Preparation of high thermal conductivity SiO2@Al support and its application in Fischer-Tropsch synthesis
-
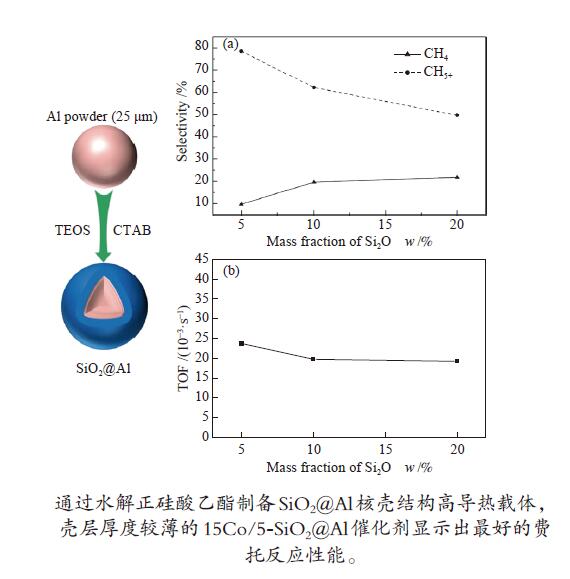
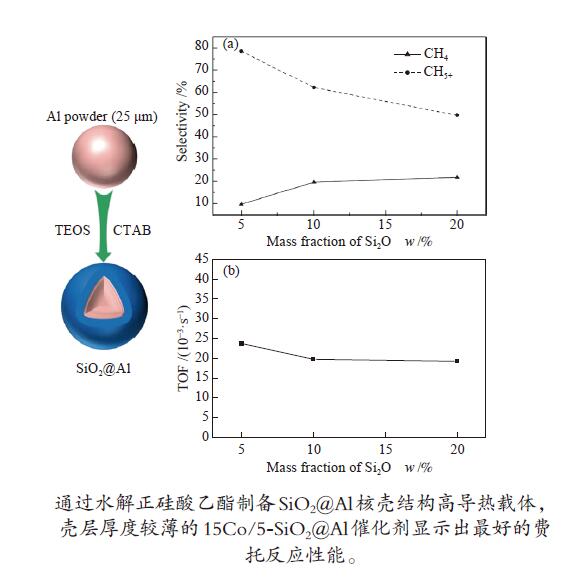
摘要: 通过在弱碱性条件下水解正硅酸乙酯(TEOS),以十六烷基三甲基溴化铵(CTAB)为模板剂成功制备了SiO2@Al核壳结构高导热载体,并采用过量浸渍法负载15%(质量分数)钴物种,进行了费托合成反应性能考察。结果表明,制备得到的核壳结构载体既保留了高金属铝含量,又具有高比表面积壳层,孔径较为均一,为2.6−2.8 nm。通过改变正硅酸乙酯的添加量可以调节载体中二氧化硅的含量,进而控制壳层厚度。随着二氧化硅壳层厚度的增加,载体的比表面积逐渐增大,催化剂还原度降低。二氧化硅壳层增强了金属钴的分散,避免金属钴在铝颗粒上的团聚失活。在相近转化率条件下,壳层厚度较薄的15Co/5-SiO2@Al催化剂显示出最好的费托反应性能,这主要归因于较薄的二氧化硅壳层和金属钴颗粒之间具有适中的金属-载体相互作用,在起到锚定分散钴物种作用的同时提高了钴物种的还原度。Abstract: High thermal conductivity supports, core-shell structure SiO2@Al, was successfully prepared by hydrolyzing tetraethyl orthosilicate (TEOS) under weak alkaline condition and using hexadecyl trimethyl ammonium bromide (CTAB) as the template. 15% (mass fraction) of cobalt was loaded on these supports by excessive impregnation method for Fischer-Tropsch synthesis reaction. The prepared catalysts have high metal aluminum content, high specific surface shell layer and relatively uniform pore size of 2.6−2.8 nm. By changing the added amount of TEOS, the content of silica in the supports and the thickness of the shell layer can be adjusted. As the thickness of the silica shell layer increases, the specific surface area of the supports gradually increases, the interaction between cobalt and the supports increases, and at the same time, the reduction degree of the catalysts decreases. The silica shell enhances the dispersion of metallic cobalt and avoids deactivation caused by agglomeration of metallic cobalt on aluminum particles. Under the condition of similar conversion rate, the catalyst 15Co/5-SiO2@Al with a thinner shell layer has the best performance in Fischer-Tropsch synthesis which is mainly due to the moderate metal-support interaction between the thin silica shell layer and metal cobalt particles. The thinner silica shell layer can anchor and disperse cobalt species to increase the reduction degree of cobalt species.
-
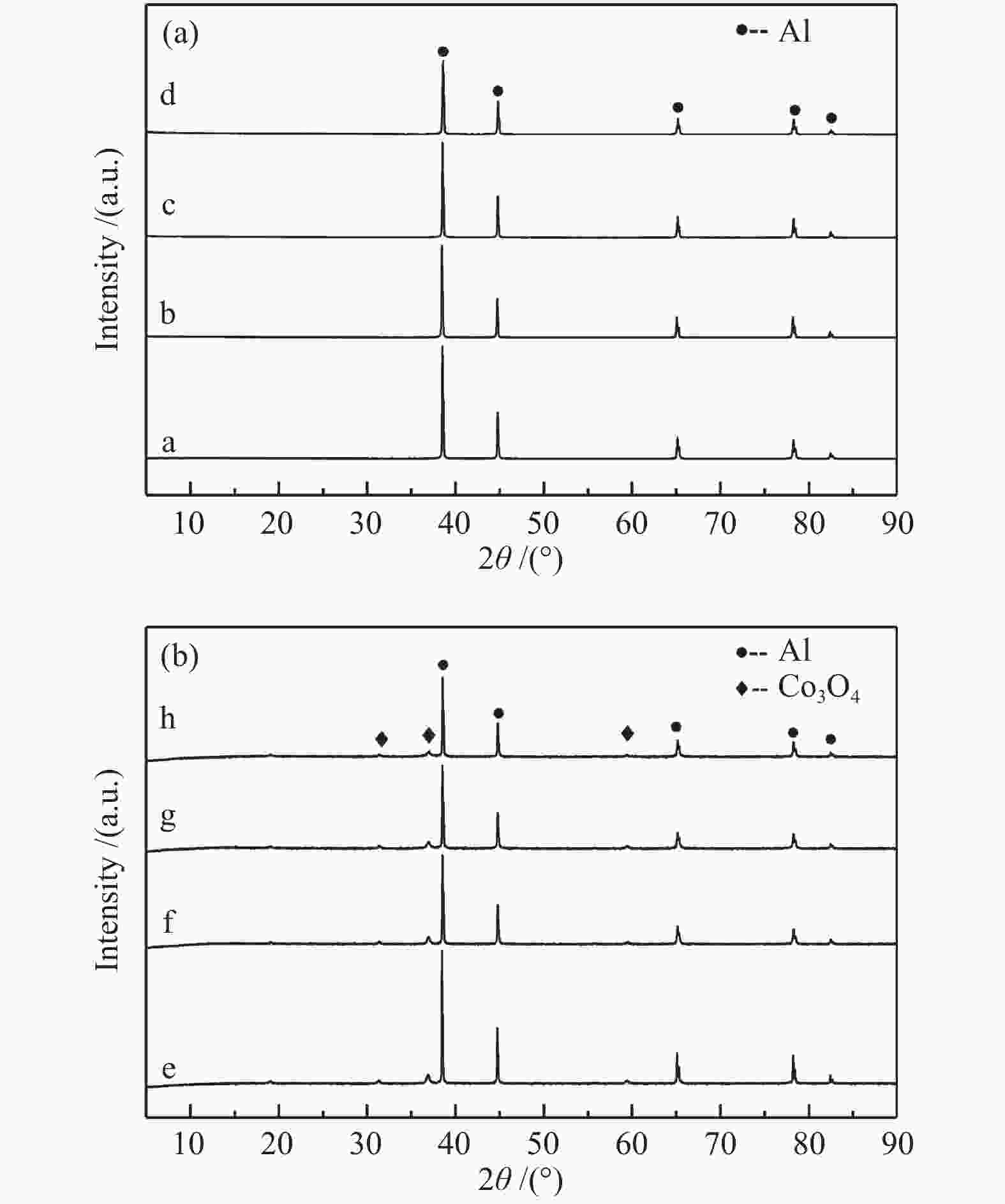
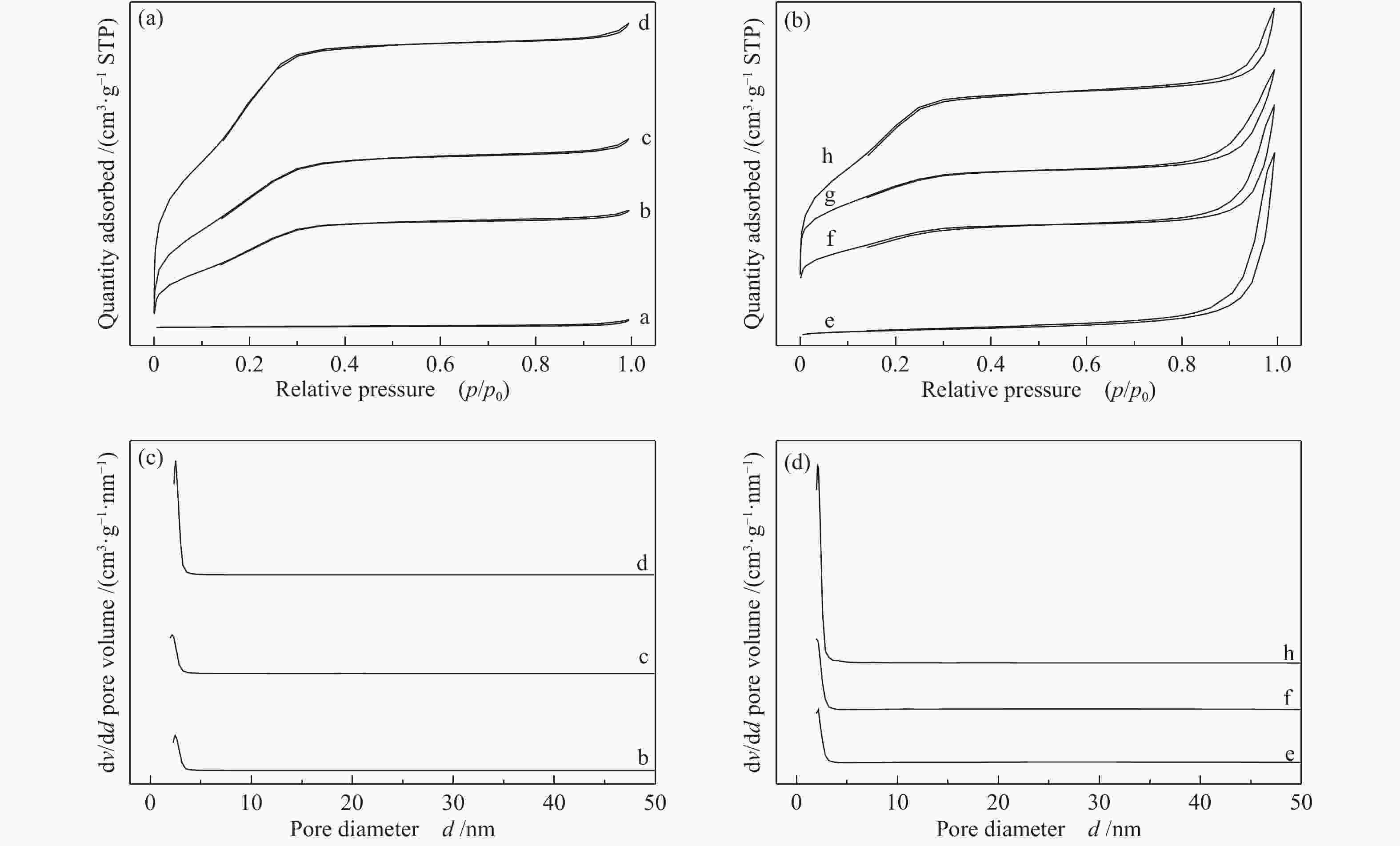
图 3 载体和催化剂的SEM照片
(a): 0-SiO2@Al; (b): 5-SiO2@Al; (c): 10-SiO2@Al; (d): 20-SiO2@Al; (e): 15Co/0-SiO2@Al; (f): 15Co/5-SiO2@Al; (g): 15Co/10-SiO2@Al; (h): 15Co/20-SiO2@Al; (i): used-15Co/0-SiO2@Al; (j): used-15Co/5-SiO2@Al; (k): used-15Co/10-SiO2@Al; (l): used-15Co/20-SiO2@Al
Figure 3 SEM images of the supports and catalysts
表 1 载体和催化剂的物化性质
Table 1 Physico-chemical properties of the supports and the catalysts
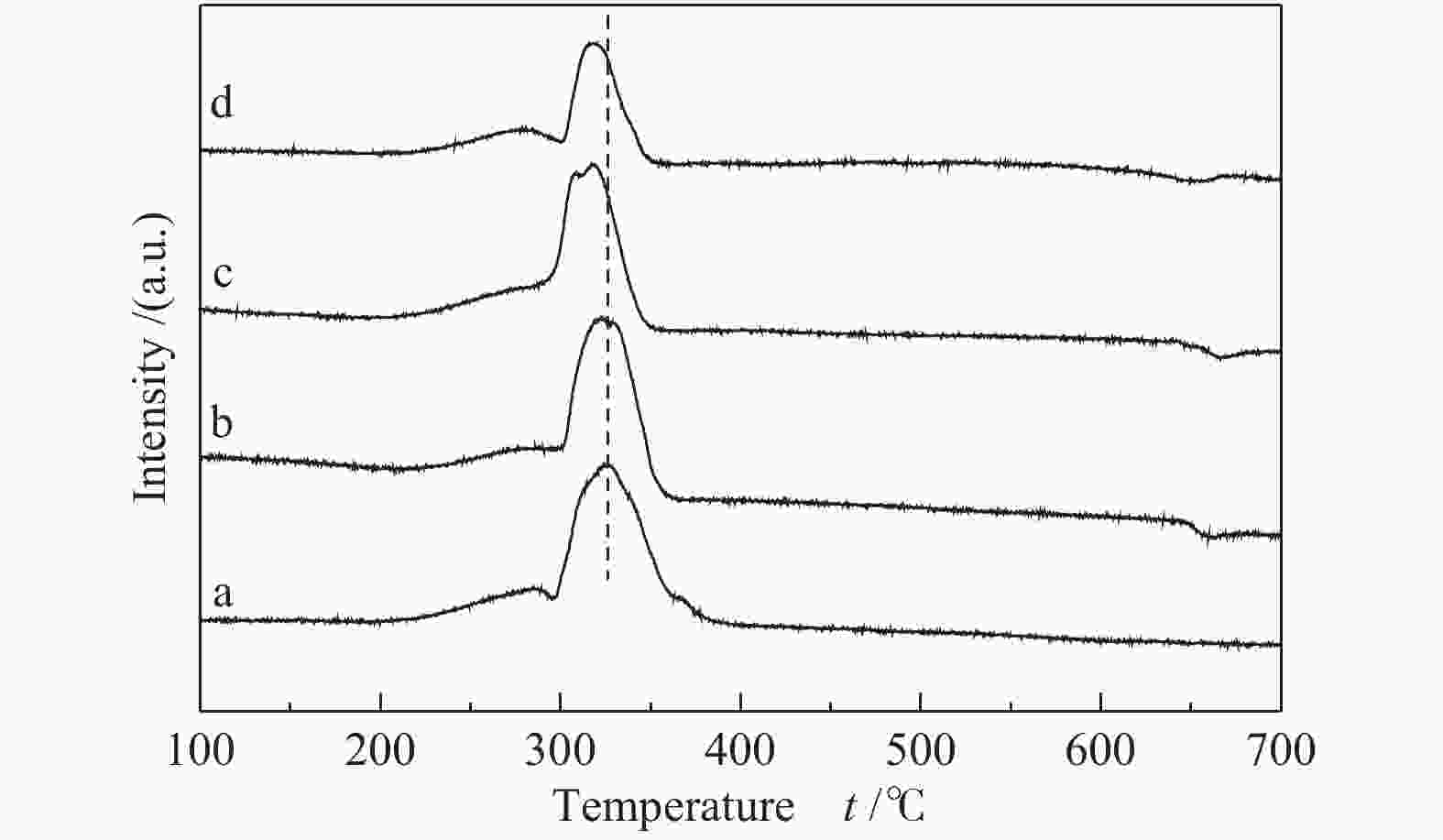
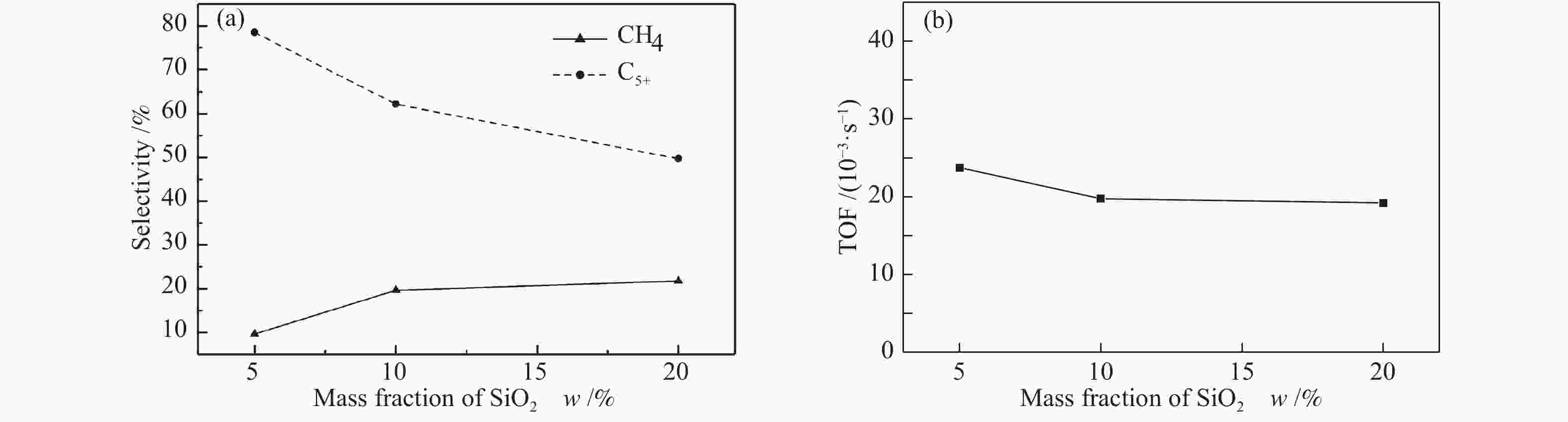
Samples ABET/(m2·g−1) vp/(cm3·g−1) dp/nm Ala/% d(Co)b/nm Reducibilityc/% Dispersiond/% ds(Co)e/nm 0-SiO2@Al 0.18 − − 98.6 − − − − 5-SiO2@Al 74.27 0.0406 2.8 93.7 − − − − 10-SiO2@Al 115.87 0.0640 2.6 90.4 − − − − 20-SiO2@Al 197.68 0.1014 2.8 82.8 − − − − 15Co/0-SiO2@Al 6.70 0.0549 34.3 − 17.15 68.72 4.1 23.49 15Co/5-SiO2@Al 45.13 0.0565 5.0 − 16.70 67.96 4.3 22.50 15Co/10-SiO2@Al 65.16 0.0610 3.7 − 16.52 57.50 5.0 19.34 15Co/20-SiO2@Al 130.09 0.0847 3.3 − 14.93 38.52 5.3 18.08 ABET: BET surface area; vp: BJH pore volume; dp: average pore diameter; a: calculated by ICP-OES; b: calculated by Scherrer equation according to the 2θ = 36.9° diffraction peak of Co3O4, d(Co) = 0.75d(Co3O4); c: calculated by H2-TPR; d : calculated by dispersion = 96/d s(Co); e: calculated from H2 chemisorption 表 2 催化剂的费托反应性能
Table 2 Performance of different catalysts on Fischer-Tropsch synthesis
Catalyst t/ ℃ xCO /% Selectivity s/% TOF/
(10−3·s−1)Criterion a CH4 C2−4 C5+ 15Co/0-SiO2@Al 185 6.18 16.20 12.30 71.51 − − 15Co/5-SiO2@Al 205 27.96 9.66 11.86 78.48 23.74 1.03 × 10−4 15Co/10-SiO2@Al 215 27.18 19.63 18.21 62.16 19.75 1.00 × 10−4 15Co/20-SiO2@Al 205 30.61 21.71 28.57 49.71 19.21 1.05 × 10−4 a: Mears criterion, calculated by the formula (1);
reaction conditions: H2/CO (molar ratio) = 2.0,GHSV = 1000 h−1,p = 2.0 MPa,time on stream = 48 h -
[1] MUNNIK P, DE JONGH P E, DE JONG K P. Recent developments in the synthesis of supported catalysts[J]. Chem Rev,2015,115(14):6687−6718. doi: 10.1021/cr500486u [2] DRY M. The Fischer-Tropsch process: 1950—2000[J]. Catal Today, 71(3/4): 227-241. [3] ZHANG L, CHU H L, QU H, ZHANG Q, XU H, CAO J, TANG Z Y, XUAN J. An investigation of efficient microstructured reactor with monolith Co/anodic γ-Al2O3/Al catalyst in Fischer-Tropsch synthesis[J]. Int J Hydrogen Energy,2018,43(6):3077−3086. doi: 10.1016/j.ijhydene.2017.12.152 [4] ZHANG Q, KANG J, WANG Y. Development of novel catalysts for fischer-tropsch synthesis: Tuning the product selectivity[J]. ChemCatChem,2010,2(9):1030−1058. doi: 10.1002/cctc.201000071 [5] DE TYMOWSKI B, LIU Y, MENY C, LEFÈVRE C, BEGIN D, NGUYEN P, PHAM C, EDOUARD D, LUCK F, PHAM-HUU C. Co–Ru/SiC impregnated with ethanol as an effective catalyst for the Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,2012,419−420:31−40. doi: 10.1016/j.apcata.2012.01.004 [6] ZHU X W, LU X J, LIU X Y, HILDEBRANDT D, GLASSER D. Study of radial heat transfer in a tubular Fischer-Tropsch synthesis reactor[J]. Ind Eng Chem Res,2010,49(21):10682−10688. doi: 10.1021/ie1004527 [7] 吴春来. 南非SASOL的煤炭间接液化技术[J]. 煤化工,2003,31(2):3−6. doi: 10.3969/j.issn.1005-9598.2003.02.001WU Chun-lai. Sasol synfuels-indirect coal liquefaction technology in South Africa[J]. Coal Chem Ind,2003,31(2):3−6. doi: 10.3969/j.issn.1005-9598.2003.02.001 [8] PANGARKAR K, SCHILDHAUER T J, VAN OMMEN J R, NIJENHUIS J, MOULIJN J A, KAPTEIJN F. Experimental and numerical comparison of structured packings with a randomly packed bed reactor for Fischer-Tropsch synthesis[J]. Catal Today,2009,147:S2−S9. doi: 10.1016/j.cattod.2009.07.035 [9] DAVIS B H. Overview of reactors for liquid phase Fischer-Tropsch synthesis[J]. Catal Today,2002,71(3-4):249−300. doi: 10.1016/S0920-5861(01)00455-2 [10] HUANG X W, ROBERTS C B. Selective Fischer-Tropsch synthesis over an Al2O3 supported cobalt catalyst in supercritical hexane[J]. Fuel Process Technol,2003,83(1-3):81−99. doi: 10.1016/S0378-3820(03)00060-2 [11] YANG J I, YANG J H, KIM H J, JUNG H, CHUN D H, LEE H T. Highly effective cobalt catalyst for wax production in Fischer-Tropsch synthesis[J]. Fuel,2010,89(1):237−243. doi: 10.1016/j.fuel.2009.07.008 [12] XIONG H F, MOTCHELAHO M A M, MOYO M, JEWELL L L, COVILLE N J. Correlating the preparation and performance of cobalt catalysts supported on carbon nanotubes and carbon spheres in the Fischer-Tropsch synthesis[J]. J Catal,2011,278(1):26−40. doi: 10.1016/j.jcat.2010.11.010 [13] MAXIME LACROIX L D, BENOIT DE TYMOWSKI, FABRICE VIGNERON, DAVID EDOUARD, DOMINIQUE BÉGIN, PATRICK NGUYEN, CHARLOTTE PHAM, SABINE SAVIN-PONCET, FRANCIS LUCK, MARC-JACQUES LEDOUX, CUONG PHAM-HUUA. Silicon carbide foam composite containing cobalt as a highly selective and re-usable Fischer-Tropsch synthesis catalyst[J]. Appl Catal A: Gen,2011,397(1/2):62−72. doi: 10.1016/j.apcata.2011.02.012 [14] CHUNG D D L. Materials for thermal conduction[J]. Appl Therm Eng,2001,21(16):1593−1605. doi: 10.1016/S1359-4311(01)00042-4 [15] WANG D, CHEN C B, WANG J G, JIA L T, HOU B, LI D B. High thermal conductive core-shell structured Al2O3@Al composite supported cobalt catalyst for Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,2016,527:60−71. doi: 10.1016/j.apcata.2016.08.027 [16] ZHONG M, WANG J G, CHEN C B, MA Z C, JIA L T, HOU B, LI D B. Incorporating silicon carbide nanoparticles into Al2O3@Al to achieve an efficient support for Co-based catalysts to boost their catalytic performance towards Fischer-Tropsch synthesis[J]. Catal Sci Technol,2019,9(21):6037−6046. doi: 10.1039/C9CY01422E [17] CHENG Z P, YANG Y, LI F S, PAN Z H. Synthesis and characterization of aluminum particles coated with uniform silica shell[J]. Trans Nonferrous Met Soc China,2008,18(2):378−382. doi: 10.1016/S1003-6326(08)60066-7 [18] JUNG J S, KIM S W, MOON D J. Fischer-Tropsch synthesis over cobalt based catalyst supported on different mesoporous silica[J]. Catal Today,2012,185(1):168−174. doi: 10.1016/j.cattod.2012.02.002 [19] BORG O, DIETZEL P, SPJELKAVIK A, TVETEN E, WALMSLEY J, DIPLAS S, ERI S, HOLMEN A, RYTTER E. Fischer-Tropsch synthesis: Cobalt particle size and support effects on intrinsic activity and product distribution[J]. J Catal,2008,259(2):161−164. doi: 10.1016/j.jcat.2008.08.017 [20] ZHANG H J, WU J, ZHOU L P, ZHANG D Y, QI L M. Facile synthesis of monodisperse microspheres and gigantic hollow shells of mesoporous silica in mixed Water-Ethanol solvents[J]. Langmuir,2007,23(3):1107−1113. doi: 10.1021/la062542l [21] PANPRANOT J, KAEWGUN S, PRASERTHDAM P. Metal-Support interaction in mesoporous silica supported cobalt Fischer-Tropsch catalysts[J]. React Kinet Catal Lett,2005,85(2):299−304. doi: 10.1007/s11144-005-0274-6 [22] CHU W, CHERNAVSKII P, GENGEMBRE L, PANKINA G, FONGARLAND P, KHODAKOV A. Cobalt species in promoted cobalt alumina-supported Fischer-Tropsch catalysts[J]. J Catal,2007,252(2):215−230. doi: 10.1016/j.jcat.2007.09.018 [23] MENDES F M T, PEREZ C A C, NORONHA F B, SCHMAL M. TPSR of CO hydrogenation on Co/Nb2O5/Al2O3 catalysts[J]. Catal Today,2005,101(1):45−50. doi: 10.1016/j.cattod.2004.12.009 [24] RAHMATI M, HUANG B Y, SCHOFIELD L M, FLETCHER T H, WOODFIELD B F, HECKER W C, BARTHOLOMEW C H, ARGYLE M D. Effects of Ag promotion and preparation method on cobalt Fischer-Tropsch catalysts supported on silica-modified alumina[J]. J Catal,2018,362:118−128. doi: 10.1016/j.jcat.2018.03.027 [25] IGLESIA E, REYES S C, MADON R J, SOLED S L. Selectivity control and catalyst desigAn in the Fischer-Tropsch synthesis: Sites, pellets, and reactors[J]. Adv Catal, 1993, 39: 221−302. [26] MEARS D. Tests for transport limitations in experimental catalytic reactors[J]. Ind Eng Chem Process Des Dev,1972,11(2):320−320. doi: 10.1021/i260042a600 [27] ZHONG M, GUO Y, WANG J, MA Z, XIA M, CHEN C, JIA L, HOU B, LI D. The Fischer-Tropsch synthesis performance over cobalt supported on silicon-based materials: the effect of thermal conductivity of the support[J]. Catal Sci Technol,2019,9(13):3482−3492. doi: 10.1039/C9CY00578A -




 下载:
下载: