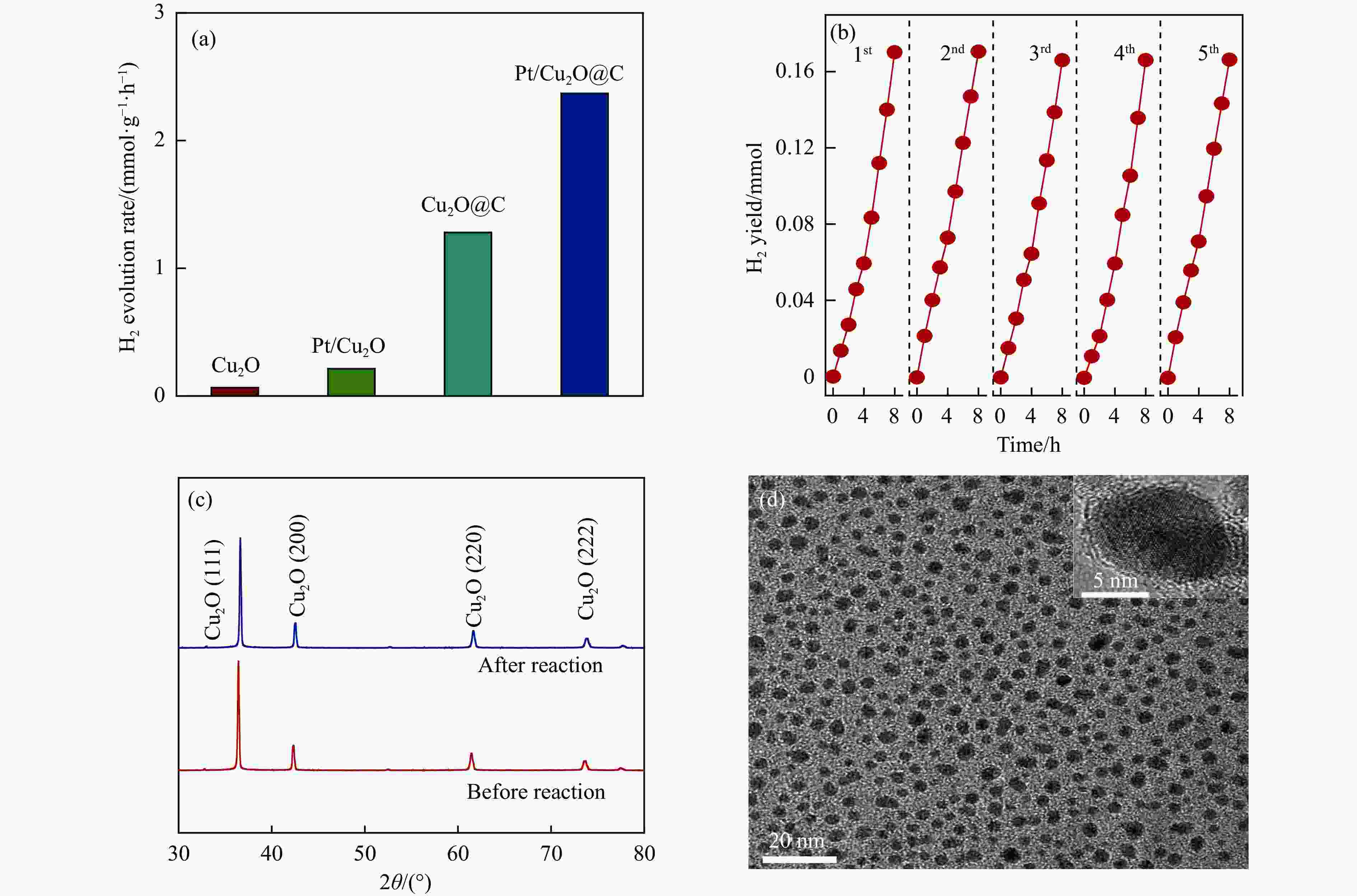
图 7 (a) 不同半导体光催化剂可见光下的光解水产氢性能;(b) Cu2O@C光解水产氢循环稳定性测试;(c) Cu2O@C稳定性测试前后的XRD谱图和 (d) 稳定性测试后的TEM图像
Figure 7 (a) Photocatalytic H2 evolution rate of as-prepared photocatalysts under visible light illumination; (b) Cycling stability test of photocatalytic hydrogen evolution over Cu2O@C nanocomposite; (c) XRD images of the Cu2O@C nanocomposite before and after cycling stability test; (d) TEM images after cycling stability test
-
[1] ZHAO Q, WANG K, WANG J, et al. Cu2O nanoparticle hyper-cross-linked polymer composites for the visible-Light photocatalytic degradation of methyl orange[J]. ACS Appl Nano Materials,2019,2(5):2706−2712. doi: 10.1021/acsanm.9b00210 [2] TAN X, YU C, ZHAO C, et al. Restructuring of Cu2O to Cu2O@Cu-metal-organic frameworks for selective electrochemical reduction of CO2[J]. ACS Appl Mater Interfaces,2019,11(10):9904−9910. [3] SIOL S, HELLMANN J C, TILLEY S D, et al. Band alignment engineering at Cu2O/ZnO heterointerfaces[J]. ACS Appl Mater Interfaces,2016,8(33):21824−21831. [4] HUANG C, YE W, LIU Q, et al. Dispersed Cu2O octahedrons on h-BN nanosheets for p-nitrophenol reduction[J]. ACS Appl Mater Interfaces,2014,6(16):14469−14476. [5] LI Q, LI X, WAGEH S, et al. CdS/Graphene nanocomposite photocatalysts[J]. Adv Energy Mater,2015,5(14):1500010−1500038. doi: 10.1002/aenm.201500010 [6] MA F, WU Y, SHAO Y, et al. 0D/2D nanocomposite visible light photocatalyst for highly stable and efficient hydrogen generation via recrystallization of CdS on MoS2 nanosheets[J]. Nano Energy,2016,27:466−474. doi: 10.1016/j.nanoen.2016.07.014 [7] LIU R, WANG P, WANG X, et al. UV and visible-light photocatalytic activity of simultaneously deposited and doped Ag/Ag(I)-TiO2 photocatalyst[J]. J Phys Chem C,2012,116(33):17721−17728. doi: 10.1021/jp305774n [8] RUDD A L, BRESLIN C B. Photo-induced dissolution of zinc in alkaline solutions[J]. Electrochimica Acta,2000,45(10):1571−1579. doi: 10.1016/S0013-4686(99)00322-9 [9] HAN C, YANG M Q, WENG B, et al. Improving the photocatalytic activity and anti-photocorrosion of semiconductor ZnO by coupling with versatile carbon[J]. Phys Chem Chem Phy,2014,16(32):16891−16903. doi: 10.1039/C4CP02189D [10] WANG Y, BAI X, QIN H, et al. Facile one-step synthesis of hybrid graphitic carbon nitride and carbon composites as high-performance catalysts for CO2 photocatalytic conversion[J]. ACS Appl Mater Interfaces,2016,8(27):17212−17219. [11] ZHANG P, LIU H, LI X. Plasmonic CuCo/carbon dots: An unconventional photocatalyst used for photocatalytic overall water splitting[J]. ACS Sustainable Chem Eng,2020,8(49):17979−17987. [12] HU Y, GAO X, YU L, et al. Carbon-coated CdS petalous nanostructures with enhanced photostability and photocatalytic activity[J]. Angew Chem Inter Ed,2013,52(21):5636−5639. doi: 10.1002/anie.201301709 [13] LI Y, LI J. Carbon-coated macroporous Sn2P2O7 as anode materials for Li-ion battery[J]. J Phys Chem C,2008,112(36):14216−14219. doi: 10.1021/jp804438v [14] GUO Y, WANG H, HE C, et al. Uniform carbon-coated ZnO nanorods: Microwave-assisted preparation, cytotoxicity, and photocatalytic activity[J]. Langmuir,2009,25(8):4678−4684. doi: 10.1021/la803530h [15] YANG R, ZHAO W, ZHENG J, et al. One-step synthesis of carbon-coated tin dioxide nanoparticles for high lithium storage[J]. J Phys Chem C,2010,114(47):20272−20276. doi: 10.1021/jp107396a [16] JIANG L, QU Y, REN Z, et al. In situ carbon-coated yolk-shell V2O3 microspheres for lithium-ion batteries[J]. ACS Appl Mater Interfaces,2015,7(3):1595−1601. [17] DOZZI M V, CANDEO A, MARRA G, et al. Effects of photodeposited gold vs platinum nanoparticles on N, F-doped TiO2 photoactivity: A time-resolved photoluminescence investigation[J]. J Phys Chem C,2018,122(26):14326−14335. doi: 10.1021/acs.jpcc.8b02997 [18] MAEDA K, TERAMURA K, LU D, et al. Roles of Rh/Cr2O3 (core/shell) nanoparticles photodeposited on visible-light-responsive (Ga1-xZnx)(N1-xOx) solid solutions in photocatalytic overall water splitting[J]. J Phys Chem C,2007,111(20):7554−7560. doi: 10.1021/jp071056j [19] HUA Q, SHANG D, ZHANG W, et al. Morphological evolution of Cu2O nanocrystals in an acid solution: Stability of different crystal planes[J]. Langmuir,2011,27(2):665−671. doi: 10.1021/la104475s [20] PAL J, SASMAL A K, GANGULY M, et al. Surface plasmon effect of Cu and presence of n-p heterojunction in oxide nanocomposites for visible light photocatalysis[J]. J Phys Chem C,2015,119(7):3780−3790. doi: 10.1021/jp5114812 [21] XUE Y, GUO Y, YI Y, et al. Self-catalyzed growth of Cu@graphdiyne core-shell nanowires array for high efficient hydrogen evolution cathode[J]. Nano Energy,2016,30:858−866. doi: 10.1016/j.nanoen.2016.09.005 [22] BUSSER G W, MEI B, POUGIN A, et al. Photodeposition of copper and chromia on gallium oxide: The role of co-catalysts in photocatalytic water splitting[J]. ChemSusChem,2014,7(4):1030−1034. doi: 10.1002/cssc.201301065 [23] PORRAS J, GIANNAKIS S, TORRES-PALMA R A, et al. Fe and Cu in humic acid extracts modify bacterial inactivation pathways during solar disinfection and photo-Fenton processes in water[J]. Appl Catal B: Environ,2018,235:75−83. doi: 10.1016/j.apcatb.2018.04.062 [24] JIANG X, ZHANG M, SHI W, et al. Microstructure and optical properties of nanocrystalline Cu2O thin films prepared by electrodeposition[J]. Nanoscale Res Lett,2014,9:219−224. doi: 10.1186/1556-276X-9-219 [25] JI X F, XU H P, LIANG S D, et al. 3D ordered macroporous Pt/ZnS@ZnO core-shell heterostructure for highly effective photocatalytic hydrogen evolution[J]. Int J Hydrogen Energy,2022,47(40):17640−17649. doi: 10.1016/j.ijhydene.2022.03.241 [26] WU J X, XI X X, ZHU W, et al. Boosting photocatalytic hydrogen evolution via regulating Pt chemical states[J]. Chem Eng J,2022,442:136334−136339. doi: 10.1016/j.cej.2022.136334 [27] SCHWARZE M, STELLMACH D, SCHRODER M, et al. Quantification of photocatalytic hydrogen evolution[J]. Phys Chem Chem Phys,2013,15(10):3466−3472. doi: 10.1039/c3cp50168j [28] FLORES-ROJAS E, SAMANIEGO-BENITEZ J E, SERRATO R, et al. Transformation of nanostructures Cu2O to Cu3Se2 through different routes and the effect on photocatalytic properties[J]. ACS Omega,2020,5(32):20335−20342. doi: 10.1021/acsomega.0c02299 [29] MAHMOUD M A, QIAN W, EL-SAYED M A. Following charge separation on the nanoscale in Cu2O-Au nanoframe hollow nanoparticles[J]. Nano Lett,2011,11(8):3285−3289. doi: 10.1021/nl201642r [30] WANG Y, ZHANG YN, ZHAO G, et al. Design of a novel Cu2O/TiO2/carbon aerogel electrode and its efficient electrosorption-assisted visible light photocatalytic degradation of 2, 4, 6-trichlorophenol[J]. ACS Appl Mater Interfaces,2012,4(8):3965−3972. [31] ZHENG Y, ZHANG L, GUAN J, et al. Controlled synthesis of Cu0/Cu2O for efficient photothermal catalytic conversion of CO2 and H2O[J]. ACS Sustainable Chem Eng,2021,9(4):1754−1761. [32] HAMID S, DILLERT R, BAHNEMANN D W. Photocatalytic reforming of aqueous acetic acid into molecular hydrogen and hydrocarbons over Co-catalyst-Loaded TiO2: Shifting the product distribution[J]. J Phys Chem C,2018,122(24):12792−12809. doi: 10.1021/acs.jpcc.8b02691 [33] LIU B, NING L, ZHANG C, et al. Enhanced visible-light photocatalytic H2 evolution in Cu2O/Cu2se multilayer heterostructure nanowires having {111} facets and physical mechanism[J]. Inorg Chem,2018,57(13):8019−8027. doi: 10.1021/acs.inorgchem.8b01197 [34] WEI T, ZHU Y N, AN X, et al. Defect modulation of Z-Scheme TiO2/Cu2O photocatalysts for durable water splitting[J]. ACS Catal,2019,9(9):8346−8354. doi: 10.1021/acscatal.9b01786 [35] YIN W J, BAI L J, ZHU Y Z et al. Embedding metal in the interface of a p-n heterojunction with a stack design for superior Z-scheme photocatalytic hydrogen evolution[J]. ACS Appl Mater Interfaces,2016,8(35):23133−23142. -




 下载:
下载: