Homogeneous reaction mechanism in arsenic/hydrogen/oxygen system
-
摘要: 采用量子化学的方法,利用量子化学软件Gaussian09和GaussView,在B3LYP/6-311G(3df, 3pd)理论水平下,对砷/氢/氧体系燃烧反应的微观机理进行研究。优化了13个基元反应的反应物、中间体、过渡态以及产物的构型,并通过振动频率和内禀反应坐标验证反应真实性。采用KiSThelP软件包基于传统过渡态理论拟合计算出动力学参数。本研究对砷燃烧体系中部分重要基元反应进行研究,便于后续建立砷的燃烧动力学模型。Abstract: Herein quantum chemistry methods were used to discuss the microscopic mechanism in the arsenic/hydrogen/oxygen reaction system during combustion by Gaussian09 and GaussView at the theoretical level of B3LYP/6-311G (3df, 3pd). Configuration of the reactants, intermediates, transition states, and products of 13 elementary reactions were optimized and the authenticity of these reactions were verified by frequency and intrinsic reaction coordinates. Finally, the kinetic parameters were calculated through KiSThelP based on the classic transition state theory. This study focused on some of the most important reations in the arsenic/hydrogen/oxygen system, which makes sense to establish arsenic dynamic model during combustion.
-
Key words:
- coal combustion /
- arsenic /
- microscopic mechanism /
- DFT
-
表 1 计算值与文献值对比
Table 1 Comparison of calculated values and literature values
Species Bond length(r/Å)
and angles(α/°)Calculated
resultReferenced
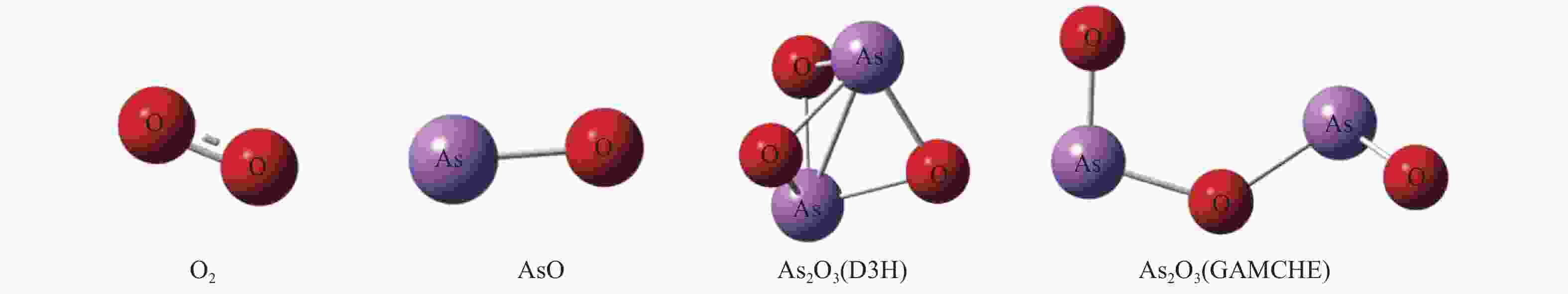
value[17]O2 r(O−O) 1.20255 1.239 AsO r(As−O) 1.62401 1.624 As2O3(D3H) r(As−O) 1.84376 1.836 r(As-As) 2.38940 2.375 α(As−O−As) 80.77769 80.6 As2O3(GAUCHE) r(As−O) 1.61949 1.610 r(O−As) 1.78284 1.794 α(As−O−As) 105.77862 106.3 α(O−As−O) 129.64809 133.8 表 2 砷参与的基元反应
Table 2 Elementary reactions of Arsenic
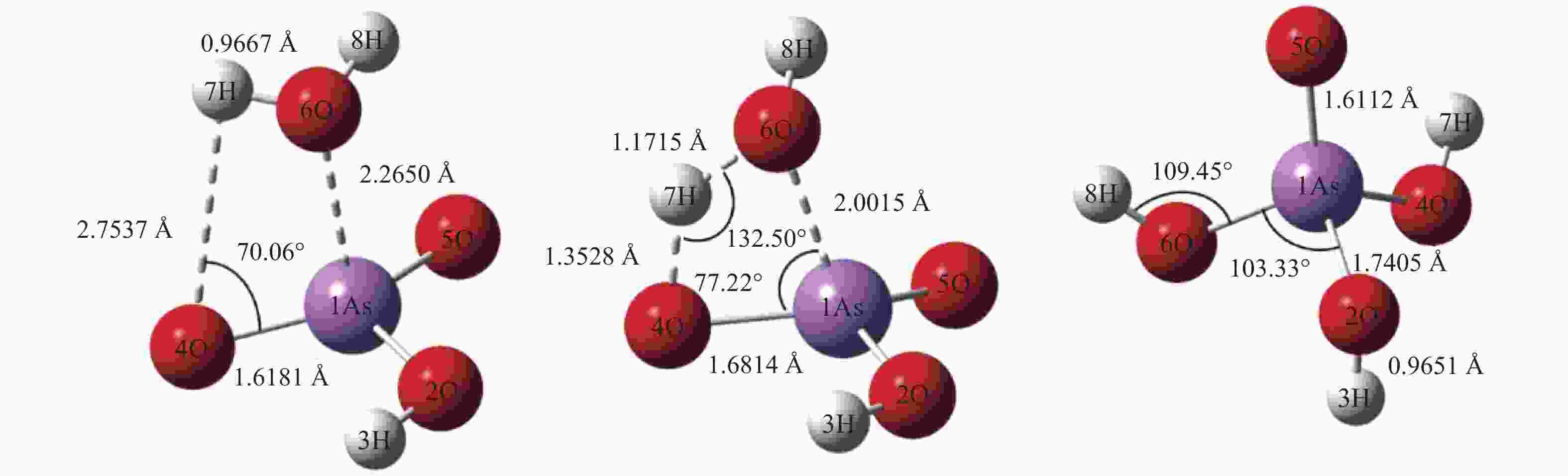
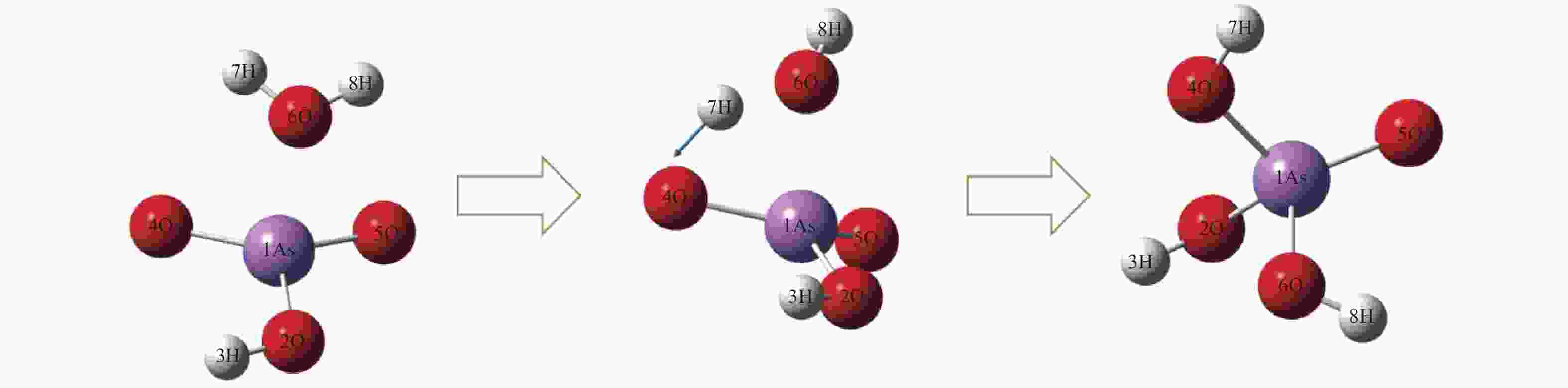
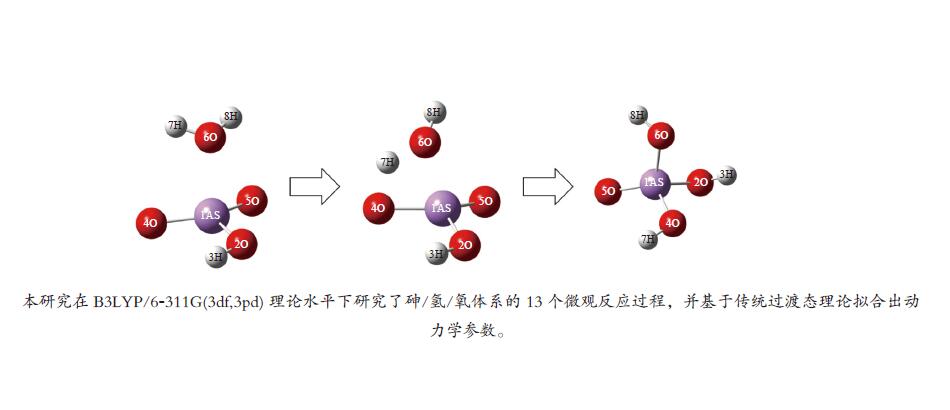
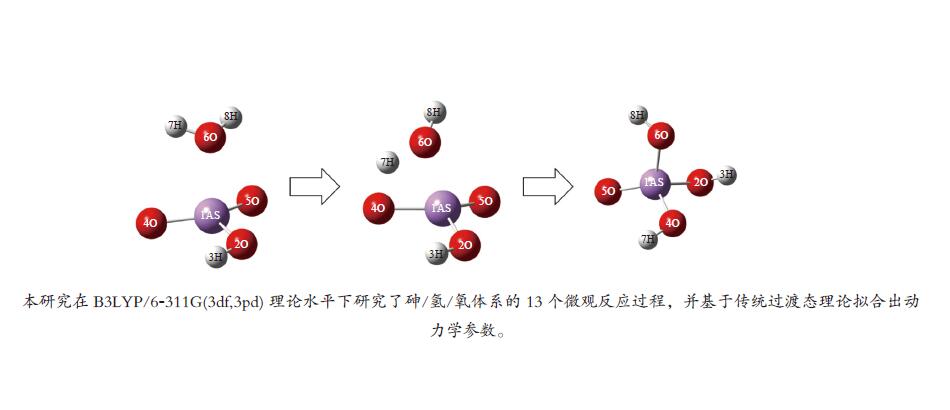
No. Elementary reactions 1 As+AsO2 = 2AsO 2 As+H2O = AsOH+H 3 AsO+H2O = AsOOH+H 4 AsOOH+H = AsO2+H2 5 AsO2+H2O = AsO2OH+H 6 AsO2OH+H = AsO(OH)2 7 AsOOH+H2O = As(OH)3 8 AsOOH+H = As(OH)2 9 As(OH)3+H = AsO(OH)2+H2 10 AsOH+H2O = As(OH)2 11 As(OH)2+H2O = As(OH)3+H 12 H3AsO4+H = AsO(OH)2+H2O 13 AsO2OH+H2O = H3AsO4 表 3 AsO2OH+H2O = H3AsO4反应各稳定点振动频率
Table 3 Frequencies of stable points of AsO2OH+H2O = H3AsO4
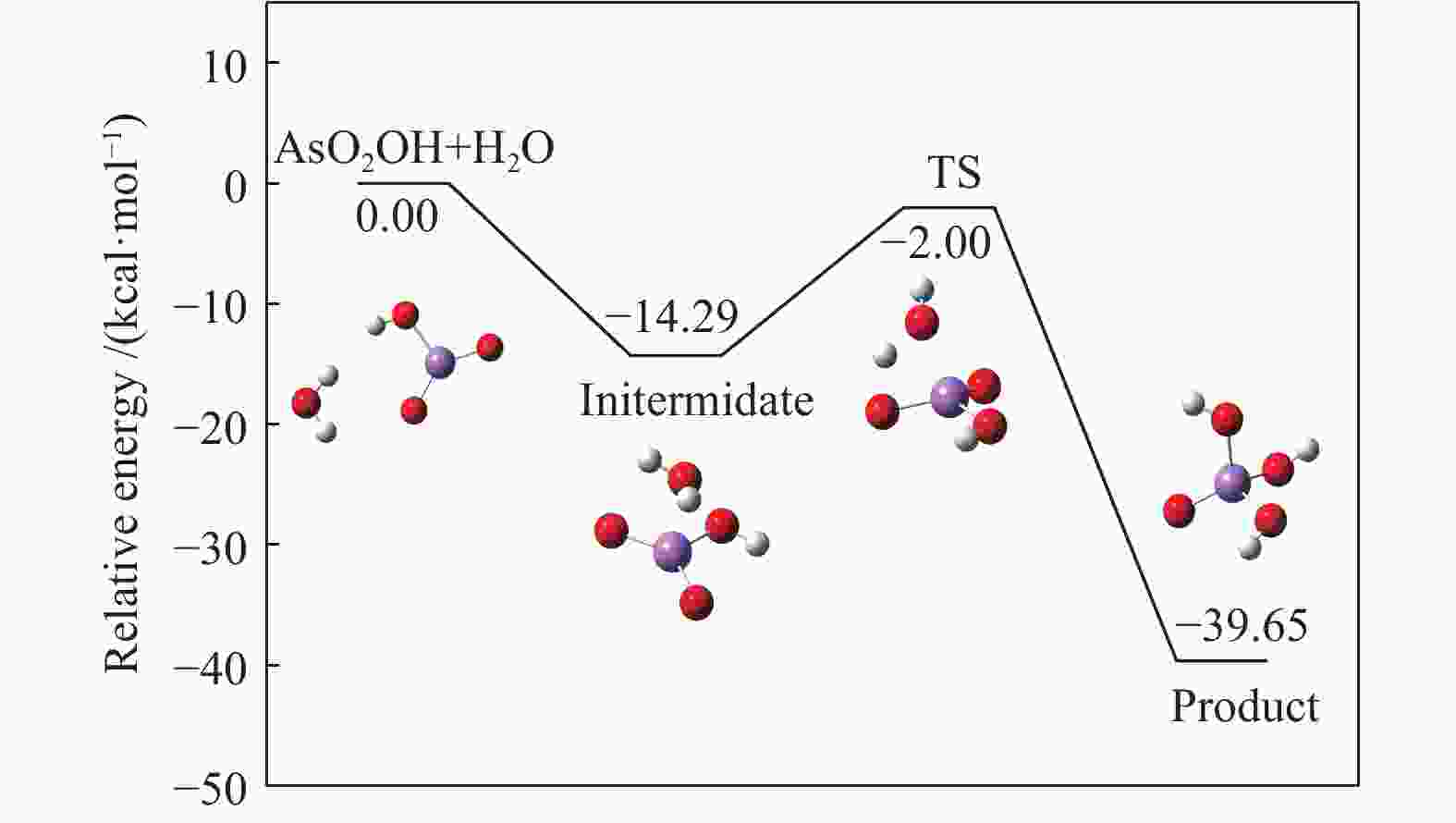
No. Vibrations frequencies/cm−1 AsO2OH 245.63, 281.93, 301.64, 459.23, 726.01, 947.36, 1018.14, 1073.63, 3771.60 H2O 1641.28, 3819.58, 3912.16 TS −1358.59, 165.93, 255.41, 266.03, 293.69, 370.60, 433.77, 553.71, 689.49,763.14, 806.75, 873.86, 1011.59, 1059.90, 1362.22, 1962.70, 3786.56, 3788.99 H3AsO4 123.08, 142.48, 208.18, 220.49, 281.94, 289.95, 300.96, 416.02, 548.75, 617.39, 718.8, 936.82, 1014.39, 1064.23, 1610.44, 3738.20, 3790.15, 3835.85 表 4 AsO2OH+H2O = H3AsO4反应过程能量变化
Table 4 Energy change in AsO2OH+H2O = H3AsO4 reaction process
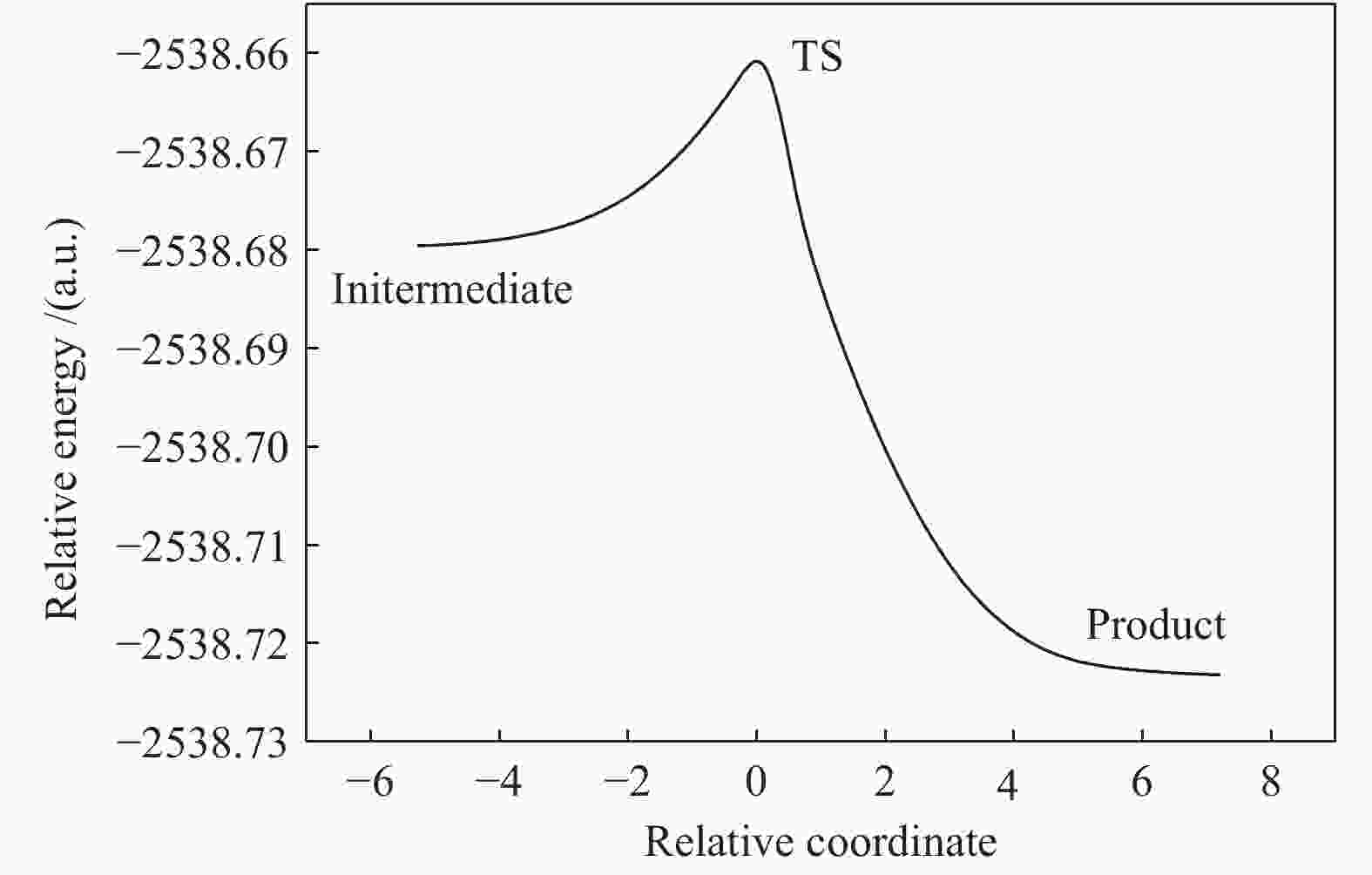
B3LYP/(a.u.) ZEP/(a.u.) Etot/(a.u.) Erel/(kcal·mol−1) AsO2OH+ H2O −2538.657127 0.041458 −2538.615669 0 Initermediate −2538.679596 0.045239 −2538.634357 −14.2883 TS −2538.660815 0.041958 −2538.618857 −2.0000 H3AsO4 −2538.723255 0.044405 −2538.678850 −39.6467 Ea, for/(kcal·mol−1) 12.2883 Ea, rev/(kcal·mol−1) 37.6467 表 5 砷参与的各基元反应反应动力学参数
Table 5 Kinetic parameters of each elementary reaction of Arsenic
No. Elementary reactions A n Ea/(cal·mol−1) 1 As+AsO2 = 2AsO 3.312 × 1012 0.01 990 2 As+H2O = AsOH+H 5.74 × 105 2.53 9548 3 AsO2OH+H2 = AsOOH+H2O 8.07 × 102 2.81 43100 4 AsOOH+H = AsO2+H2 5.06 × 107 1.85 7303 5 AsO2+H2O = AsO2OH+H 5.00 ×10−2 3.95 48637 6 AsO2OH+H = AsO(OH)2 1.38 × 109 1.57 382 7 AsOOH+H2O = As(OH)3 1.09 × 109 0.53 7330 8 AsOOH+H = As(OH)2 6.56 × 108 1.57 119 9 As(OH)3+H = AsO(OH)2+H2 6.62 × 107 1.9 5403 10 AsOH+H2O = As(OH)2 3.991 × 10−1 4.04 40720 11 As(OH)2+H2O = As(OH)3+H 3.22 × 10−2 4.04 34765 12 H3AsO4+H = AsO(OH)2+H2O 2.97 × 108 1.67 10179 13 AsO2OH+H2O = H3AsO4 9.80 × 109 0.52 7982 -
[1] CHEN J, LIU G, KANG Y, WU B, SUN R, ZHOU C, WU D. Coal utilization in China: Environmental impacts and human health[J]. Environ Geochem Health,2014,36(4):735−753. doi: 10.1007/s10653-013-9592-1 [2] 田贺忠, 曲益萍. 2005年中国燃煤大气砷排放清单[J]. 环境科学,2014,36(4):735−753.TIAN He-zhong, QU Yi-ping. Inventories of atmospheric arsenic emissions from coal combustion in China, 2005[J]. Environ Sci,2014,36(4):735−753. [3] 黄秋婵, 韦友欢, 吴颖珍. 砷污染对人体健康的危害效应研究[J]. 微量元素与健康研究,2009,26(4):65−67.HUANG Qiu-chan, WEI You-huan, WU Ying-zhen. Research on the harmful effects of arsenic pollution on human health[J]. Stud Trace Elem Health,2009,26(4):65−67. [4] 肖雨亭. 脱硝催化剂的砷中毒[J]. 电力科技与环保,2019,35(5):6−10. doi: 10.3969/j.issn.1674-8069.2019.05.002XIAO Yu-ting. Arsenic poisoning in SCR catalyst[J]. Electr Power Environ Prot,2019,35(5):6−10. doi: 10.3969/j.issn.1674-8069.2019.05.002 [5] 刘迎晖, 郑楚光, 游小清, 郭欣. 燃煤过程中易挥发有毒痕量元素的相互作用[J]. 燃烧科学与技术,2001,(4):243−247. doi: 10.3321/j.issn:1006-8740.2001.04.007LIU Ying-hui, ZHENG Chu-guang, YOU Xiao-qing, GUO Xin. Interaction between most volatile toxic trace elements during coal combustion[J]. J Combust Sci Technol,2001,(4):243−247. doi: 10.3321/j.issn:1006-8740.2001.04.007 [6] DUKER A A, CARRANZE E J M, HALE M. Arsenic geochemistry and health[J]. Environ Int,2005,31(5):631−641. doi: 10.1016/j.envint.2004.10.020 [7] 王臣, 徐明厚, 刘晶, 郑楚光. 基于量子化学的燃煤过程痕量元素反应机理研究及其进展[J]. 热力发电,2005,(4):15−19+40. doi: 10.3969/j.issn.1002-3364.2005.04.004WANG Chen, XU Ming-hou, LIU Jing, ZHENG Chu-guang. Study on reaction mechanisms of trace elements during process of coal combustion based on quantum and advancement thereof[J]. Therm Power Gener,2005,(4):15−19+40. doi: 10.3969/j.issn.1002-3364.2005.04.004 [8] 吕雪峰, 于溯源. 铬/氢/空气焚烧动力学模型机理分析及反应速率常数计算[J]. 热科学与技术,2011,10(4):356−359. doi: 10.3969/j.issn.1671-8097.2011.04.015LV Xue-feng, YU Su-yuan. Reaction mechanisms and rate constants in Cr/H/Air incineration kinetic model[J]. J Therm Sci Technol,2011,10(4):356−359. doi: 10.3969/j.issn.1671-8097.2011.04.015 [9] 刘晶. 燃烧过程中痕量元素释放与反应机理的研究[D]. 武汉: 华中科技大学, 2007.LIU Jing. Study on trace elements release and reaction mechanism during coal combustion[D]. Wuhan: Huazhong University of Science and Technology, 2007. [10] SLIGER R N, KRAMLICH J C, MARINOV N M. Towards the development of a chemical kinetic model for the homogeneous oxidation of mercury by chlorine species[J]. Fuel Process Technol,2000,65:423−438. [11] 陈嵩涛. 燃煤烟气中铅的反应机理研究[D]. 北京: 华北电力大学, 2015.CHEN Song-tao.Reaction mechanism study on trace elements of Pb in the coal flue gas[D]. Beijing: North China Electric Power University, 2015. [12] NIKSA S, HELBLE JJ, FUJIWARA N. Kinetic modeling of homogeneous mercury oxidation: The importance of NO and H2O in predicting oxidation in coal-derived systems[J]. Environ Sci Technol,2001,35(18):3701−3706. doi: 10.1021/es010728v [13] 周素芹, 谷亚昕, 固旭. 煤炭燃烧过程中N2O消除反应机理的密度泛函理论研究(英文)[J]. 无机化学学报,2011,27(6):1202−1206.ZHOU Su-qin, GU Ya-xin, GU Xu. Mechanism of N2O Destruction under coal combustion conditions: Density functional theory study[J]. Chin J Inorg Chem,2011,27(6):1202−1206. [14] 邹潺, 王春波, 邢佳颖. 煤燃烧过程中砷与氮氧化物的反应机理[J]. 燃料化学学报,2019,47(2):138−143. doi: 10.3969/j.issn.0253-2409.2019.02.002ZOU Chan, WANG Chun-bo, XING Jia-ying. Reaction mechanism of arsenic and nitrous oxides during coal combustion[J]. J Fuel Chem Technol,2019,47(2):138−143. doi: 10.3969/j.issn.0253-2409.2019.02.002 [15] URBAN DR, WILCOX J. Theoretical study of the kinetics of the reactions Se + O2 → Se + O and As + HCl → AsCl + H[J]. J Phys Chem A,2006,110(28):8797−8801. doi: 10.1021/jp0628986 [16] PENDERGAST M, PRZYBYLEX M, LINDBLAD M, WILCOX J. Theoretical predictions of arsenic and selenium species under atmospheric conditions[J]. Atmospheric Environ,2008,42(10):2349−2357. doi: 10.1016/j.atmosenv.2007.12.028 [17] 闫傲, 张月, 王春波, 白涛, 赵斌. O2对燃煤烟气中As2O3均相反应生成途径影响研究[J]. 燃料化学学报,2020,48(1):11−17. doi: 10.3969/j.issn.0253-2409.2020.01.002YAN Ao, ZHANG Yue, WANG Chun-bo, BAI Tao, ZHAO Bin. Influence of O2 on the formation of As2O3 by homogeneous reaction with As and AsO in the coal-fired flue gas[J]. J Fuel Chem Technol,2020,48(1):11−17. doi: 10.3969/j.issn.0253-2409.2020.01.002 [18] SÉBASTIEN C, FRÉDÉRIC B, ERIC H. KiSThelP: A program to predict thermodynamic properties and rate constants from quantum chemistry results[J]. J Comput Chem,2014,35(1):82−93. doi: 10.1002/jcc.23470 [19] 卢天. 使用KiSThelP结合Gaussian基于过渡态理论计算反应速率常数[EB/OL]. http://sobereva.com/246, 2018-2-16/2020-10-7.LU Tian. Using KiSThelP combined with Gaussian to calculate the reaction rate constant based on transition state theory[EB/OL]. http://sobereva.com/246, 2018-2-16/2020-10-7. [20] ZHENG J, ALECU I M, LYNCH B J, ZHAO Y, TRUHLAR D G. Database of frequency scale factors for electronic model chemistries[EB/OL]. https://comp.chem.umn.edu/freqscale/version3b1.htm. -




 下载:
下载: