Study on pyrolysis and combustion characteristics of different plant fibers
-
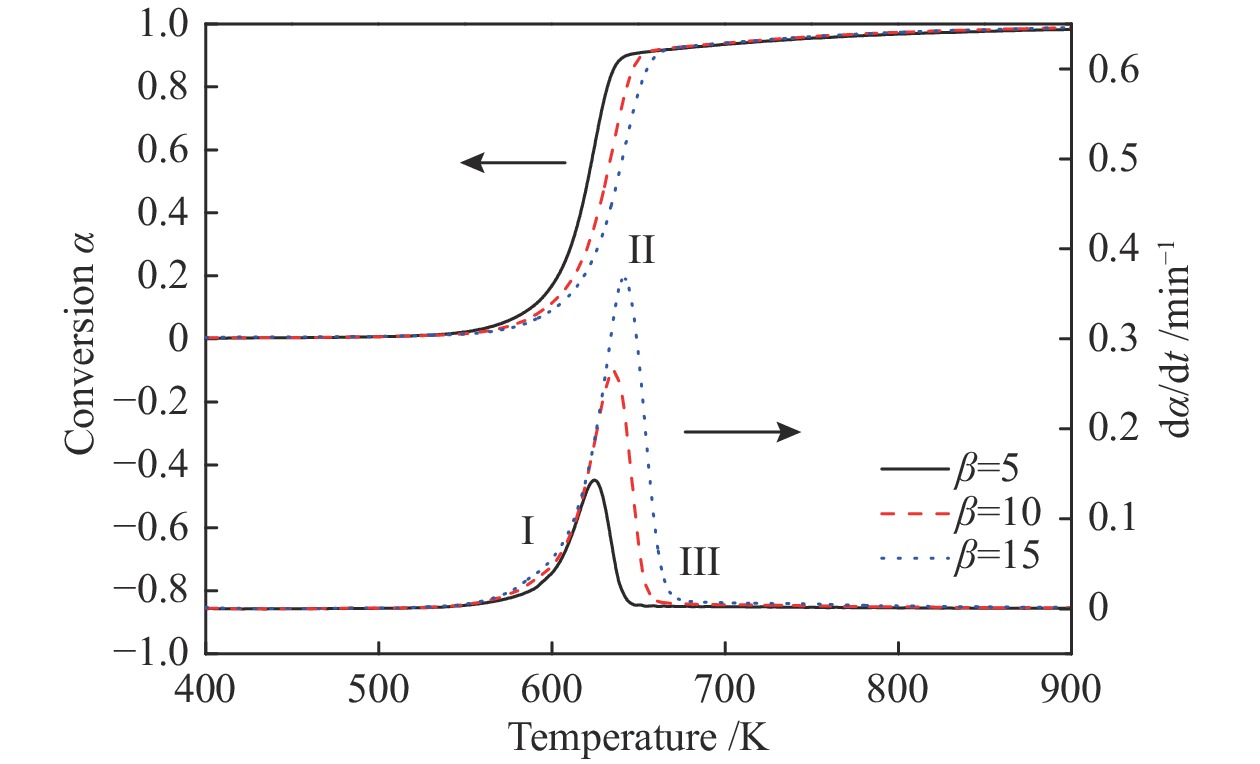
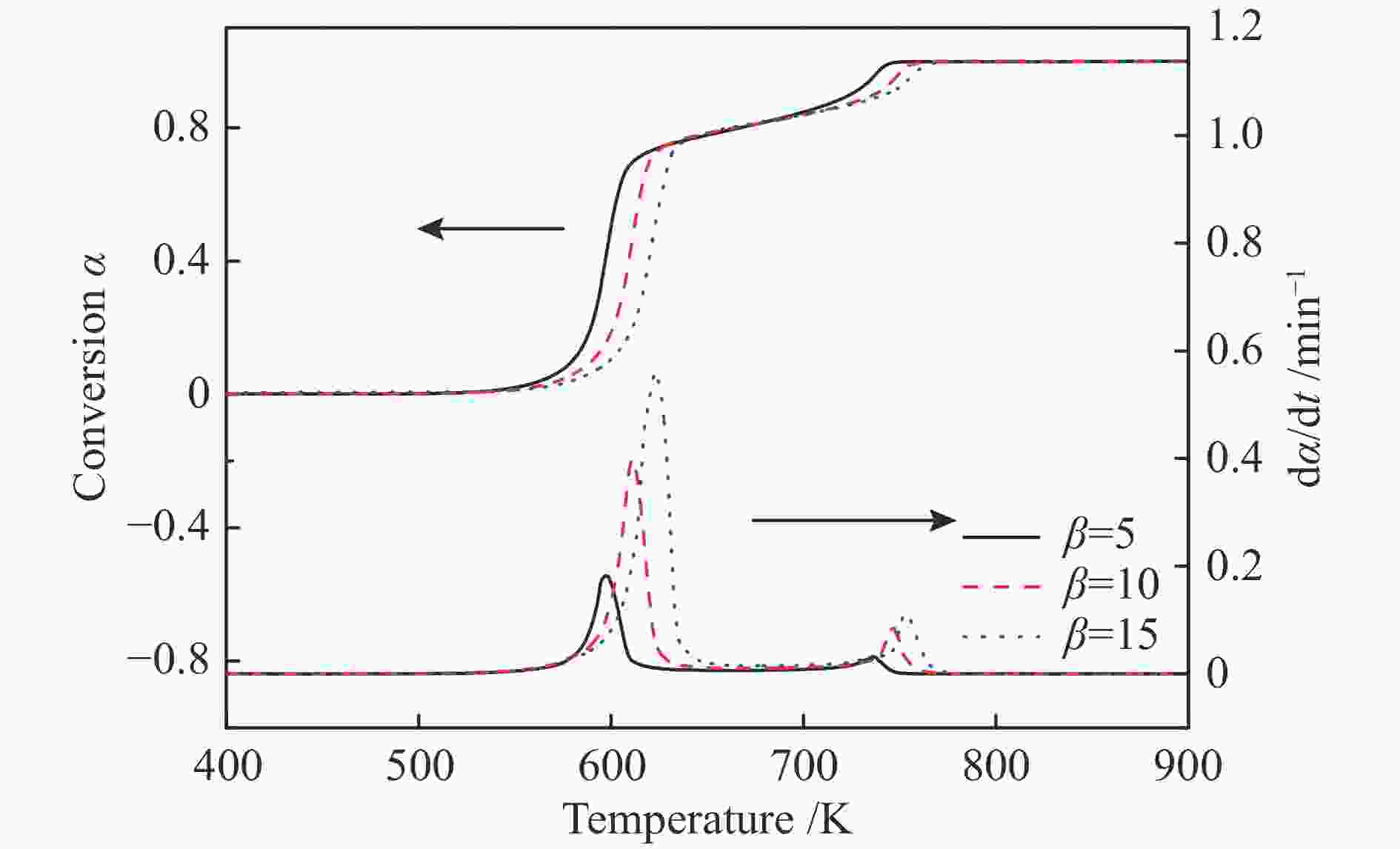
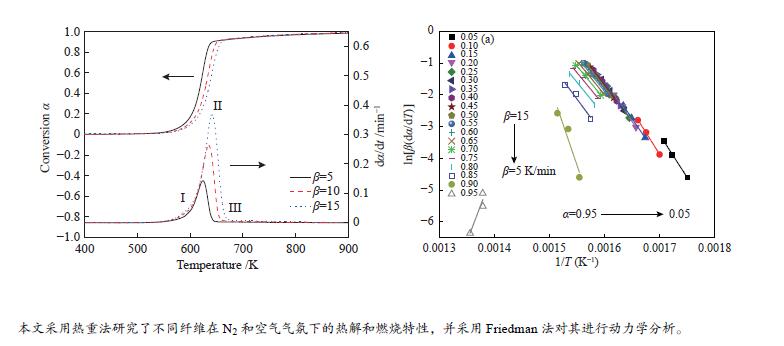
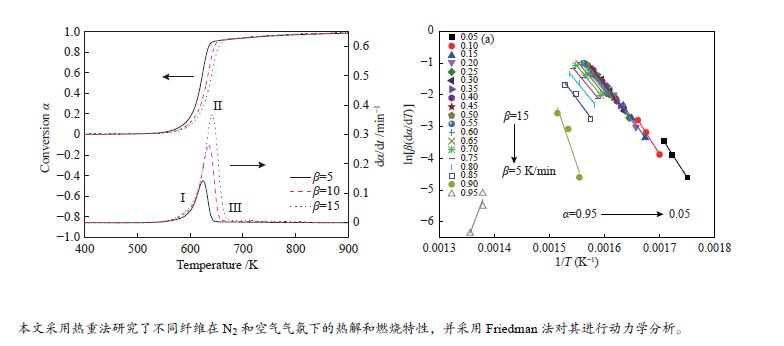
摘要: 本研究采用不等温热重法研究六种纤维(针叶、阔叶、竹、亚麻、草和棉)在N2和空气气氛下的热解和燃烧特性,并采用Friedman法对其进行动力学分析。结果表明,纤维不同的热解和燃烧特性参数与其自身结构组成有关。纤维在热解和燃烧过程中,其挥发分析出温度Ts、终止温度Th、DTG峰温Tmax、固定碳燃烧峰温、最大质量损失速率、热解指数P和燃烧指数S均随着升温速率的增加而增加;在N2气氛下,亚麻纤维Tmax最大,竹纤维Tmax最小,棉纤维的Ts最大,草纤维的最大热解质量损失速率−(dm/dt)max、热解指数P和燃烧指数S均最小;在转化率为0.05−0.85条件下,阔叶纤维平均表观活化能最小(173.3 kJ/mol),竹纤的最大(201.10 kJ/mol)。在空气气氛下,所有纤维的热解过程的Tmax均低于N2条件下,在转化率为0.05−0.65时,纤维在空气中热解的表观活化能Eα低于其在N2条件下的表观活化能。Abstract: In order to study the pyrolysis and combustion characteristics of different fibers, the kinetics of six kinds of plant fibers (coniferous, broadleaf, bamboo, flax, grass and cotton) in N2 and air atmosphere were studied by non-isothermal thermogravimetric (TG) method using Friedman method. The results showed that the fibers had different pyrolysis and combustion characteristic parameters, which were related to their own structural compositions. In the process of pyrolysis and combustion of fibers, the initial volatilization temperature (Ts), terminal decomposition temperature (Th), DTG peak temperature (Tmax), fixed carbon combustion peak temperature, maximum mass loss rate, pyrolysis character index (P) and combustion character index (S) increased with the increase of heating rates; In N2 atmosphere, the flax fiber Tmax and bamboo fiber
Tmax were shown to be the highest and lowest among all fibers, respectively, and Ts of cotton fiber was the largest; Grass fiber had the smallest maximum pyrolysis mass loss rate (−(dm/dt)max), pyrolysis index (P), and combustion index (S); Between the conversion of 0.05−0.85, the average apparent activation energies (E) of broadleaf fiber and bamboo fiber were the smallest (173.30 kJ/mol) and highest (201.10 kJ/mol), respectively. In air atmosphere, Tmax of all fibers in the pyrolysis process was lower than that in N2. The apparent activation energy (Eα) of fiber pyrolysis in air atmosphere was shown to be lower than that in N2 when the conversion was between 5% and 65%. -
Key words:
- plant fiber /
- pyrolysis characteristic /
- combustion characteristic /
- kinetic study /
- Friedman method
-
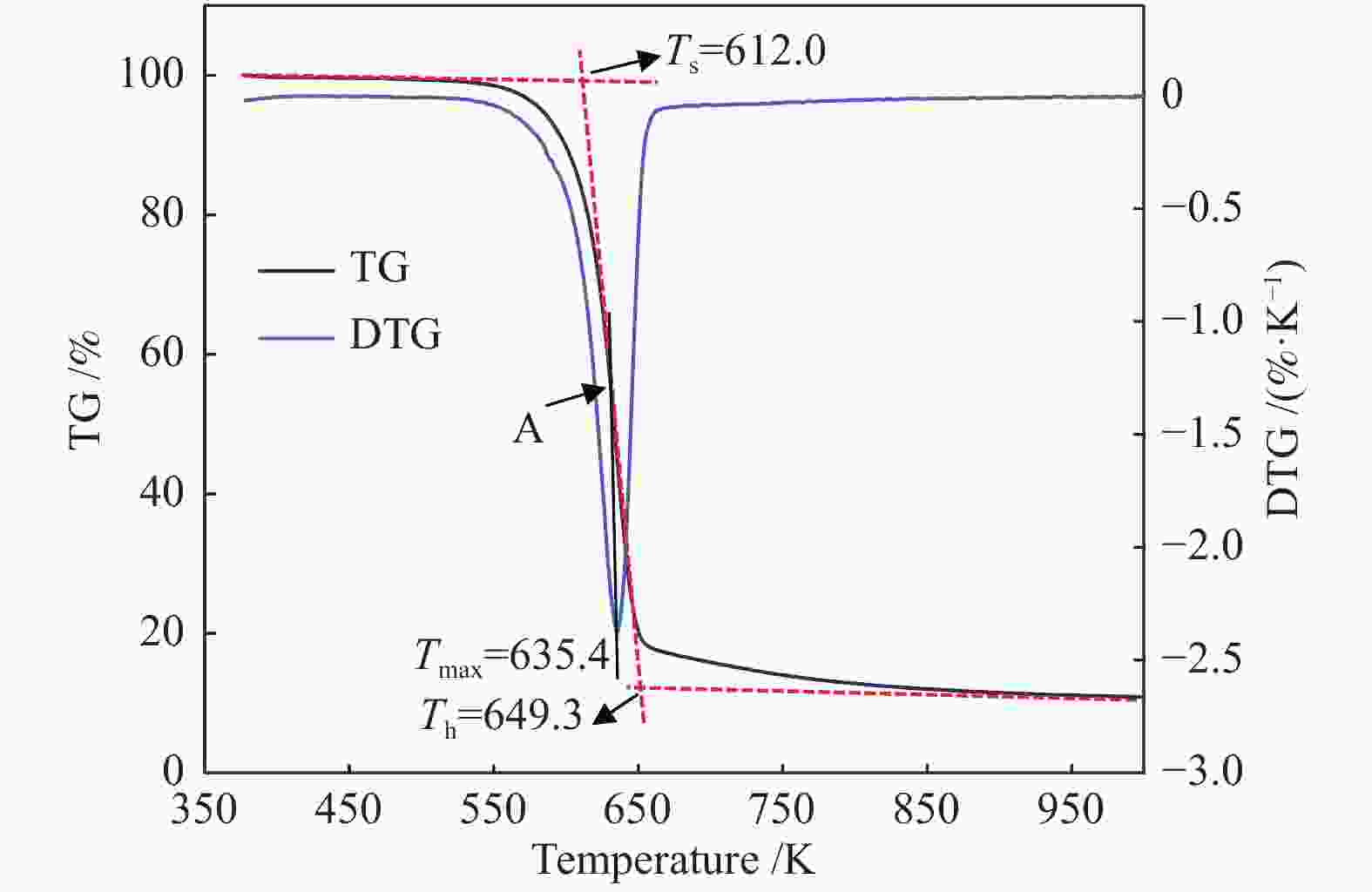
表 1 纤维薄片热解特性参数*
Table 1 Pyrolysis characteristic parameters of fiber sheets
Sample β/(K·min−1) Ts/K Th/K Tmax/K −(dm/dt)max/(%·min−1) △T1/2/K P×10−6/(%·min−1·K−3) Coniferous fiber 5 602.8 637.1 624.6 12.5 25.3 1.31 10 612.0 649.3 635.4 22.9 27.9 2.11 15 617.4 657.3 641.6 32.1 30.5 2.65 Broadleaf fiber 5 597.8 634.0 621.4 11.9 26.2 1.22 10 608.9 648.1 634.1 22.2 29.1 1.98 15 613.5 655.5 640.1 31.0 31.7 2.49 Bamboo fiber 5 595.2 633.3 619.2 11.0 27.7 1.08 10 604.6 646.2 631.2 20.3 30.7 1.74 15 609.3 652.4 636.5 29.1 32.6 2.30 Flax fiber 5 607.6 645.2 630.8 11.5 29.3 1.03 10 618.3 658.3 643.2 21.8 31.7 1.73 15 625.0 666.0 649.1 31.6 27.7 2.81 Grass fiber 5 591.7 637.8 619.8 9.0 33.0 0.74 10 601.2 651.1 631.9 16.9 36.1 1.23 15 607.4 659.8 639.7 24.1 39.0 1.59 Cotton fiber 5 610.0 643.4 629.8 13.2 27.1 1.27 10 620.6 656.4 640.8 24.8 29.9 2.09 15 627.1 664.0 648.1 35.2 31.4 2.76 *β—heating rate;Ts—initial decomposition temperature;Th—terminal decomposition temperature;Tmax—peak temperature;−(dm/dt)max—maximum mass loss rate;△T1/2—peak width at half-height; P—pyrolysis index 表 2 纤维的组成信息
Table 2 Information of fiber composition (%)
Sample Cellulose Hemicellulose Lignin Coniferous fiber 74.56 ± 0.23 13.23 ± 0.43 9.32 ± 1.61 Broadleaf fiber 75.8 ± 0.31 15.07 ± 0.55 5.55 ± 1.21 Bamboo fiber 74.05 ± 0.52 18.28 ± 0.46 5.86 ± 0.71 Flax fiber 92.19 ± 0.34 4.32 ± 0.16 2.33 ± 0.51 Grass fiber 71.9 ± 0.17 17.59 ± 0.55 4.28 ± 0.50 Cotton fiber 95.84 ± 0.97 0.23 ± 0.01 1.17 ± 0.01 表 3 Friedman法得到纤维在N2气氛下热解的表观活化能Eα(kJ/mol)和相关系数R2
Table 3 Apparent activation energy Eα(kJ/mol) of different fibers pyrolysis under N2 atmosphere obtained by Friedman method and correlation coefficient R2
Conversion Coniferous Broadleaf Bamboo Flax Grass Cotton Eα R2 Eα R2 Eα R2 Eα R2 Eα R2 Eα R2 0.05 224.16 0.9962 185.76 0.9769 214.39 0.9837 182.96 0.9952 183.16 0.9989 206.94 0.9994 0.1 228.85 0.9992 196.07 0.9927 227.60 0.9968 193.57 0.9997 209.72 0.9971 186.28 1.0000 0.15 211.04 1.0000 188.48 0.9910 216.08 0.9949 184.16 1.0000 203.67 0.9985 189.93 0.9998 0.2 210.38 0.9999 188.72 0.9954 218.02 0.9962 188.78 1.0000 184.92 0.9998 188.52 0.9996 0.25 203.58 0.9998 185.37 0.9963 213.00 0.9967 187.00 0.9999 186.89 0.9992 186.82 0.9995 0.3 197.55 0.9995 181.42 0.9972 205.12 0.9976 186.65 1.0000 182.30 0.9993 185.16 0.9998 0.35 193.79 0.9994 178.06 0.9980 201.10 0.9963 184.48 0.9999 176.78 0.9992 189.52 1.0000 0.4 190.80 0.9998 173.30 0.9980 198.15 0.9974 184.77 1.0000 173.92 0.9986 191.19 0.9990 0.45 188.00 0.9994 171.94 0.9983 193.86 0.9962 186.93 0.9999 170.33 0.9987 187.55 0.9994 0.5 186.02 0.9995 169.93 0.9986 193.62 0.9974 187.58 0.9996 168.08 0.9989 181.27 1.0000 0.55 184.37 0.9996 168.42 0.9977 192.50 0.9968 185.69 1.0000 168.19 0.9988 177.59 0.9999 0.6 179.99 0.9987 167.40 0.9982 191.93 0.9979 182.11 1.0000 167.02 0.9986 176.19 1.0000 0.65 177.07 0.9978 165.31 0.9989 191.79 0.9975 179.71 0.9996 167.47 0.9988 176.97 0.9999 0.7 174.73 0.9953 161.26 0.9995 192.96 0.9972 178.66 0.9991 170.44 0.9986 177.59 1.0000 0.75 175.14 0.9898 160.78 1.0000 196.99 0.9981 177.87 0.9987 173.45 0.9983 178.37 1.0000 0.8 177.50 0.9845 162.33 1.0000 205.95 0.9992 180.06 0.9975 179.63 0.9982 184.03 0.9999 0.85 194.01 0.9744 178.59 0.9994 245.74 1.0000 188.62 0.9935 194.08 0.9976 199.28 0.9998 0.9 422.16 0.9265 334.18 1.0000 1227.04 0.8234 261.73 0.9700 287.75 0.9931 267.37 0.9945 0.95 −378.69 0.8906 −396.67 0.5410 −400.47 0.9187 −227.41 0.6301 259.91 0.7083 −232.31 0.8507 E
(α: 0.05−0.85)193.79 173.30 201.10 184.77 176.78 186.28 表 4 纤维在空气气氛、不同升温速率条件下的燃烧特性参数*
Table 4 Combustion characteristic parameters of different fiber sheets under airatmosphere at different heating rates
Sample β/(K·min−1) Pyrolysis stage Combustion stage −(dm/dt)mean /(%·min−1) S×10−8/(%2·
min−2·K−3)Ts,1/K Th,1 /K Tmax,1/K −(dm/dt)max1 /(%·min−1) Ts,2/K Th,2/K Tmax,2/K −(dm/dt)max2 /(%·min−1) Coniferous fiber 5 585.7 606.2 597.7 17.47 724.1 743.0 736.3 3.05 1.09 7.49 10 598.9 619.4 611.1 38.05 737.8 752.8 746.6 8.1 2.08 29.31 15 608.9 631.4 623.0 53.61 745.5 761.9 752.9 10.40 3.02 57.40 Broadleaf fiber 5 582.4 604.0 595.9 17.68 716.1 725.5 723.5 6.21 1.12 8.02 10 596.2 618.7 609.6 34.65 719.7 734.4 729.2 8.64 2.16 28.65 15 605.1 627.9 617.5 52.19 726.8 744.8 734.9 11.13 3.13 59.91 Bamboo fiber 5 580.4 605.3 595.1 14.13 712.0 721.0 718.4 6.99 1.11 6.49 10 595.7 620.0 611.0 30.85 719.4 739.4 727.7 7.44 2.18 25.59 15 602.5 628.7 618.6 44.25 726.2 746.8 734.1 10.74 3.13 51.08 Flax fiber 5 587.4 609.7 600.6 16.44 707.0 753.6 737.0 1.60 1.05 6.67 10 601.8 625.0 615.2 32.73 724.1 770.5 752.7 2.97 2.03 23.82 15 609.6 634.7 626.0 47.97 731.3 780.3 758.9 3.99 2.97 49.14 Grass fiber 5 574.4 605.8 592.7 11.28 680.8 717.3 696.9 2.15 1.10 5.26 10 588.5 619.9 607.8 23.36 697.2 715.4 704.2 7.19 2.16 20.35 15 596.3 628.1 615.8 35.98 696.7 721.9 705.9 9.28 3.15 44.12 Cotton fiber 5 591.9 610.8 602.9 20.18 726.7 766.7 753.8 1.62 1.04 7.82 10 606.7 626.9 618.7 39.25 743.9 783.5 771.3 3.06 2.02 27.48 15 615.3 636.4 628.1 58.5 754.6 793.9 783.2 4.24 2.98 58.10 *β—heating rate;Ts—initial decomposition temperature;Th—terminal decomposition temperature;Tmax—peak temperature;−(dm/dt)max—maximum mass loss rate;−(dm/dt)mean—average mass loss rate; S—combustion index 表 5 Friedman法得到纤维在空气气氛下燃烧的表观活化能Eα(kJ/mol)和相关系数R2
Table 5 Apparent activation energy Eα(kJ/mol) of different fibers pyrolysis under air atmosphere obtained by Friedman method and the relative correlation coefficient R2
α Coniferous fiber Broadleaf fiber Bamboo fiber Flax fiber Grass fiber Cotton fiber Eα R2 Eα R2 Eα R2 Eα R2 Eα R2 Eα R2 0.05 165.8 0.9704 169.82 0.9997 162.9 0.9968 140.65 0.9973 175.09 0.9861 124.09 0.9967 0.1 131.33 0.9902 137.01 1.0000 141.92 0.9934 127.15 0.9973 157.47 0.9998 116.35 0.9996 0.15 126.12 0.9892 132.08 0.9995 127.99 0.9999 127.8 0.9998 139.1 0.9985 118.75 1.0000 0.2 137.41 0.9988 129.79 0.9998 130.31 0.9970 128.12 1.0000 139.09 1.0000 124.62 0.9996 0.25 128.74 0.9941 126 0.9991 130.19 0.9962 139.41 0.9980 143.09 0.9982 136.99 0.9987 0.3 130.92 0.9999 125.18 0.9760 144.95 0.9845 143.59 0.9997 141.19 0.9841 132.65 0.9990 0.35 135.64 0.9958 137.72 0.9960 147.87 0.9891 134.99 0.9936 160.18 0.9918 128.32 0.9990 0.4 138.64 0.9659 150.25 0.9996 151.23 0.9989 130.72 0.9899 131.63 0.9987 133.58 0.9999 0.45 146.93 0.9777 148.61 0.9998 165.88 0.9998 144.26 0.9989 152.3 0.9996 143.65 0.9999 0.5 157.98 0.9718 154.04 0.9999 176.13 0.9995 157.97 1.0000 187.55 0.9868 157.26 0.9998 0.55 146.28 0.9775 165.25 0.9822 179.75 0.9997 168.62 0.9993 177.2 0.9999 169.86 0.9993 0.6 165.47 0.9748 183.67 0.9985 175.76 0.9999 175.52 0.9975 184.03 0.9999 186.15 0.9985 0.65 233.98 0.9846 207.04 0.9978 255.59 0.9895 210.39 0.9975 202.51 0.9965 221.75 0.9976 0.7 413.4 0.9493 326.3 0.9966 484.12 0.9163 410.94 0.9949 305.77 0.9972 369.8 0.9988 0.75 769.22 0.5416 519.19 0.9999 253.9 0.0863 1233.16 0.8073 422.58 0.9997 1075.39 0.9484 0.8 −631.82 0.9956 248 0.0484 56.53 0.0199 −184.38 0.2396 388.45 0.9101 −317.51 0.8451 0.85 326.48 0.9175 259.24 0.9843 242.62 0.9477 213.68 0.9199 263.18 0.9930 79.59 0.0708 0.9 188.04 0.9989 244.68 0.8630 205.25 0.9859 192.75 0.9856 398.79 0.9999 176.18 0.9989 0.95 239.06 0.9869 243.56 0.8928 −110.48 0.5280 180.26 0.9991 379.87 0.9482 170.41 0.9971 -
[1] 周顺, 徐迎波, 王程辉, 田振峰, 徐志强, 何庆. 比较研究纤维素、果胶和淀粉的燃烧行为和机理[J]. 中国烟草学报,2011,17(5):1−9. doi: 10.3969/j.issn.1004-5708.2011.05.001ZHOU Shun, XU Ying-bo, WANG Cheng-hui, TIAN Zhen-feng, XU Zhi-qiang, HE Qing. A comparative study of the combustion behavior and mechanism of cellulose, pectin and starch[J]. Acta Tab Sin,2011,17(5):1−9. doi: 10.3969/j.issn.1004-5708.2011.05.001 [2] 吕当振, 姚洪, 王泉斌, 李志远, 彭钦春, 刘小伟, 徐明厚. 纤维素、木质素含量对生物质热解气化特性影响的实验研究[J]. 工程热物理学报,2008,29(10):1771−1774. doi: 10.3321/j.issn:0253-231X.2008.10.042LÜ Dang-zhen, YAO Hong, WANG Quan-bin, LI Zhi-Yuan, PENG Qin-Chun, LIU Xiao-wei, XU Ming-hou. Effect of cellulose and lignin content on pyrolysis and gasification characteristics for several types of biomass[J]. J Eng Therm,2008,29(10):1771−1774. doi: 10.3321/j.issn:0253-231X.2008.10.042 [3] 赵坤, 肖军, 沈来宏, 瞿婷婷. 基于三组分的生物质快速热解实验研究[J]. 太阳能学报,2011,32(5):7710−717.ZHAO Shen, XIAO Jun, SHEN Lai-hong, QU Ting-ting. Experimental study of biomass rapid pyrolysis based on three components[J]. Acta Energ Sol Sin,2011,32(5):7710−717. [4] VYAZOVKIN S. Modification of the integral isoconversional method to account for variation in activation energy[J]. J Comput Chem,2001,22(2):178−183. doi: 10.1002/1096-987X(20010130)22:2<178::AID-JCC5>3.0.CO;2-# [5] MA Z, WANG J, YANG Y, ZHANG Y, ZHAO C, YU Y, WANG S. Comparison of the thermal degradation behaviors and kinetics of palm oil waste under nitrogen and air atmosphere in TGA-FTIR with a complementary use of model-free and model-fitting approaches[J]. J Anal Appl Pyrolysis,2018,134:12−24. doi: 10.1016/j.jaap.2018.04.002 [6] DAMARTZIS T, VAMVUKA D, SFAKIOTAKIS S, ZABANIOTOU A. Thermal degradation studies and kinetic modeling of cardoon (Cynara cardunculus) pyrolysis using thermogravimetric analysis (TGA)[J]. Bioresour Technol,2011,102(10):6230−6238. doi: 10.1016/j.biortech.2011.02.060 [7] CHEN Y, DUAN J, LUO Y-H. Investigation of agricultural residues pyrolysis behavior under inert and oxidative conditions[J]. J Anal Appl Pyrolysis,2008,83:165−174. doi: 10.1016/j.jaap.2008.07.008 [8] MIRANDA M I G, SAMIOS D, OLIVEIRA PI EIRO T, VAGHETTI J C P, PIATNICKI C M S. Kinetics of oxidation and decomposition of soybean biodiesel evaluated by the TTT superposition theory and the Freeman-Carroll method[J]. J Mol Liq,2017,245:121−128. doi: 10.1016/j.molliq.2017.07.073 [9] DA ROZA M B, NICOLAU A, ANGELONI L M, SIDOU P N, SAMIOS D. Thermodynamic and kinetic evaluation of the polymerization process of epoxidized biodiesel with dicarboxylic anhydride[J]. Mol Phys,2012,110(11/12):1375−1381. doi: 10.1080/00268976.2011.647717 [10] HU M, CHEN Z, WANG S, GUO D, MA C, ZHOU Y, CHEN J, LAGHARI M, FAZAL S, XIAO B, ZHANG B, MA S. Thermogravimetric kinetics of lignocellulosic biomass slow pyrolysis using distributed activation energy model, Fraser-Suzuki deconvolution, and iso-conversional method[J]. Energy Convers Manage,2016,118:1−11. doi: 10.1016/j.enconman.2016.03.058 [11] SBIRRAZZUOLI N. Determination of pre-exponential factors and of the mathematical functions f(α) or G(α) that describe the reaction mechanism in a model-free way[J]. Thermochim Acta,2013,564:59−69. doi: 10.1016/j.tca.2013.04.015 [12] MISHRA G, BHASKAR T. Non isothermal model free kinetics for pyrolysis of rice straw[J]. Bioresource Technol,2014,169:614−621. doi: 10.1016/j.biortech.2014.07.045 [13] 赵增立, 李海滨, 吴创之, 陈勇. 蔗渣的热解与燃烧动力学特性研究[J]. 燃料化学学报,2005,33(3):314−319. doi: 10.3969/j.issn.0253-2409.2005.03.012ZHAO Zeng-li, LI Hai-bin, WU Chuang-zhi, CHEN Yong. Study on the kinetic characteristics of bagasse pyrolysis and combustion[J]. J Fuel Chem Technol,2005,33(3):314−319. doi: 10.3969/j.issn.0253-2409.2005.03.012 [14] DUAN L, CHEN J, JIANG Y, LI X, LONGHURST P, LEI M. Experimental and kinetic study of thermal decomposition behaviour of phytoremediation derived Pteris vittata[J]. J Therm Anal Calorim,2016,128(2):1207−1216. [15] COLLAZZO G C, BROETTO C C, PERONDI D, JUNGES J, DETTMER A, DORNELLES FILHO A A, FOLETTO E L, GODINHO M. A detailed non-isothermal kinetic study of elephant grass pyrolysis from different models[J]. Appl Therm Eng,2017,110:1200−1211. doi: 10.1016/j.applthermaleng.2016.09.012 [16] GUO G, LIU C, WANG Y, XIE S, ZHANG K, CHEN L, ZHU W, DING M. Comparative investigation on thermal degradation of flue-cured tobacco with different particle sizes by a macro-thermogravimetric analyzer and their apparent kinetics based on distributed activation energy model[J]. J Therm Anal Calorim,2019,138(5):3375−3388. doi: 10.1007/s10973-019-08215-7 [17] GUO G, ZHANG K, LIU C, XIE S, LI X, LI B, SHU J, NIU Y, ZHU H, DING M, ZHU W. Comparative investigation on thermal decomposition of powdered and pelletized biomasses: Thermal conversion characteristics and apparent kinetics[J]. Bioresour Technol,2020,301:1−9. [18] CHEN D, ZHENG Y, ZHU X. In-depth investigation on the pyrolysis kinetics of raw biomass. Part I: Kinetic analysis for the drying and devolatilization stages[J]. Bioresource Technol,2013,131:40−46. doi: 10.1016/j.biortech.2012.12.136 [19] CAI J, WU W, LIU R, HUBER G W. A distributed activation energy model for the pyrolysis of lignocellulosic biomass[J]. Green Chem,2013,15:1331−1340. doi: 10.1039/c3gc36958g [20] SLUITER A, HAMES B, RUIZ R, SCARLATA C, SLUITER J, TEMPLETON D, CROCKER D, Determination of structural carbohydrates and lignin in biomass[Z]. Golden: National Renewable Energy Laboratory, 2008. [21] LIANG M, YANG T, ZHANG G, ZHANG K, WANG L, LI R, HE Y, WANG J, ZHANG J. Effects of hydrochloric acid washing on the structure and pyrolysis characteristics of tobacco stalk[J]. Biomass Convers Bior,2021,1−14. [22] MA B-G, LI X-G, XU L, WANG K, WANG X-G. Investigation on catalyzed combustion of high ash coal by thermogravimetric analysis[J]. Thermochim Acta,2006,445(1):19−22. doi: 10.1016/j.tca.2006.03.021 [23] 秦国鑫, 李斌, 鲁端峰, 谢国勇, 银董红, 王兵. 烟草生物质燃烧特性与机理研究[J]. 烟草科技,2015,48(1):76−81.QIN Guo-xin, LI Bin, LU Duan-feng, XIE Guo-yong, YIN Dong-hong, WANG Bing. Combustion property and mechanism of tobacco biomass[J]. Tob Sci Technol,2015,48(1):76−81. [24] 谭洪, 王树荣, 骆仲泱, 岑可法. 生物质三组分热裂解行为的对比研究[J]. 燃料化学学报,2006,34(1):61−65. doi: 10.3969/j.issn.0253-2409.2006.01.013TAN Hong, WANG Shu-rong, LUO Zhong-yang, CEN Ke-fa. Pyrolysis behavior of cellulose, xylan and lignin[J]. JFuel Chem Technol,2006,34(1):61−65. doi: 10.3969/j.issn.0253-2409.2006.01.013 [25] 胡睿, 万诗琪, 毛峰, 王杰. 农业废弃物水洗前后热解特性的变化[J]. 燃料化学学报,2021,49(9):1239−1249.HU Rui, WAN Shi-qi, MAO Feng, WANG Jie. Changes in pyrolysis characteristics of agricultural residues before and after water washing[J]. J Fuel ChemTechnol,2021,49(9):1239−1249. [26] 郑泉兴, 张建平, 李巧灵, 刘秀彩, 黄朝章, 蓝洪桥, 许寒春, 于德德, 刘雯, 叶仲力, 刘江生, 伊晓东, 李斌, 谢卫, 邓楠. 离子色谱-积分脉冲安培法在纸浆纤维单糖组成分析中的应用[J]. 中国造纸,2020,39(7):37−43. doi: 10.11980/j.issn.0254-508X.2020.07.006ZHENG Quan-xing, ZHANG Jian-ping, LI Qiao-ling, LIU Xiu-cai, HUANG Chao-zhang, LAN Hong-qiao, XU Han-chun, YU De-de, LIU Wen, YE Zhong-li, LIU Jiang-sheng, YI Xiao-dong, LI Bin, XIE Wei, DENG Nan. Application of ion chromatography-integrated pulsed amperometric method in the analysis of monosaccharide composition of pulp fiber[J]. China Pulp Paper,2020,39(7):37−43. doi: 10.11980/j.issn.0254-508X.2020.07.006 [27] 龚德鸿, 许成, 顾红艳. 烟梗的热解特性分析[J]. 贵州大学学报(自然科学版),2011,28(4):33−36.GONG De-hong, XU Cheng, GU Hong-yan. Analysis on pyrolysis characteristics of tobacco stem[J]. J Guizhou Univ (Nat Sci),2011,28(4):33−36. [28] 于娟, 章明川, 沈轶, 范卫东, 周月桂. 生物质热解特性的热重分析[J]. 上海交通大学学报,2002,36(10):1475−1478. doi: 10.3321/j.issn:1006-2467.2002.10.022YU Juan, ZHANG Ming-chuan, SHEN Yi, FAN Wei-dong, ZHOU Yue-gui. Thermogravimetric analysis of pyrolysis characteristics of biomass[J]. J Shanghai Jiaotong Univ,2002,36(10):1475−1478. doi: 10.3321/j.issn:1006-2467.2002.10.022 [29] DIBLASI C. Modeling chemical and physical processes of wood and biomass pyrolysis[J]. Prog Energ Combust,2008,34(1):47−90. doi: 10.1016/j.pecs.2006.12.001 [30] VYAZOVKIN S, CHRISSAFIS K, LORENZO M L D, KOGA N, PIJOLAT M, RODUIT B, SBIRRAZZUOLI N, SU OL J J. ICTAC kinetics committee recommendations for collecting experimental thermal analysis data for kinetic computations[J]. Thermochim Acta,2014,590:1−23. doi: 10.1016/j.tca.2014.05.036 [31] XIONG S, ZHANG S, WU Q, GUO X, DONG A, CHEN C. Investigation on cotton stalk and bamboo sawdust carbonization for barbecue charcoal preparation[J]. Bioresour Technol,2014,152:86−92. doi: 10.1016/j.biortech.2013.11.005 -




 下载:
下载: