Catalytic activity of Ni/SSZ-13 catalyst for CO2 methanation
-
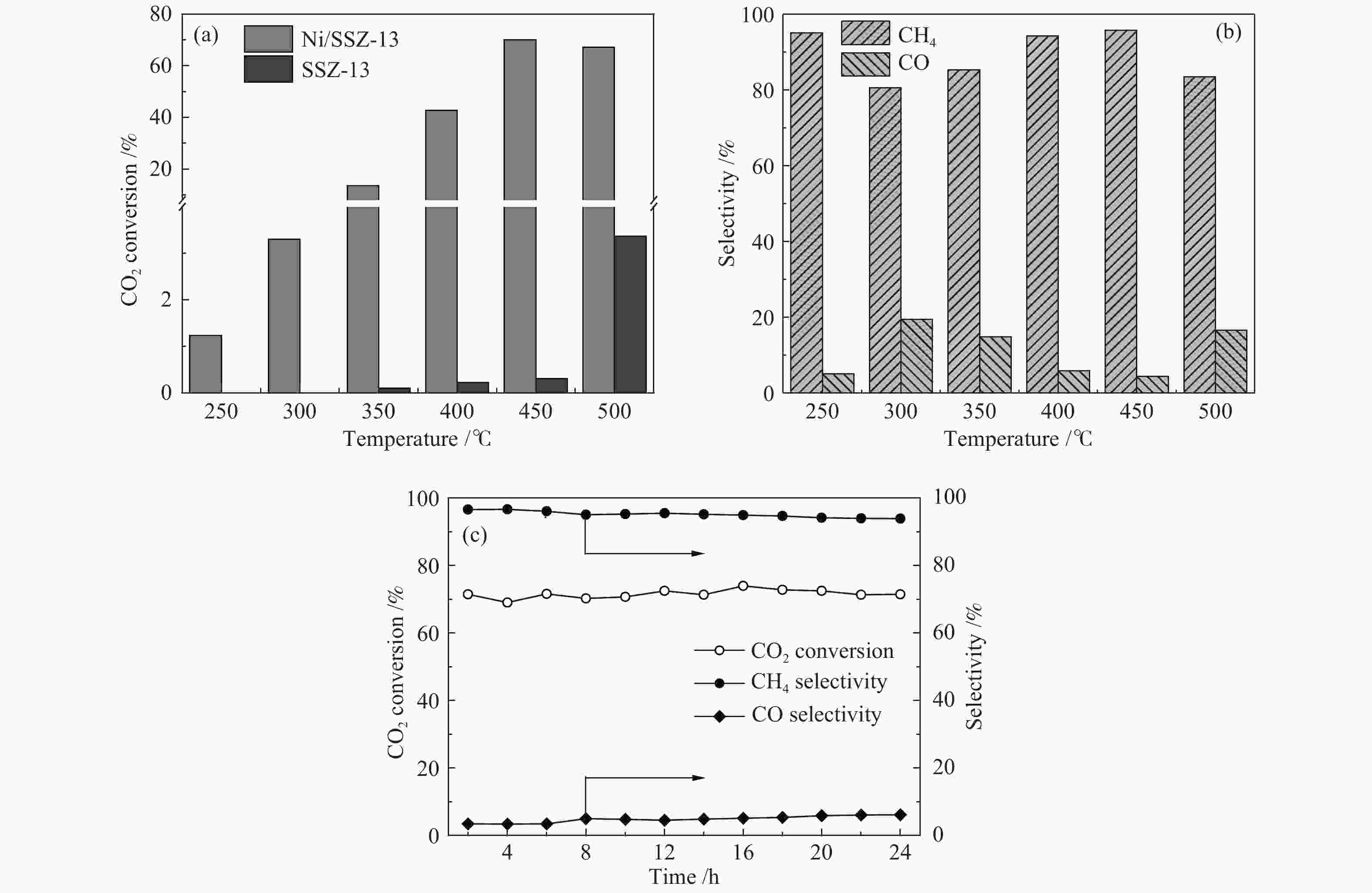
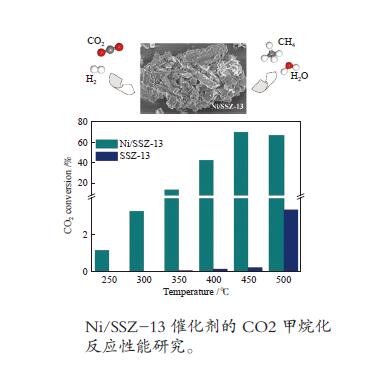
摘要: 采用浸渍法制备了Ni/SSZ-13催化剂,并研究了催化剂的CO2甲烷化反应性能。通过N2物理吸附-脱附、X射线衍射、扫描电子显微镜、透射电子显微镜、X射线光电子能谱等表征分析方法对催化剂的质构特性及物理化学性质进行了研究。结果表明,Ni/SSZ-13催化剂在250 ℃开始表现出CO2甲烷化催化活性;在450 ℃,Ni/SSZ-13表现出最佳的催化反应活性,CO2转化率和CH4选择性分别为70%和95%。SSZ-13载体表现出长方体结构,孔结构主要为微孔,并含有部分介孔,为镍物质的分散提供较大的比表面积。煅烧后的催化剂在500 ℃的还原过程中,NiO被H2还原成金属Ni,金属Ni是CO2甲烷化的主要活性组分。Abstract: Ni/SSZ-13 catalyst was prepared by the impregnation method and used to catalyze CO2 methanation in a fixed-bed reactor. The microstructure and physical-chemical properties of catalyst were characterized by N2 physical adsorption, X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray photoelectron spectroscopy (XPS). The results show that Ni/SSZ-13 catalyst initiates catalytic activity towards CO2 methanation at 250 ℃. Ni/SSZ-13 catalyst shows the optimal performance for CO2 methanation at 450 ℃. CO2 conversion rate and CH4 selectivity are 70% and 95%, respectively. SSZ-13 support shows a cuboid structure. Moreover, SSZ-13 support mainly possesses microporous and contains some mesopores, which provide larger surface area for nickel species dispersion. During the reduction process of the calcined catalyst at 500 ℃, NiO is reduced by H2 into metal Ni, which is the main active component for CO2 methanation.
-
Key words:
- carbon dioxide /
- methanation /
- Ni/SSZ-13 /
- catalyst /
- catalytic activity
-
图 4 SSZ-13载体和Ni/SSZ-13催化剂的SEM、TEM和EDS元素分布图
Figure 4 SEM, TEM images and EDS elemental mapping of SSZ-13 zeolite and Ni/SSZ-13 catalyst
(a): SEM image of SSZ-13 support; (b): SEM image of fresh Ni/SSZ-13 catalyst; (c): SEM image of spent Ni/SSZ-13 catalyst; (d): TEM image of fresh Ni/SSZ-13 catalyst; (e): TEM image of spent Ni/SSZ-13 catalyst; (f)−(h): EDS elemental mapping of SSZ-13 support; (i)−(l): EDS elemental mapping of fresh Ni/SSZ-13 catalyst
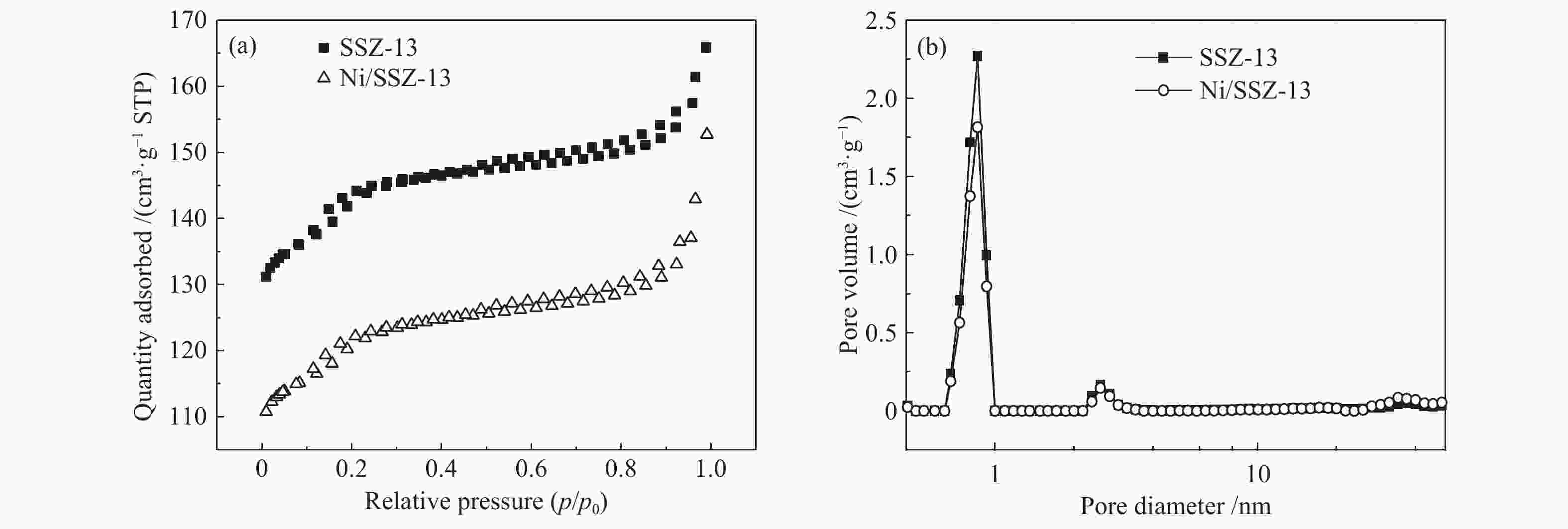
表 1 样品的比表面积及孔结构参数
Table 1 The specific surface area and pore structure parameters of SSZ-13 zeolite and Ni/SSZ-13 catalyst
Samples Specific surface
area A/(m2·g−1)Micropore Area
A/(m2·g−1)Pore volume
v/(cm3·g−1)Micropore volume
v/(cm3·g−1)Average pore
size d/nmSSZ-13 449.20 337.69 0.28 0.17 3.61 Ni/SSZ-13 385.51 284.03 0.24 0.14 4.53 表 2 Ni/SSZ-13催化剂的表面元素含量
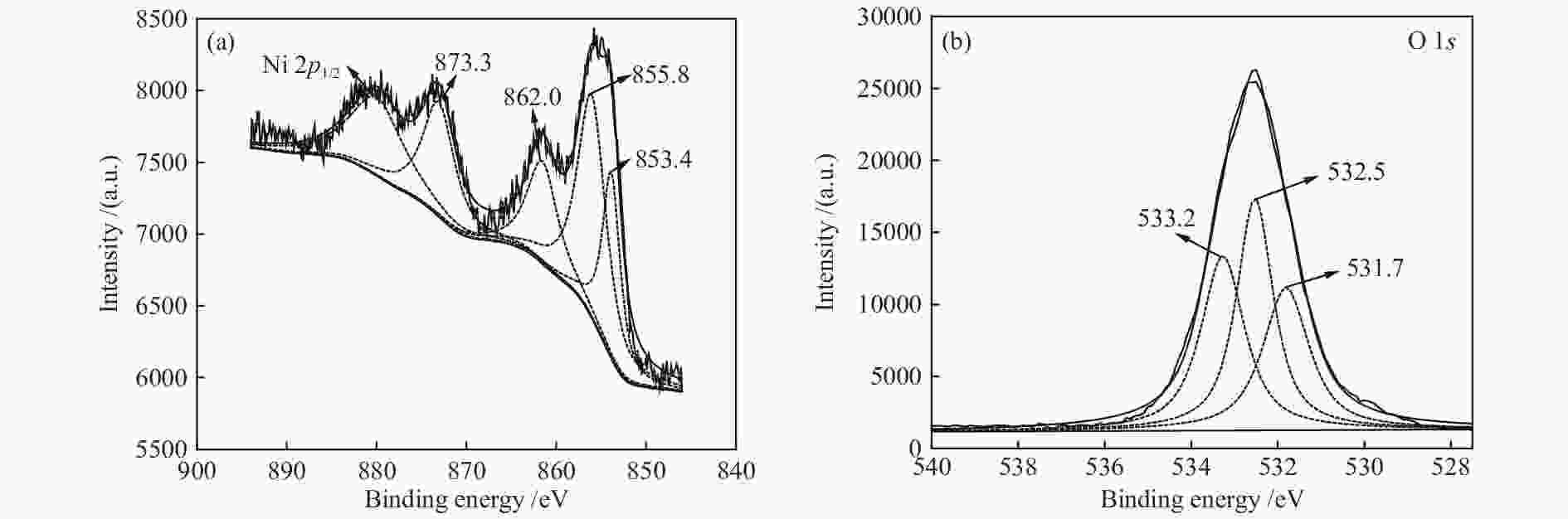
Table 2 Surface atom concentration of Ni/SSZ-13 catalyst
C 1s O 1s Al 2p Si 2p Ni 2p Peak BE 284.77 532.58 74.79 103.32 854.94 Atomic/% 23.52 51.43 1.14 20.65 3.27 -
[1] BAINS P, PSARRAS P, WILCOX J. CO2 capture from the industry sector[J]. Prog Energy Combust Sci,2017,63:146−172. doi: 10.1016/j.pecs.2017.07.001 [2] ZHONG J, YANG X, WU Z, LIANG B L, HUANG Y Q, ZHANG T. State of the art and perspectives in heterogeneous catalysis of CO2 hydrogenation to methanol[J]. Chem Soc Rev,2020,49:1385−1413. doi: 10.1039/C9CS00614A [3] NGUYEN T N, DINH C T. Gas diffusion electrode design for electrochemical carbon dioxide reduction[J]. Chem Soc Rev,2020,49(21):7488−1504. doi: 10.1039/D0CS00230E [4] MARQUES MOTA F, KIM D H. From CO2 methanation to ambitious long-chain hydrocarbons: Alternative fuels paving the path to sustainability[J]. Chem Soc Rev,2019,48(1):205−259. doi: 10.1039/C8CS00527C [5] NSCH S, SCHNEIDER J, MATTHISCHKE S, SCHLÜTER M, GÖTZ M L, JONATHAN P, PRASEETH B, Siegfried. Review on methanation – From fundamentals to current projects[J]. Fuel,2016,166:276−296. doi: 10.1016/j.fuel.2015.10.111 [6] FRONTERA P, MACARIO A, FERRARO M, ANTONUCCI P. Supported catalysts for CO2 methanation: A review[J]. Catalysts,2017,7(2):59. [7] YANG Y, LIU J, LIU F, WU D W. Reaction mechanism of CO2 methanation over Rh/TiO2 catalyst[J]. Fuel,2020,276:118093. doi: 10.1016/j.fuel.2020.118093 [8] AGNELLI M, KOLB M, MIRODATOS C. CO hydrogenation on a nickel catalyst. : 1. Kinetics and modeling of a low-temperature sintering process[J]. J Catal,1994,148(1):9−21. doi: 10.1006/jcat.1994.1180 [9] VAN DEELEN T W, HERN NDEZ MEJ A C, DE JONG K P. Control of metal-support interactions in heterogeneous catalysts to enhance activity and selectivity[J]. Nat Catal,2019,2(11):955−970. doi: 10.1038/s41929-019-0364-x [10] IPEK B, POLLOCK R A, BROWN C M, UNER D, LOBO R F. H2 adsorption on Cu(I)-SSZ-13[J]. J Phys Chem C,2018,122(1):540−548. doi: 10.1021/acs.jpcc.7b09963 [11] ARTZ J, M LLER T E, THENERT K, KLEINEKORTE J, MEYS R, STERNBERG A, BARDOW A, LEITNER W. Sustainable conversion of carbon dioxide: An integrated review of catalysis and life cycle assessment[J]. Chem Rev,2018,118(2):434−504. doi: 10.1021/acs.chemrev.7b00435 [12] 付长亮 王, 王少鹏, 王东娇, 赵雅文, 梅梦迪. 不同金属改性Ni/KIT-6催化剂的制备及其甲烷化性能研究[J]. 燃料化学学报,2020,48(4):476−482. doi: 10.3969/j.issn.0253-2409.2020.04.011FU Chang-liang, WANG Li-ping, WANG Shao-peng, WANG Dong-jiao, ZHAO Ya-wen MEI Meng-di. Preparation of Ni/ Kit-6 catalysts modified by different metals and study on their methanation properties[J]. J Fuel Chem Technol,2020,48(4):476−482. doi: 10.3969/j.issn.0253-2409.2020.04.011 [13] 曹红霞. CO/CO2甲烷化催化剂的制备、表征及性能研究[D]. 徐州: 中国矿业大学, 2018.CAO Hong-xia. Study on preparation, characterization and performance of catalyst for CO/CO2 methanation[D]. Xuzhou: China University of Mining and Technology, 2018. [14] 聂望欣. 新型二氧化碳甲烷化催化剂制备及性能的研究[D]. 上海: 上海大学, 2018.NIE Wang-xin. synthesis and catalytic properties of novel catalysts for carbon dioxide methanation[D]. Shanghai: Shanghai University, 2018. -




 下载:
下载: