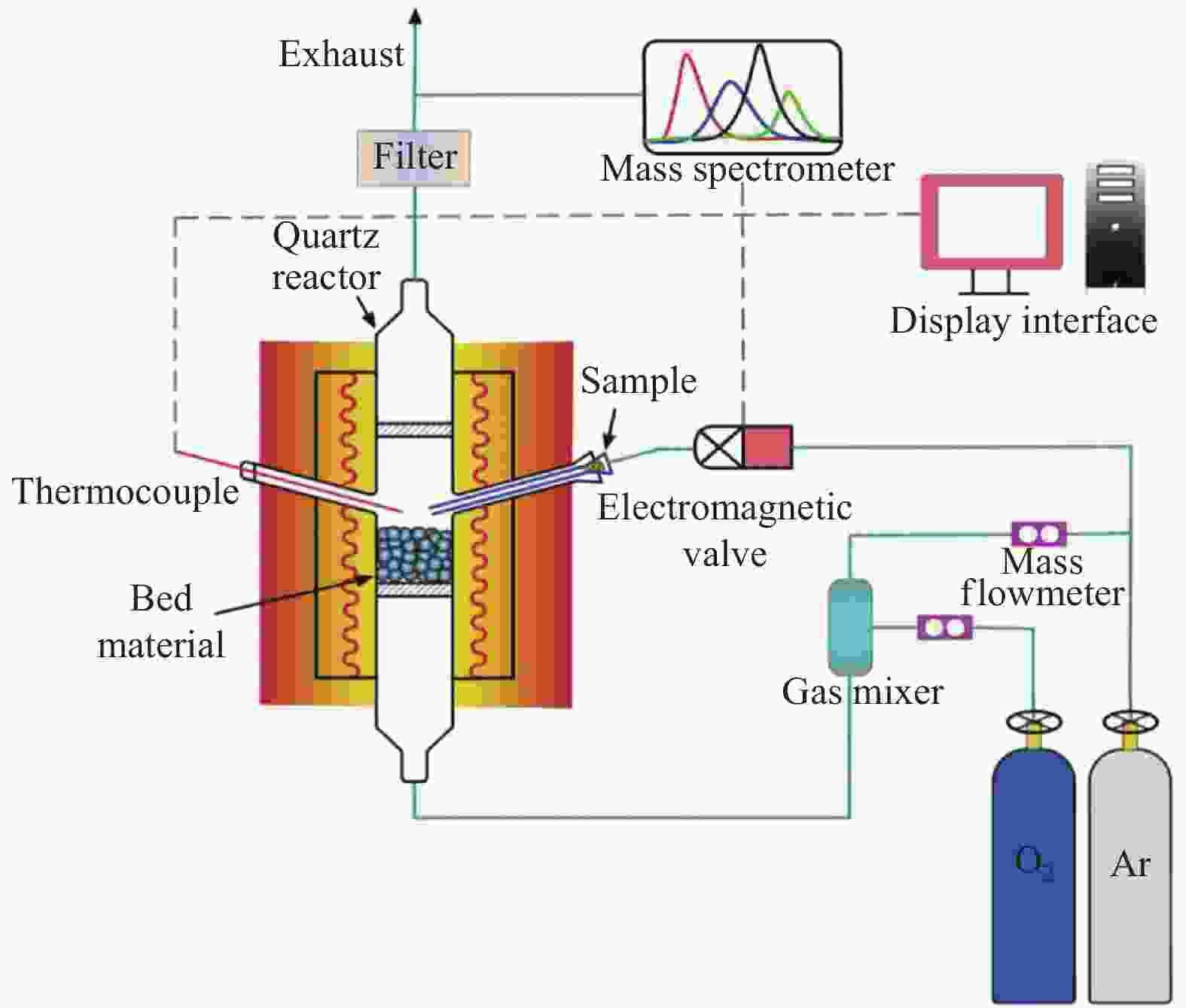
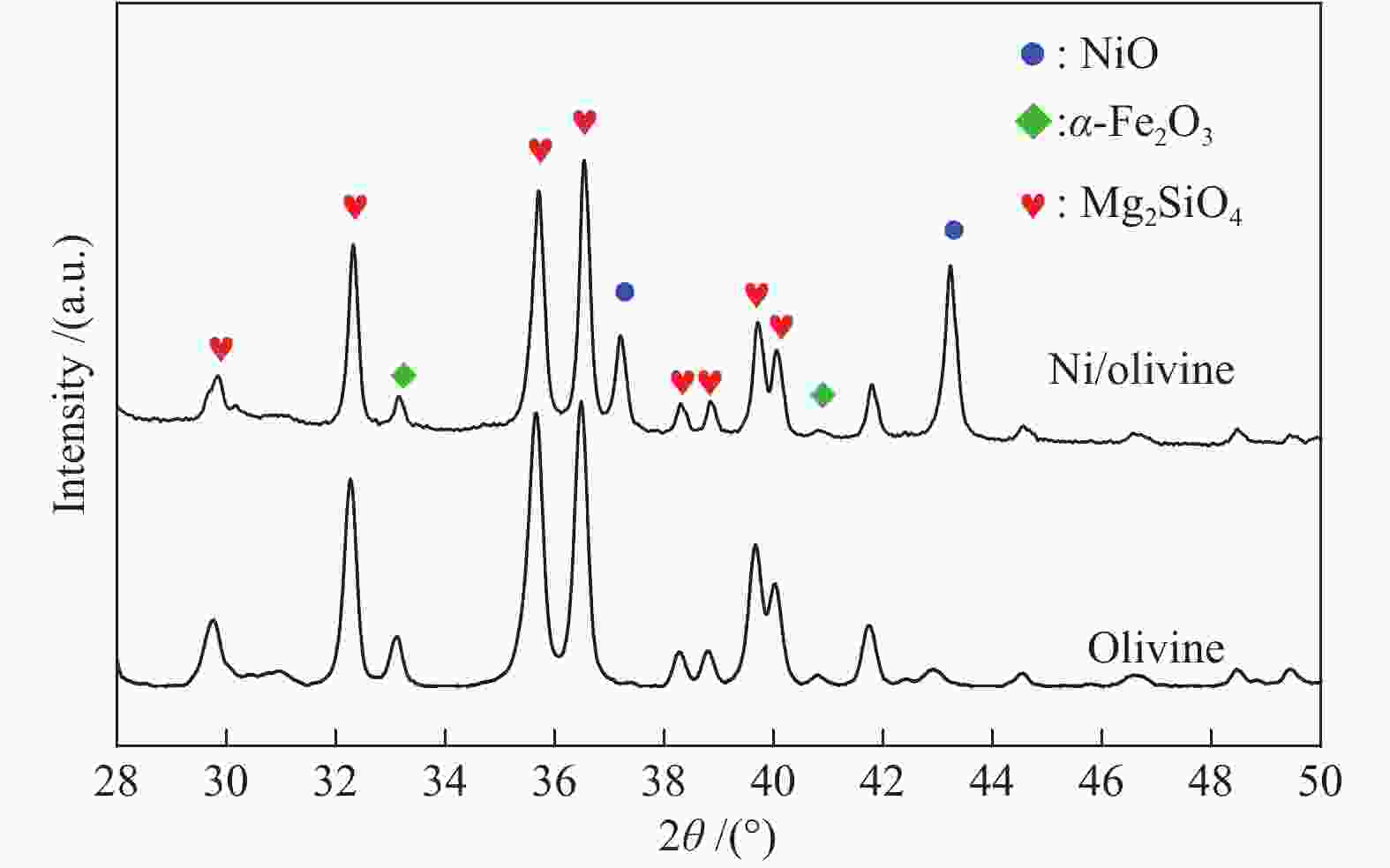
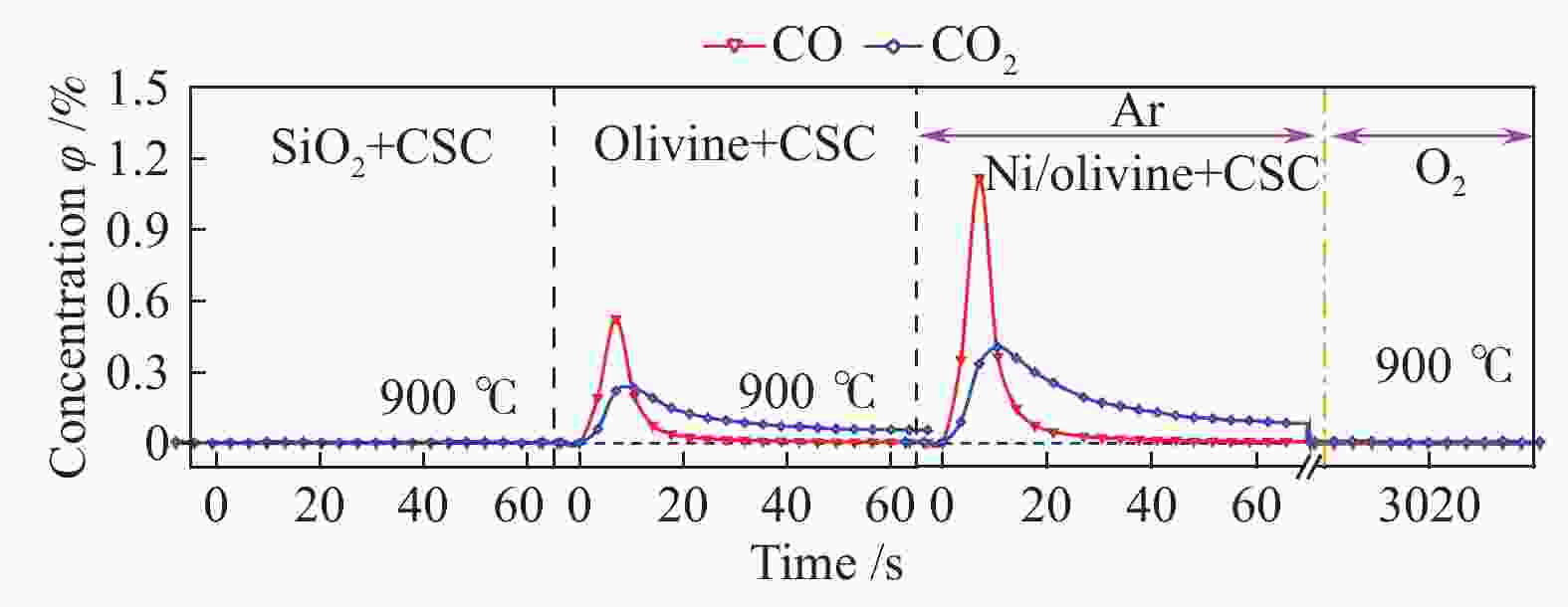
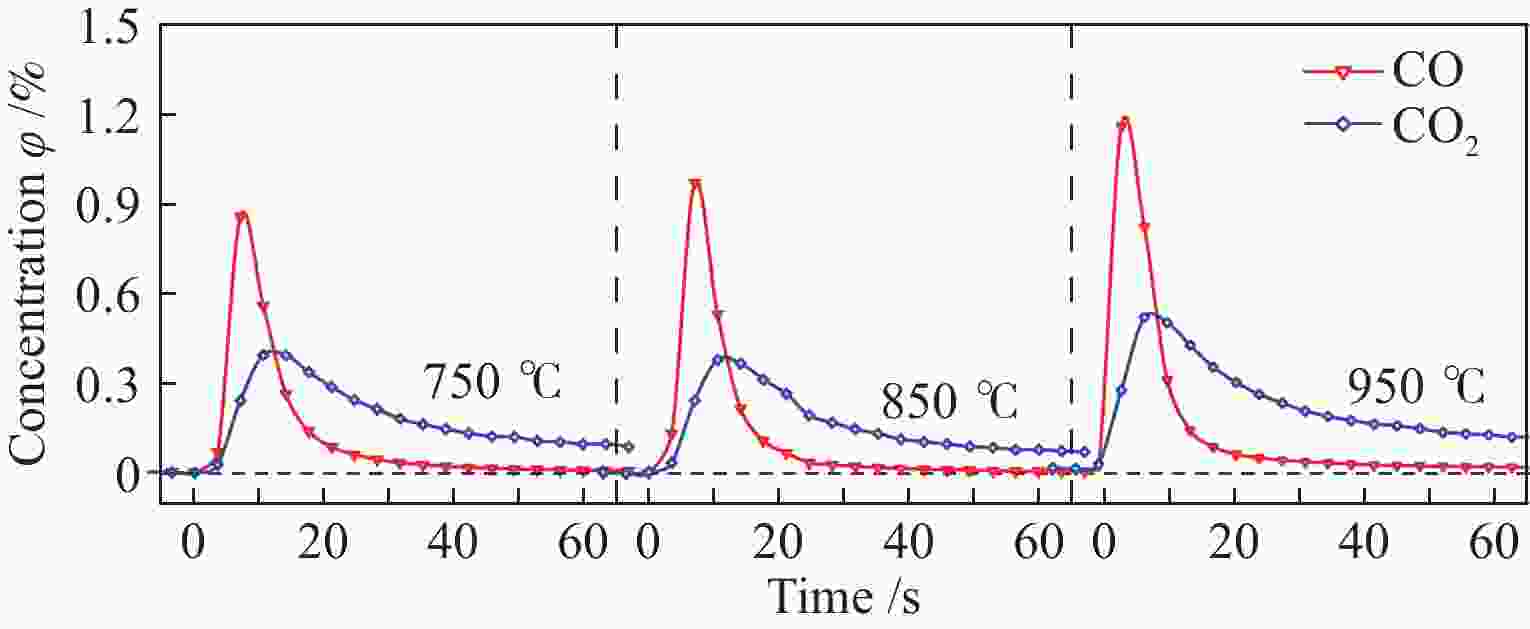
Kinetics of solid-solid reaction between cotton char and Ni/olivine in chemical looping gasification
-
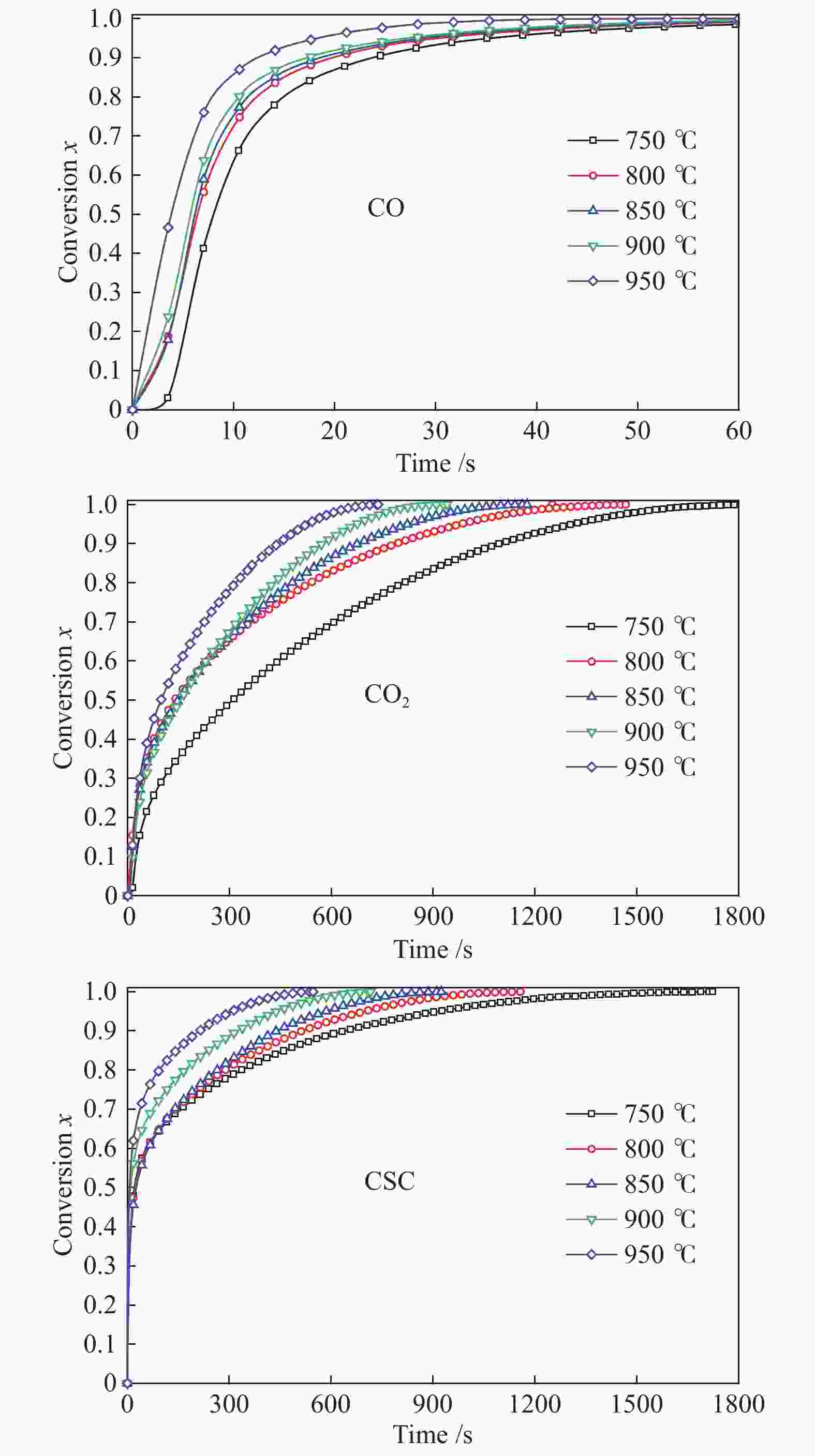
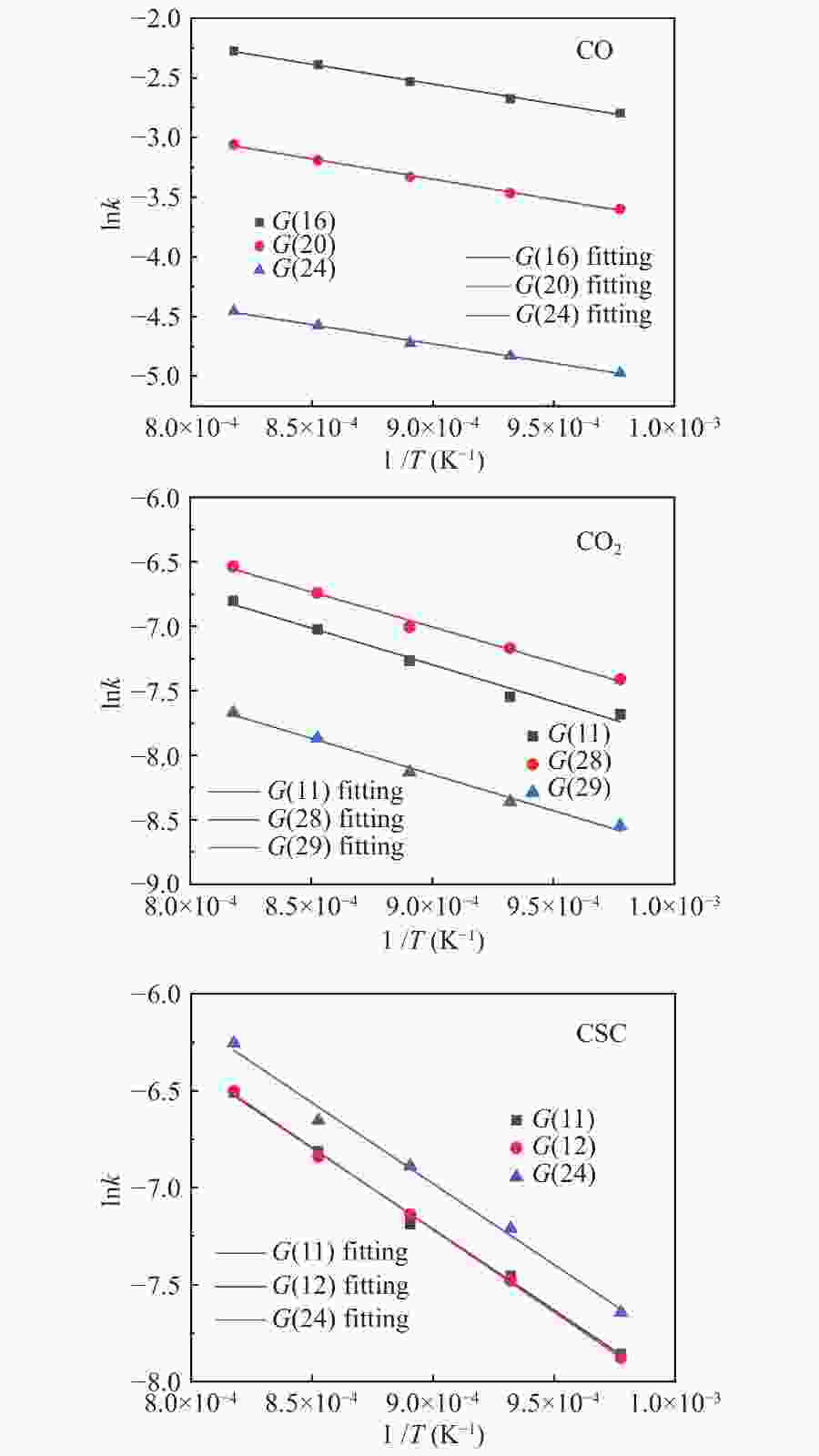
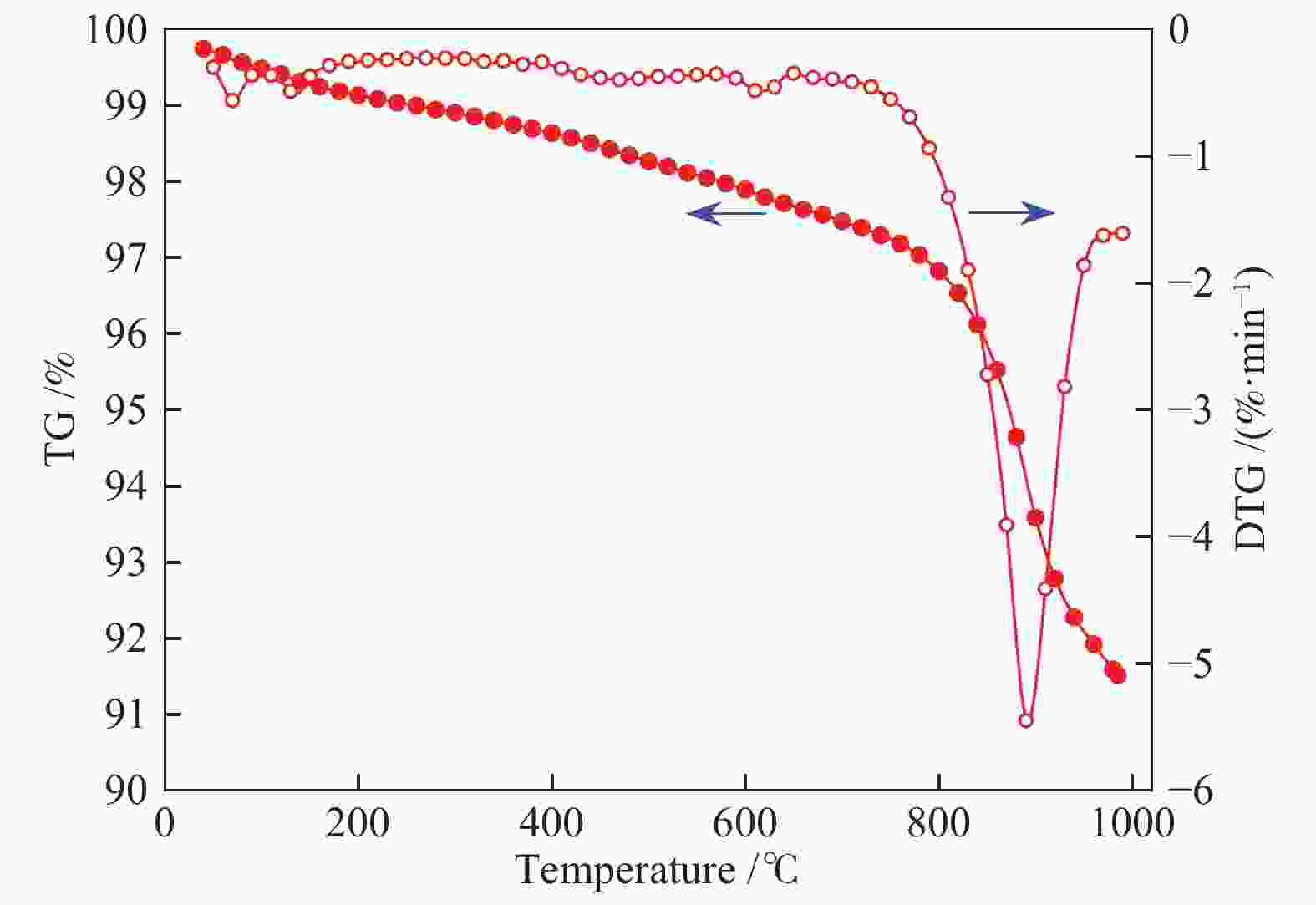
摘要: 本实验利用微型流化床反应分析仪(MFBRA)研究了棉秆半焦(CSC)与载镍橄榄石(Ni/olivine)的固-固化学链反应特性,利用模型拟合法在等温条件下对29种模型函数进行拟合计算,从中选取了最优的三种模型,计算出棉秆半焦和载氧体的固-固反应动力学。结果表明,CO和CO2是CSC与Ni/olivine反应的主要气体产物,固-固反应过程中,先析出CO后再析出CO2,CSC并不会完全转换成CO2,产气中CO的浓度比CO2大;随着反应温度的升高,产气中CO和CO2的浓度和产率增加。CO、CO2和CSC利用三种不同模型函数计算出来的活化能平均值分别为27.5、46.4和69.8 kJ/mol。利用热重研究了CSC和Ni/olivine非等温反应特性及动力学,结果表明,CSC和Ni/olivine的反应从750 ℃开始,在890 ℃时反应速率达到了峰值,非等温反应活化能为72.05 kJ/mol,这与MFBRA等温动力学活化能基本相似,说明生物质化学链气化过程中,半焦和镍基载氧体的固-固反应较容易发生。Abstract: Direct solid-solid reaction characteristics of cotton stalk char (CSC) and nickel loaded olivine (Ni/olivine) were studied using micro-fluidized bed reaction analyzer (MFBRA). Model fitting method was used to fit 29 model functions under isothermal conditions. Three optimal models were selected to calculate kinetics of the solid-solid reaction of cotton char and oxygen carries. The results show that CO and CO2 are main gas products of CSC and Ni/olivine. CO is first and then CO2 is evolved during the reaction. CSC does not completely convert into CO2, and concentration of CO is higher than that of CO2. With increasing reaction temperature, concentration and yield of CO and CO2 in the product gas increase obviously. The average activation energies for CO, CO2 and CSC are 27.5, 46.4 and 69.8 kJ/mol, respectively. The non-isothermal reaction characteristics and kinetics of CSC and Ni/olivine were studied by thermogravimetric analyzer (TGA). The results show that reaction of CSC and Ni/olivine starts at 750 ℃, and reached the peak at 890 ℃. The activation energy of non-isothermal reaction is 72.05 kJ/mol, which is in accordance with the result of MFBRA. It indicates that the solid-solid reaction between char and nickel-based oxygen carrier easily occurs in during chemical looping gasification of biomass.
-
表 1 29种固-固反应模型函数
Table 1 29 model functions of solid -solid reactions
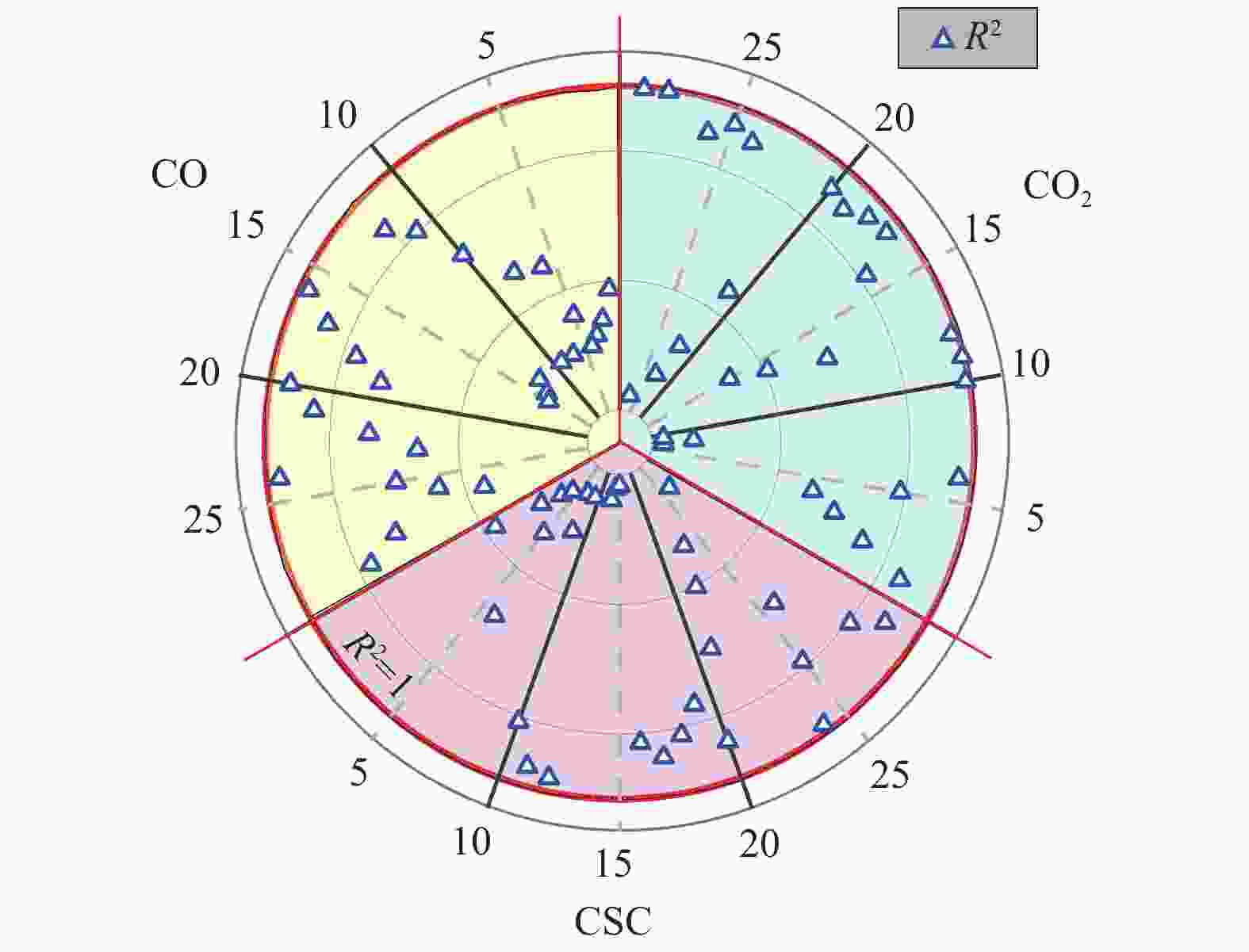
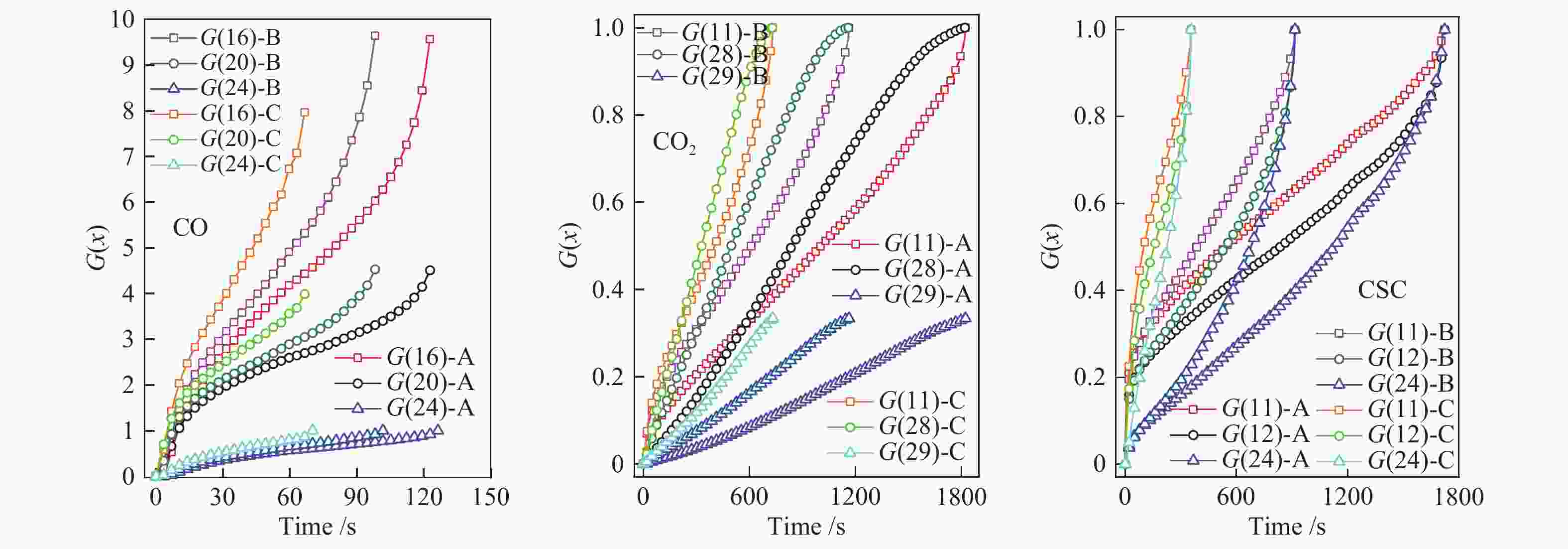
Number Function name Mechanism G(x) 1 Maple power order (Exponential nucleation) Phase boundary reaction (One-dimensional), R1, n = 1 $ x $ 2 Maple power order (Exponential nucleation) n = $ \dfrac{1}{2} $ $ {x}^{\frac{1}{2}} $ 3 Maple power order (Exponential nucleation) n = $ \dfrac{1}{3} $ $ {x}^{\frac{1}{3}} $ 4 Maple power order (Exponential nucleation) n = $ \dfrac{1}{4} $ $ {x}^{\frac{1}{4}} $ 5 Maple power order (Exponential nucleation) n = $ \dfrac{2}{3} $ $ {x}^{\frac{2}{3}} $ 6 Parabola order One-dimensional diffusion, 1D, D1 Deceleration curve of α-t $ {x}^{2} $ 7 2 order Chemical reaction, F2, Deceleration curve of α-t $ {\left(1-x\right)}^{-1} $ 8 2/3 order Chemical reaction $ {{\left(1-x\right)}}^{-\frac{1}{2}} $ 9 Reaction order Chemical reaction $ {\left(1-x\right)}^{-1}-1 $ 10 Shrink cylinder (area) Phase boundary reaction, Cylindrical Symmetry, R2, Deceleration curve of α-t, n = $ \dfrac{1}{2} $ $ 1-{\left(1-x\right)}^{\frac{1}{2}} $ 11 Shrink ball (volume) Phase boundary reaction, Spherical Symmetry, R3, Deceleration curve of α-t, n = $ \dfrac{1}{3} $ $ 1-{\left(1-x\right)}^{\frac{1}{3}} $ 12 Reaction order n = $ \dfrac{1}{4} $ $ 1-{\left(1-x\right)}^{\frac{1}{4}} $ 13 Reaction order n = 2 $ 1-{\left(1-x\right)}^{2} $ 14 Reaction order n = 3 $ 1-{\left(1-x\right)}^{3} $ 15 Reaction order n = 4 $ 1-{\left(1-x\right)}^{4} $ 16 Maple single law, First order Random nucleation and subsequent growth, There is only one core on each particle, A1, F1, Sigmoid curve of α-t, n = 1, m = 1 $ -{\rm{ln}}\left(1-x\right) $ 17 Avrami-erofeev equation Random nucleation and subsequent growth, A2, Sigmoid curve of α-t, n = $ \dfrac{1}{2} $, m = 2 $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{\frac{1}{2}} $ 18 Avrami-erofeev equation Random nucleation and subsequent growth, A3, Sigmoid curve of α-t, n = $ \dfrac{1}{3} $, m = 3 $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{\frac{1}{3}} $ 19 Avrami-erofeev equation Random nucleation and subsequent growth, A4, Sigmoid curve of α-t, n = $ \dfrac{1}{4} $, m = 4 $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{\frac{1}{4}} $ 20 Avrami-erofeev equation Random nucleation and subsequent growth, A1.5, n = $ \dfrac{2}{3} $ $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{\frac{2}{3}} $ 21 Avrami-erofeev equation Random nucleation and subsequent growth, n = 2 (Code: AE2) $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{2} $ 22 Avrami-erofeev equation Random nucleation and subsequent growth, n = 3 (Code: AE3) $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{3} $ 23 Avrami-erofeev equation Random nucleation and subsequent growth, n = 4 (Code: AE4) $ {\left[-{\rm{ln}}\left(1-x\right)\right]}^{4} $ 24 Jander equation Spherical Symmetry, 3D, D3, Deceleration curve of α-t, n = 2 $ {\left[1-{\left(1-x\right)}^{\frac{1}{3}}\right]}^{2} $ 25 Jander equation Three-dimensional diffusion, 3D, n = $ \dfrac{1}{2} $ $ {\left[1-{\left(1-x\right)}^{\frac{1}{3}}\right]}^{\frac{1}{2}} $ 26 Jander equation Three-dimensional diffusion, 2D, n = $ \dfrac{1}{2} $ $ {\left[1-{\left(1-x\right)}^{\frac{1}{2}}\right]}^{\frac{1}{2}} $ 27 Z-L-T equation Three-dimensional diffusion, 3D $ {\left[{{\left(1-x\right)}^{-}}^{\frac{1}{3}}-1\right]}^{2} $ 28 Valensi equation Two-dimensional diffusion, Cylindrical Symmetry, 2D, D2, Deceleration curve of α-t $ x + \left(1-x\right){\rm{ln}}\left(1-x\right) $ 29 Ginstling-brounshtein equation Spherical Symmetry, 3D, D4, Deceleration curve of α-t $ 1-\dfrac{2}{3}x-{\left(1-x\right)}^{\frac{2}{3}} $ 表 2 基于MFBRA等温反应动力学参数
Table 2 Kinetic parameters of isothermal reaction by MFBRA
Sample Model function E/(kJ·mol−1) A R2 CO G(16) 27.54 1.53 0.995 G(20) 28.20 0.74 0.996 G(24) 26.71 0.16 0.994 CO2 G(11) 47.23 0.11 0.977 G(28) 45.22 0.12 0.989 G(29) 46.62 0.04 0.989 CSC G(11) 69.41 1.35 0.995 G(12) 70.51 1.51 0.999 G(24) 69.36 1.70 0.991 表 3 基于热重非等温反应动力学参数
Table 3 Kinetic parameters of non-isothermal reaction by TGA
Sample E/(kJ·mol−1) A R2 CSC 72.05 1.33 0.953 -
[1] 吴志强, 张博, 杨伯伦. 生物质化学链转化技术研究进展[J]. 化工学报, 2019, 70(8): 2835−2853.WU Zhi-qiang, ZHANG Bo, YANG Bo-lun. Research progress on biochemical chain transformation technology[J]. CIESC J, 2019, 70(8): 2835−2853. [2] LUO S W, ZENG L, FAN L S. Chemical looping technology: Oxygen carrier characteristics[J]. Annu Rev Chem Biomol Eng,2015,6(1):53−75. doi: 10.1146/annurev-chembioeng-060713-040334 [3] WANG P, MEANS N, SHEKHAWAT D, BERRY D, MASSOUDI M. Chemical-looping combustion and gasification of coals and oxygen carrier development: A brief review[J]. Energies,2015,8(10):10605−10635. doi: 10.3390/en81010605 [4] LUO S W, SUN Z C, BAOX G, FAN L S. Role of metal oxide support in redox reactions of iron oxide for chemical looping applications: experiments and density functional theory calculations[J]. Energy Environ Sci,2011,4(9):3661−3667. doi: 10.1039/c1ee01325d [5] VARDANYAN A, KHACHATRYAN A, ZARUHI M. CHEMICAL OXIDATION INTEGRATED INTO BIOLEACHING OF PYRITE AND CHALCOPYRITE USING IMMOBILIZED BIOMASS[J]. Environ Eng Manage J,2018,17(4):897−904. doi: 10.30638/eemj.2018.090 [6] ZHANG P, LI S, GUO P. Investigating the kinetics of liquid-free, OSDA-free ZSM-5 zeolite synthesis from iron ore tailings[J]. Int J Chem Kinet,2020,52(6):403−412. doi: 10.1002/kin.21359 [7] NELSON T, WATT J G V D, LAUDAL D, FEILEN H, SRINIVASACHAR S. Reactive jet and cyclonic attrition analysis of ilmenite in chemical looping combustion systems[J]. Int J Green Gas Control,2019,91:102837. doi: 10.1016/j.ijggc.2019.102837 [8] PAN Y, TURSUN Y, ABDUHANI H, DILNUR T. Chemical looping gasification of cotton stalk with bimetallic Cu/Ni/olivine as oxygen carrier[J]. Int J Energy Res,2020,44:7268−7282. doi: 10.1002/er.5439 [9] CHEN L Y, BAO J H, KONG L, COMBS M, HEATHER S, ZHEN F, LIU K L. The direct solid-solid reaction between coal char and iron-based oxygen carrier and its contribution to solid-fueled chemical looping combustion[J]. Appl Energy,2016,184:9−18. doi: 10.1016/j.apenergy.2016.09.085 [10] GUO Q J, CHENG Y, LIU Y Z, JIA W H, RYU, H J. Coal chemical looping gasification for syngas generation using an iron-based oxygen carrier[J]. Ind Eng Chem Res,2014,53(1):78−86. doi: 10.1021/ie401568x [11] TIAN X, ZHAO H B, WANG K, MA J C, ZHENG C G. Performance of cement decorated copper ore as oxygen carrier in chemical-looping with oxygen uncoupling[J]. Int J Green Gas Control,2015,41:210−218. [12] 冉景煜, 张松, 秦昌雷, 禹建功, 付范萱, 杨琳. 生物质半焦/铜基载氧体气化反应特性研究[J]. 燃料化学学报,2014,42(11):1316−1323.RAN Jing-yu, ZHANG Song, QIN Chang-lei, YU Jian-gong, FU Fan-xun, YANG Lin. Gasification rea ctivity of biomass char with oxygen carrier CuO[J]. J Fuel Chem Technol,2014,42(11):1316−1323. [13] 程丹琰, 雍其润, 龚本根, 赵永椿, 张军营. 煤和生物质化学链气化中铜基载氧体与灰分的碳热反应研究[J]. 燃料化学学报,2020,48(1):18−27.CHENG Dan-yan, YONG Qi-run, GONG Ben-gen, ZHAO Yong-chun, ZHANG Jun-ying. Carbothermal interaction between Cu-based oxygen carrier and ash minerals in the chemical-looping gasification of coal and biomass[J]. J Fuel Chem Technol,2020,48(1):18−27. [14] 曾玺, 王芳, 韩江则, 张聚伟, 刘云义, 汪印, 余剑, 许光文. 微型流化床反应分析及其对煤焦气化动力学的应用[J]. 化工学报,2013,64(1):289−296. doi: 10.3969/j.issn.0438-1157.2013.01.032ZENG Xi, WANG Fang, HAN Jiang-ze, ZHANG Ju-wei, LIU Yun-yi, WANG Yin, YU Jian, XU Guang-wen. Micro fluidized bed reaction analysis and its application to coal char gasification kinetics[J]. CIESC J,2013,64(1):289−296. doi: 10.3969/j.issn.0438-1157.2013.01.032 [15] GUO F Q, DONG Y P, LV Z C, FAN P F, YANG S, DONG L. Kinetic behavior of biomass under oxidative atmosphere using a micro-fluidized bed reactor[J]. Energy Conver Manage,2016,108:210−218. doi: 10.1016/j.enconman.2015.11.014 [16] 王芳, 曾玺, 王永刚, 余剑, 岳君容, 张建岭, 许光文. 微型流化床与热重测定煤焦非等温气化反应动力学对比[J]. 化工学报,2015,66(5):1716−1722.WANG Fang, ZENG Xi, WANG Yong-gang, YU Jian, YUE Jun-rong, XU Guang-wen. Comparation of non-isothermal coal char gasification in micro fluidized bed and thermogravimetric analyzer[J]. CIESC J,2015,66(5):1716−1722. [17] ZENG X, WANG F, AdAMUA M H, ZHANG L J, HAN Z N, XU G W. High-temperature drying behavior and kinetics of lignite tested by the micro fluidization analytical method[J]. Fuel,2019,253:180−188. doi: 10.1016/j.fuel.2019.05.025 [18] HE K, ZHENG Z, CHEN Z. Multistep reduction kinetics of Fe3O4 to Fe with carbon monoxide in a micro fluidized bed reaction analyzer[J]. Powder Technol,2019,360:1−10. [19] GUO F Q, PENG K Y, ZHAO X M, JIANG X C, LIN Q, GUO C L, RAO Z H. Influence of impregnated copper and zinc on the pyrolysis of rice husk in a micro-fluidized bed reactor: Characterization and kinetics[J]. Int J Hydrogen Energy,2018,43(46):21256−21268. doi: 10.1016/j.ijhydene.2018.10.013 [20] KUBA M, KIRNBAUER F, HOFBAUER H. Influence of coated olivine on the conversion of intermediate products from decomposition of biomass tars during gasification[J]. Biomass Convers Biorefin,2017,7(1):11−21. doi: 10.1007/s13399-016-0204-z [21] 王芳 曾玺, 韩江则, 张聚伟, 刘云义, 汪印, 李奡明, 余剑, 许光文. 微型流化床与热重测定煤焦-CO2气化反应动力学的对比研究[J]. 燃料化学学报, 2013, 41(4): 407−413.ZENG Xi, WANG Fang, HAN Jiang-ze, ZHANG Ju-wei, WANG Yin, LI Ao-ming, YU Jian, XU Guang-wen. Comparation of char gasification kinetics studied by micro fluidized bed and by thermogravimetric analyzer.[J] J Fuel Chem Technol, 2013, 41(4): 407−413. [22] YU J, YAO C B, ZENG X, GENG S, DONG L, WANG Y, GAO S Q, XU G W. Biomass pyrolysis in a micro-fluidized bed reactor: Characterization and kinetics[J]. Chem Eng J,2011,168(2):839−847. doi: 10.1016/j.cej.2011.01.097 [23] LIU Y, GUO F Q, LI X L, LIT T, PENG K Y, GUO C L, CHANG J F. Catalytic effect of iron and nickel on gases formation from fast biomass pyrolysis in a micro-fluidized bed reactor: A kinetic study[J]. Energy Fuels,2017,31(11):12278−12287. doi: 10.1021/acs.energyfuels.7b02214 [24] YAN X Y, HU J J, ZHANG Q G, ZHAO S H, DANG J T, WANG W. Chemical-looping gasification of corn straw with Fe-based oxygen carrier: Thermogravimetric analysis[J]. Bioresour Technol,2020,303:122904. [25] 郭飞强, 刘元, 郭成龙, 董玉平. 微型流化床内碱金属和碱土金属对稻壳热解动力学的影响特性[J]. 化工学报,2017,68(10):3795−3804.GUO Fei-qiang, LIU Yuan, GUO Cheng-long, DONG Yu-ping. Influence of AAEM on kinetic characteristics of rice husk pyrolysis in micro-fluidized bed reactor[J]. CIESC J,2017,68(10):3795−3804. [26] 糜梦星, 邢献军, 张学飞, 陈泽宇, 朱成成, 付一轩. 基于分布式改良Coats-Redfern法的梧桐叶燃烧动力学研究[J]. 太阳能学报, 2019, 40(9): 2672-2679.MI Meng-xing, XING Xian-jun, ZHANG Xue-fei, CHEN Ze-yu, ZHU Cheng-cheng, FU Yi-xuan. Study on combustion kinetics of phoenix tree’s leaves based on distributed Coats-Redfern (Modified) method.[J] Acta Solar Energy, 2019, 40(9): 2672-2679. [27] 杨小芹, 徐绍平, 胡冠, 刘长厚. 不同矿源橄榄石对催化苯水蒸气重整的影响[J]. 催化学报,2009,30(6):497−502. doi: 10.3321/j.issn:0253-9837.2009.06.005YANG Xiao-qing, XU Shao-ping, HU Guan, LIU Chang-hou. Effects of olivines from different quarries on the steam reforming of benzene[J]. Chin J Catal,2009,30(6):497−502. doi: 10.3321/j.issn:0253-9837.2009.06.005 [28] DEVI L, PTASINSKI K J, JANSSEN F J J G. Pretreated olivine as tar removal catalyst for biomass gasifiers: investigation using naphthalene as model biomass tar[J]. Fuel Process Technol,2005,86(6):707−730. doi: 10.1016/j.fuproc.2004.07.001 [29] MENG J G, WANG X B, ZHAO X B, ZHANG A Q, HUANG Z. Highly abrasion resistant thermally fused olivine as in-situ catalysts for tar reduction in a circulating fluidized bed biomass gasifier[J]. Bioresour Technol,2018,268:212−220. doi: 10.1016/j.biortech.2018.07.135 [30] TURSUN Y, XU S P, ABULIKEMU A, DILNUR T. Biomass gasification for hydrogen rich gas in a decoupled triple bed gasifier with olivine and NiO/olivine[J]. Bioresour Technol,2019,272:241−248. [31] 赵海波, 刘黎明, 徐迪, 郑楚光, 刘国军, 蒋林林. 气体燃料化学链燃烧技术中的溶胶凝胶Ni基氧载体研究[J]. 燃料化学学报,2008,36(3):261−266. doi: 10.1016/S1872-5813(08)60020-1ZHAO Hai-bo, LIU Li-ming, XU Di, ZHEN Chun-guang, LIU Guo-jun, JIANG Lin-lin. NiO/NiAl204 oxygen carriers prepared by sol-gel for chemical-looping combustion fueled by gas[J]. J Fuel Chem Technol,2008,36(3):261−266. doi: 10.1016/S1872-5813(08)60020-1 [32] WANG F, ZENG X, WANG Y G, YU J, XU G W. Characterization of coal char gasification with steam in a micro-fluidized bed reaction analyzer[J]. Fuel Process Technol,2016,141:2−8. [33] ZHANG Y M, SUN G G, GAO S Q, XU G W. Regeneration Kinetics of Spent FCC Catalyst via Coke Gasification in a Micro Fluidized Bed[J]. Procedia Eng,2015,102:1758−1765. [34] 余剑, 李强, 段正康, 朱剑虹, 岳君容, 李养明, 许光文. 微型流化床中的等温微分反应特性[J]. 中国科学: 化学,2011,41(1):152−160.YU Jian, LI Qiang, DUAN Zheng-kang, ZHU Jian-hong, YUE Jun-rong, LI Yang-ming, XU Guang-wen. Isothermal differential characteristics of the reaction in micro fluidized bed[J]. Sci Sin Chem,2011,41(1):152−160. -





 下载:
下载: