Preparation and properties of carbon-based electrocatalysts from gasification fine slag for oxygen reduction
-
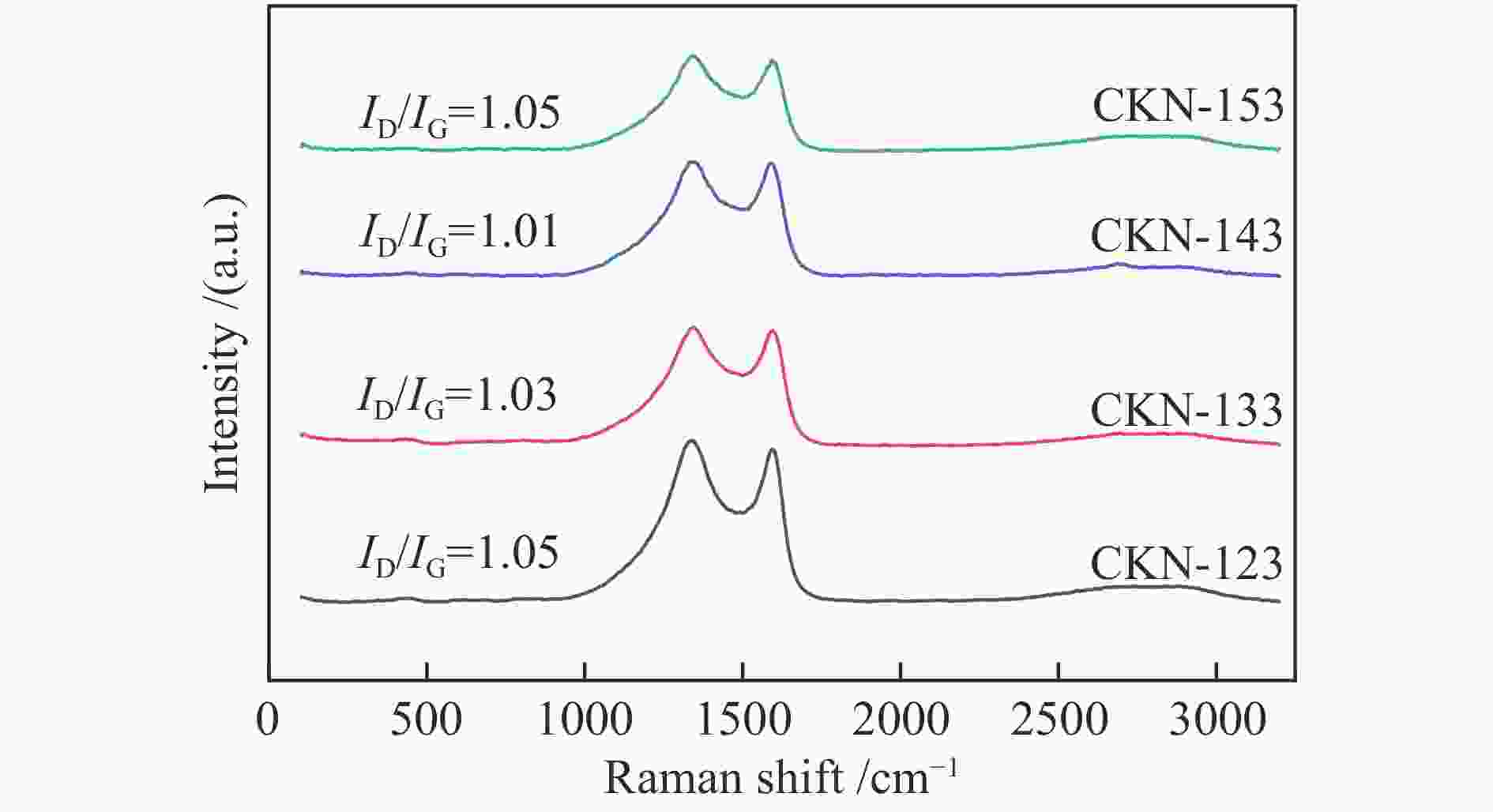
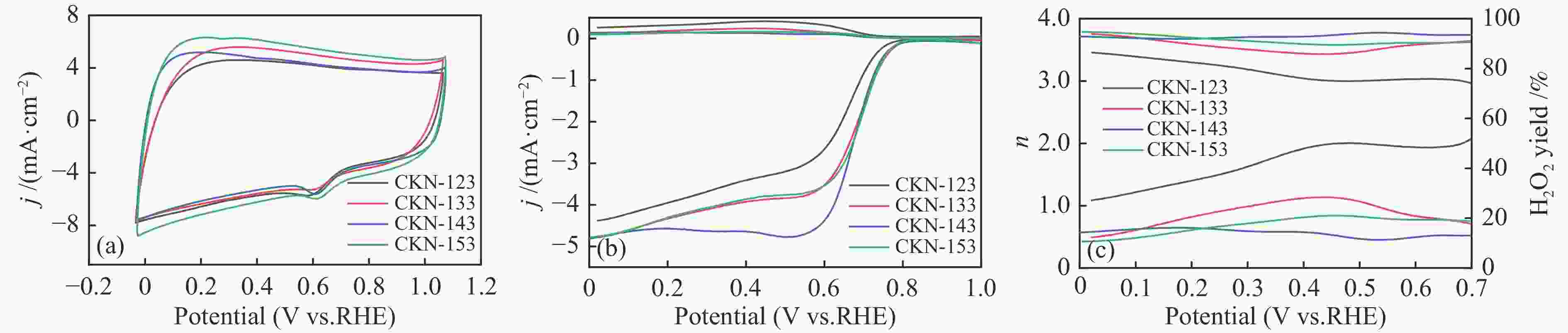
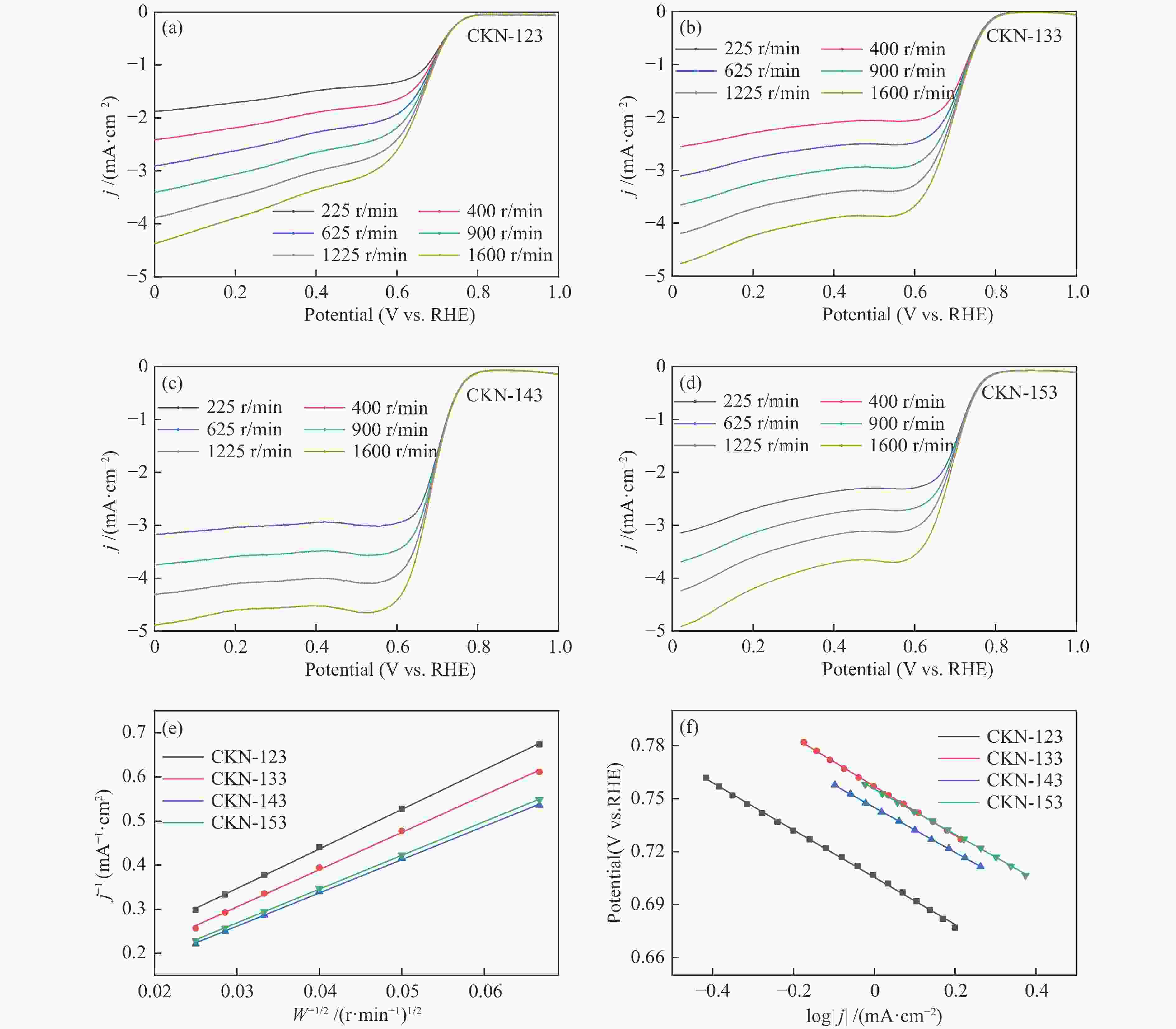
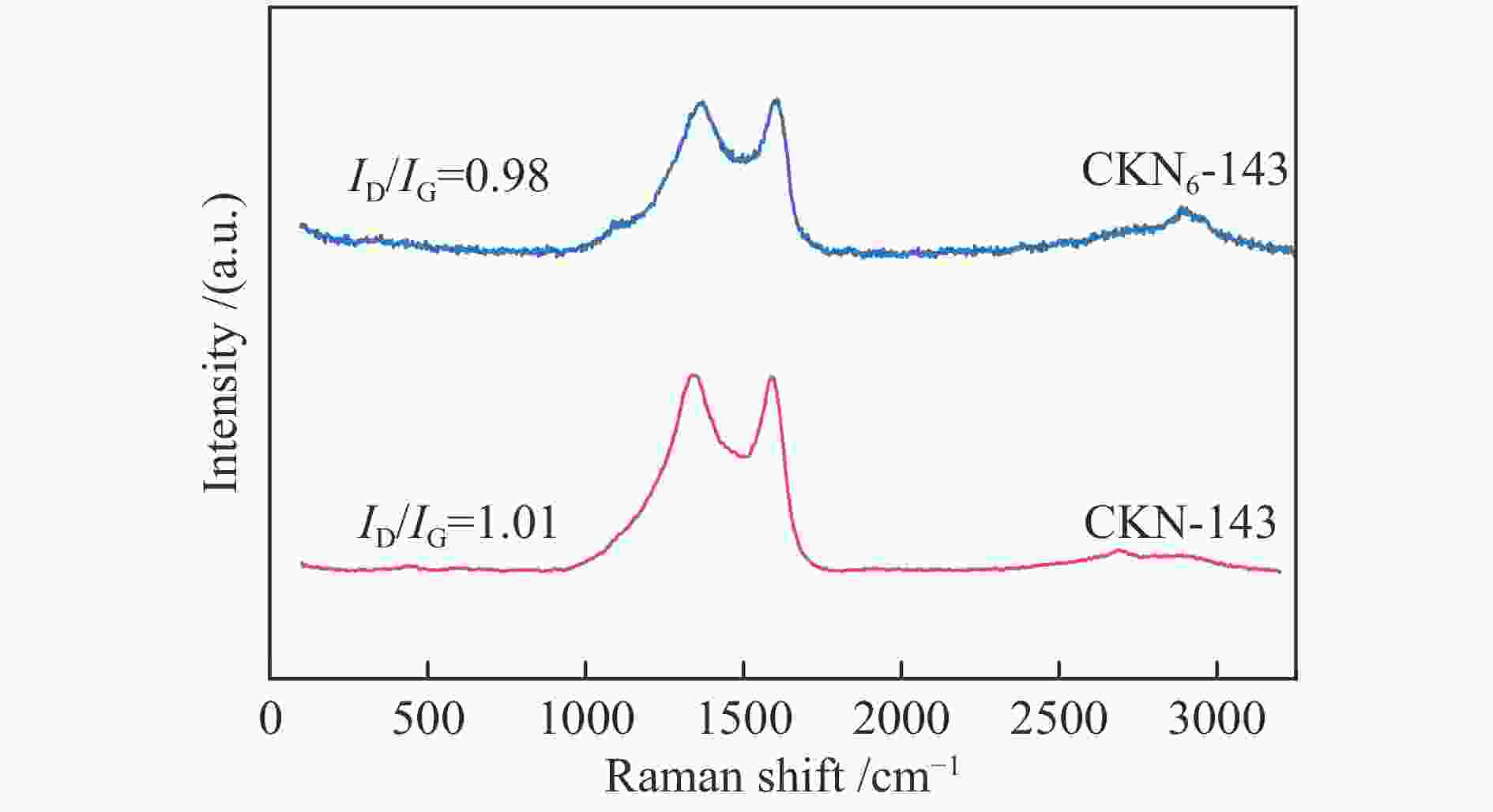
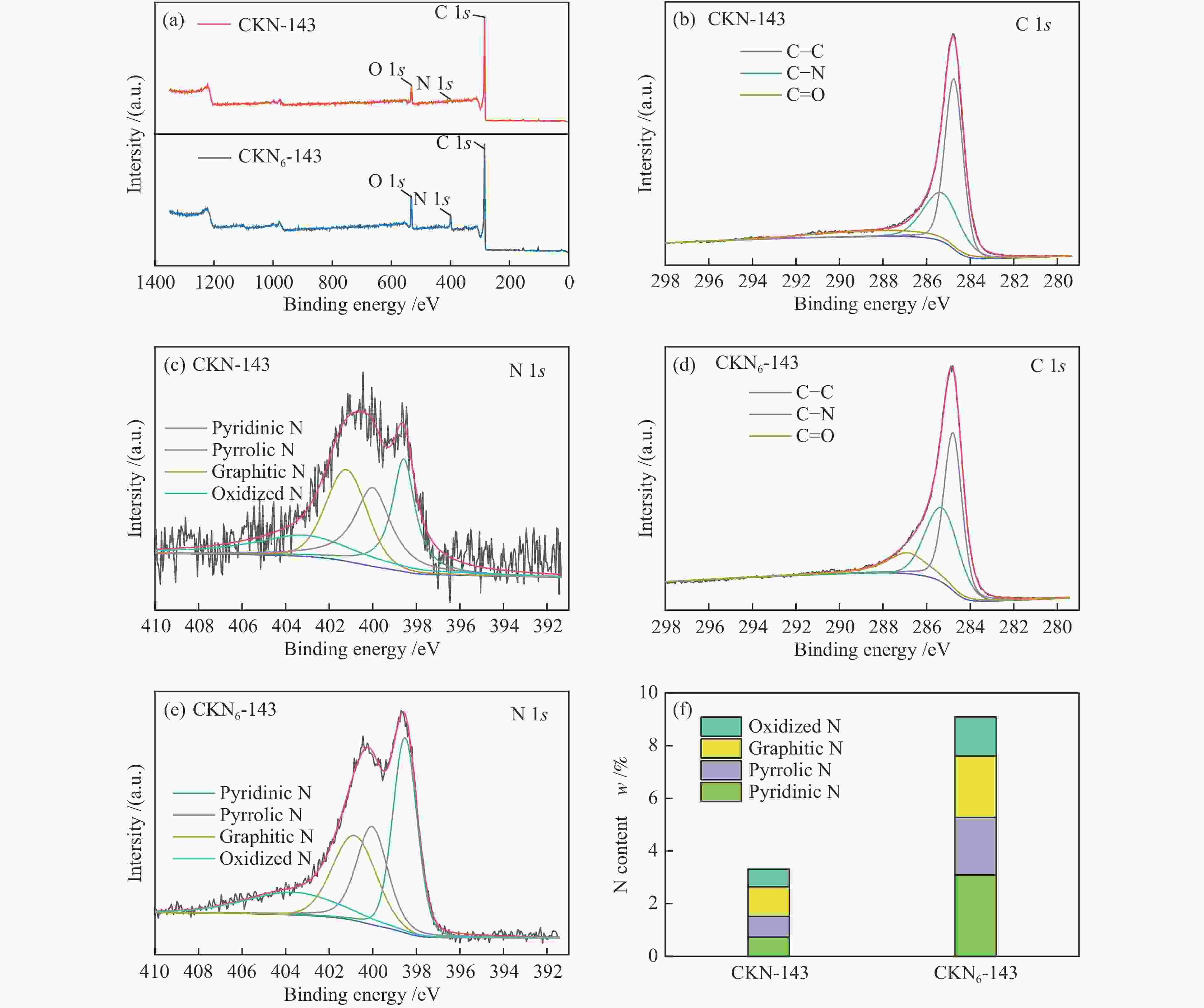
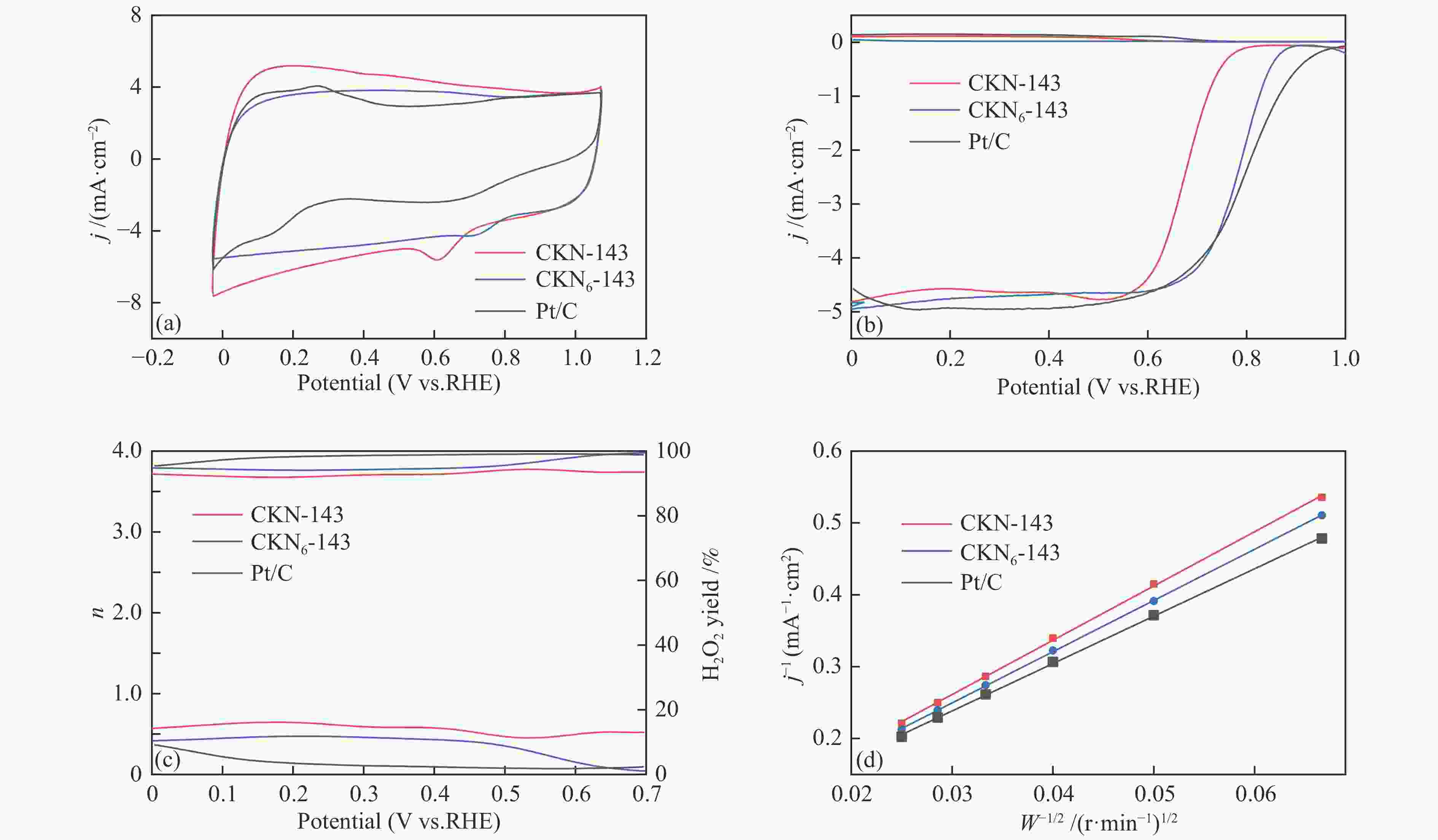
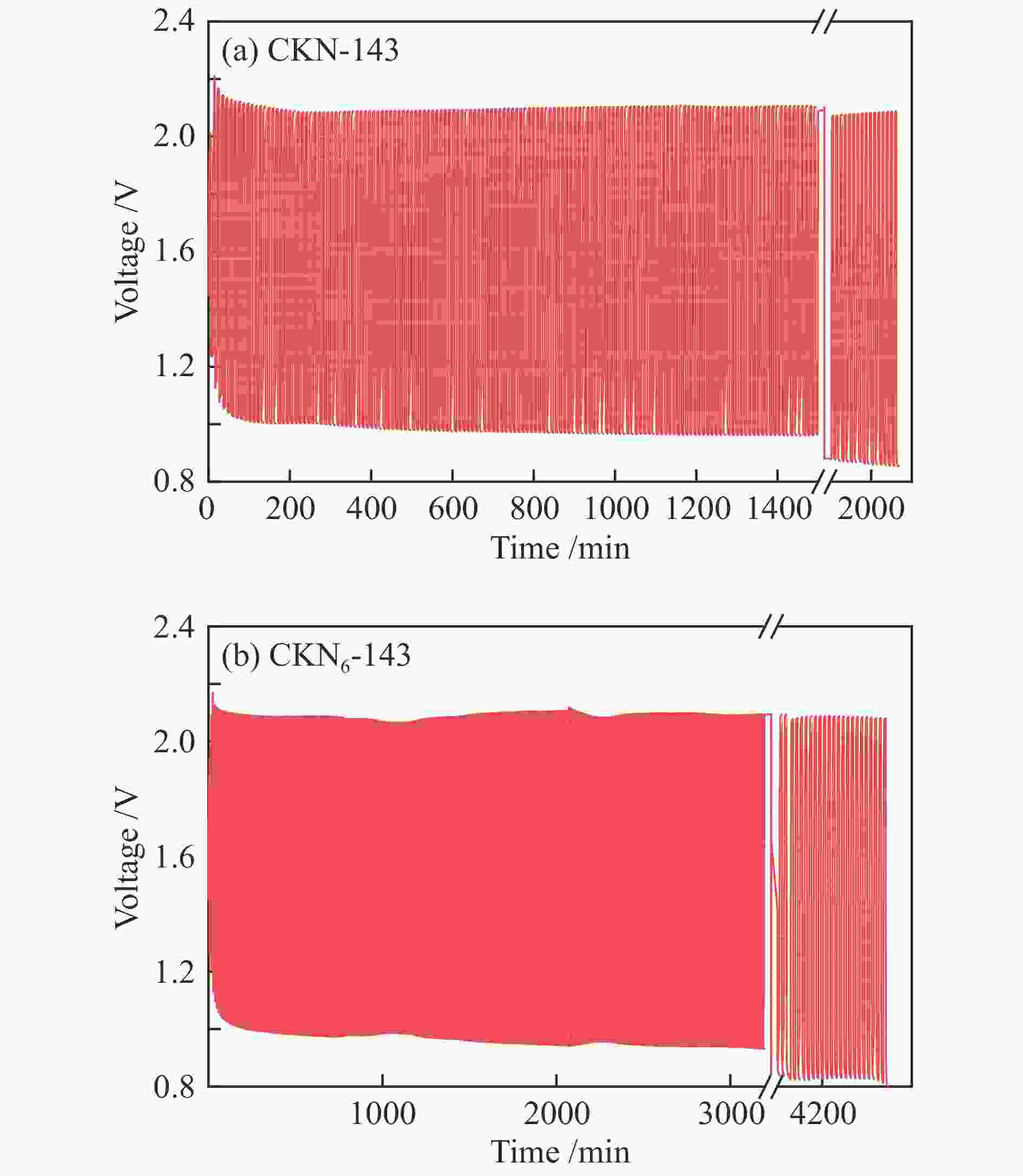
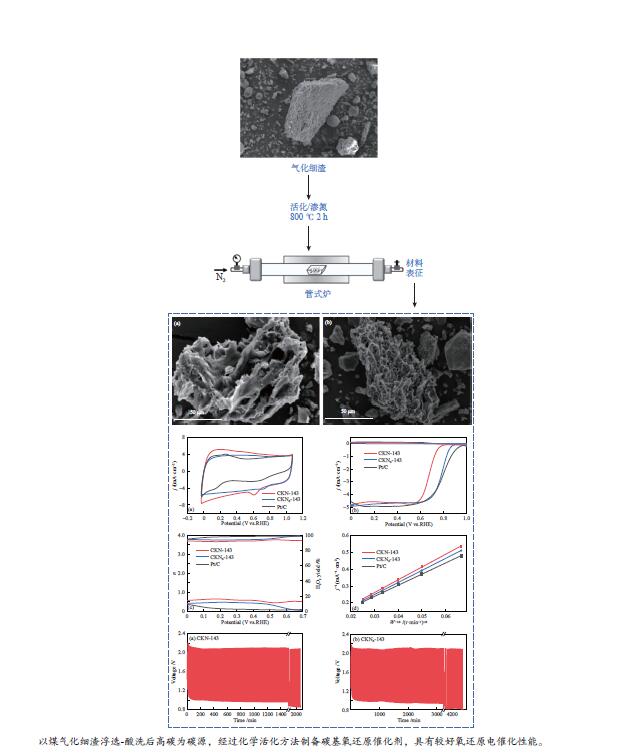
摘要: 气化细渣是煤气化过程产生的一种含碳量较高的固体废弃物。经炭灰分离得到的高炭是制备炭材料的潜在碳源。本研究以气化细渣浮选-酸洗后的高炭为前驱体,通过高温活化制备了氮掺杂碳基催化剂,结合拉曼光谱、XPS和SEM等表征,探究了活化剂比例和氮源对催化剂理化特性的影响,揭示了两者与催化剂氧还原性能的内在关联,验证了气化细渣作为原料制备碳基氧还原催化剂的可行性。结果表明,随着活化剂KOH比例的增加,碳基催化剂的氧还原催化性能先增加后减小,当高炭和KOH质量比为1∶4时其催化性能最优。此外,相较于氯化铵,以三聚氰胺作为氮源具有更强的氮掺杂效应,使得CKN6-143催化剂的起始电位可达0.87 V(vs. RHE),极限扩散电流密度为4.95 mA/cm2,平均电子转移数为3.82,表现出良好的电催化性能,为气化细渣的高值化利用奠定了基础。Abstract: Gasification fine slag is a kind of solid waste with high carbon content produced in coal gasification process. The residual carbon obtained by carbon ash separation is a potential carbon source for preparing high quality carbon materials. In this study, the carbon residue after flotation of fine gasification slag was used as the precursor to prepare N-doped carbon-based electrocatalysts through high temperature activation. Combined with the characterization of Raman spectroscopy, XPS and SEM, the influence of activator ratio and nitrogen source on the physical and chemical structure of the catalyst was explored. The intrinsic relationship between activation formula and oxygen reduction performance of catalyst was revealed. The feasibility of preparing carbon-based electrocatalysts with gasification fine slag as raw material was verified. The results demonstrate that the oxygen reduction catalytic performance of carbon materials increases first and then decreases with the increase of the proportion of KOH. The optimal catalytic performance was achieved when the mass ratio of carbon residue to KOH was 1:4. In addition, melamine has stronger nitrogen doping effect as a nitrogen source than NH4Cl, making the initial potential of CKN6-143 up to 0.87 V (vs. RHE), the limiting current density is 4.95 mA/cm2, and the average electron transfer number is 3.82, indicating that CKN6-143 has good electrocatalytic performance. The results provide a possibility for preparing oxygen reduction catalyst with gasification fine slag.
-
Key words:
- gasification fine slag /
- oxygen reduction reaction /
- porous carbon /
- N-doped
-
表 1 原料的工业分析和元素分析
Table 1 Proximate and ultimate analyses of raw materials
Sample Proximate analysis wad/% Ultimate analysis wad/% M V A FC C H O N S FS 3.52 13.63 46.75 36.10 38.27 2.25 8.22 0.61 0.38 AC 2.29 23.46 25.67 48.58 51.37 2.82 12.95 2.07 2.83 HC 2.78 25.20 14.65 57.37 59.08 3.34 16.90 2.21 1.04 表 2 原料的灰成分分析
Table 2 Ash composition analysis of raw materials
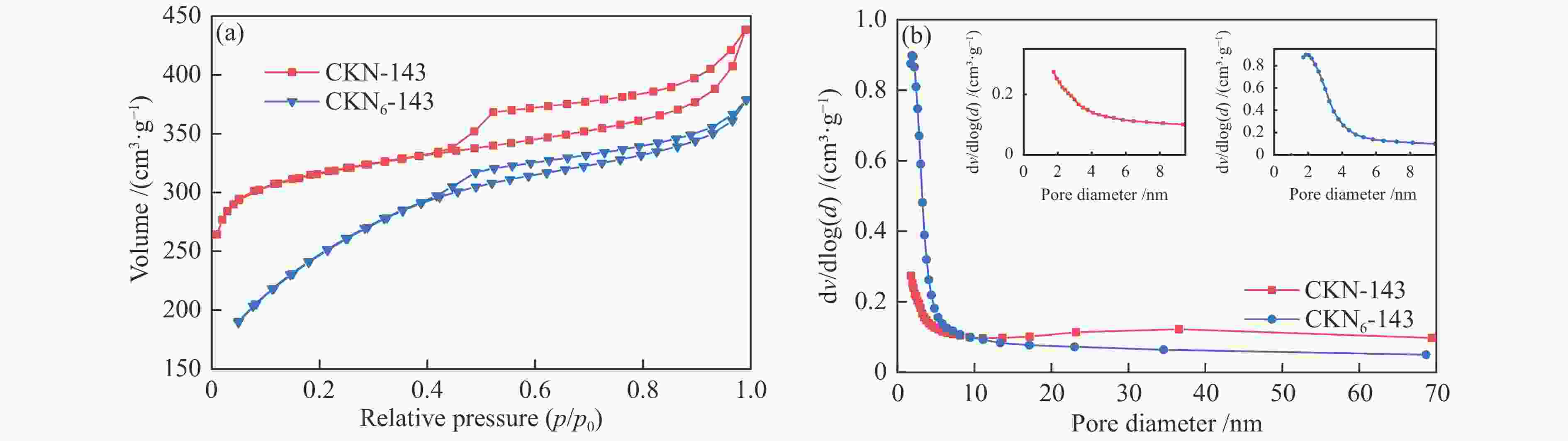
Sample Content w/% SiO2 Al2O3 Fe2O3 CaO SO3 Na2O MgO K2O others FS 44.14 17.81 16.32 13.91 2.18 2.24 1.12 1.04 1.24 AC 44.98 18.48 12.86 14.31 2.46 2.10 1.29 1.18 2.34 HC 81.04 5.85 3.14 3.49 3.38 0.21 0.23 0.71 1.95 表 3 CKN-143和CKN6-143的比表面积和孔容
Table 3 Specific surface area (SSA) and pore volume of CKN-143 and CKN6-143
Catalyst A/(m2·g−1) Total pore
volume/(cm3·g−1)Micropore
volume/(cm3·g−1)Mesoporous and macroporous
volume/(cm3·g−1)CKN-143 987.49 0.68 0.38 0.30 CKN6-143 847.54 0.59 0.07 0.52 -
[1] 曲江山, 张建波, 孙志刚, 杨晨年, 史达, 李少鹏, 李会泉. 煤气化渣综合利用研究进展[J]. 洁净煤技术,2020,26(1):184−193.QU Jiang-shan, ZHANG Jian-bo, SUN Zhi-gang, YANG Chen-nian, SHI Da, LI Shao-peng, LI Hui-quan. Research progress on comprehensive utilization of coal gasification slag[J]. Clean Coal Technol,2020,26(1):184−193. [2] LUCIE B. Unburned carbon from coal combustion ash: An overview[J]. Fuel Process Technol,2015,134:136−158. doi: 10.1016/j.fuproc.2015.01.028 [3] ZHANG Y C, LI H X, GAO S T, GENG, Y, WU C L. A study on the chemical state of carbon present in fine ash from gasification[J]. Asia-Pac J Chem Eng,2019,14(4):2336. [4] 李恒, 赵丽丽, 龚岩, 崔启迪, 郭庆华, 于广锁. 表面活性剂复配对气化细渣浮选影响的试验研究[J]. 中国电机工程学报, https://doi.org/10.13334/j.0258-8013.pcsee.211309.LI Heng, ZHAO Li-li, GONG Yan, CUI Qi-di, GUO Qing-hua, YU Guang-suo. Experimental study on the performance of surfactants on the flotation effect of gasification fine slag[J]. Proc CSEE, https://doi.org/10.13334/j.0258-8013.pcsee.211309. [5] 黄宇宸. 煤转化固体产物的气化反应活性及电化学性能研究[D]. 上海: 华东理工大学, 2020.HUANG Yu-chen. On study gasification reaction activity and electrochemical performance of coal conversion solid product[D]. Shanghai: East China University of Science and Technology, 2020. [6] 史兆臣, 戴高峰, 王学斌, 董永胜, 李攀, 于伟, 谭厚章. 煤气化细渣的资源化综合利用技术研究进展[J]. 华电技术,2020,42(7):63−73. doi: 10.3969/j.issn.1674-1951.2020.07.009SHI Zhao-chen, DAI Gao-feng, WANG Xue-bin, DONG Yong-sheng, LI Pan, YU Wei, TAN Hou-zhang. Review on the comprehensive resources utilization technology of coal gasification fine slag[J]. Huadian Technol,2020,42(7):63−73. doi: 10.3969/j.issn.1674-1951.2020.07.009 [7] 王萌. 质子交换膜燃料电池设计与综合优化研究[D]. 北京: 北京科技大学, 2019.WANG Meng. Design and comprehensive optimization of proton exchange membrane fuel cells[D]. Beijing: University of Science and Technology Beijing, 2019. [8] 孙玥. 碳基氧还原催化剂的制备和性能研究[D]. 济南: 济南大学, 2020.SUN Yue. Study on preparation and properties of carbon-based oxygen reduction catalysts[D]. Jinan: University of Jinan, 2020. [9] 丁晓波, 覃远航. N、S共掺杂碳基高效氧还原催化剂的制备[J]. 武汉工程大学学报,2021,43(4):362−366.DING Xiao-bo, QIN Yuan-hang. Preparation of N, S Co-doped carbon as highly efficient catalyst for oxygen reduction[J]. J Wuhan Inst Technol,2021,43(4):362−366. [10] KUANPING G, FENG D, ZHENHAI X, DURSTOCK M, LIMING D. Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction[J]. Science,2009,323(5915):760−764. doi: 10.1126/science.1168049 [11] CAO Y L, MAO S J, LI M M, CHEN Y Q, WANG Y. Metal/porous carbon composites for heterogeneous catalysis: old catalysts with improved performance promoted by N-doping[J]. ACS Catal,2017,7(12):8090−8112. doi: 10.1021/acscatal.7b02335 [12] RUBIO B, IZQUIERDO M T, MAYORAL M C, BONA M T, MARTÍNEZ-TARAZONA R M. Preparation and characterization of carbon-enriched coal fly ash[J]. J Environ Manage,2008,88(4):1562−1570. doi: 10.1016/j.jenvman.2007.07.027 [13] DENG D H, PAN X L, YU L A, CUI Y, JIANG Y P, QI J, LI W X, FU Q A, MA X C, XUE Q K, SUN G Q, BAO X H. Toward N-doped graphene via solvothermal synthesis[J]. Chem Mater,2010,23(5):1188−1193. [14] REYIMJAN A S, ALFRED B A, NALINI P S, SWAMINATHA P K, BRANKO N P. O2 reduction on graphite and nitrogen-doped graphite: experiment and theory[J]. J Phys Chem B,2006,110(4):1787−1793. doi: 10.1021/jp055150g [15] XING T, ZHENG Y, LI L H, COWIE B C C, GUNZELMANN D, QIAO S Z, HUANG S M, CHEN Y. Observation of active sites for oxygen reduction reaction on nitrogen-doped multilayer graphene[J]. ACS Nano,2014,8(7):6856−6862. doi: 10.1021/nn501506p [16] JIANG T, JIANG W, LI Y, XU Y, WANG Y. Facile regulation of porous N-doped carbon-based catalysts from covalent organic frameworks nanospheres for highly-efficient oxygen reduction reaction[J]. Carbon,2021,180(23):92−100. [17] JINTAO Z, TONG Z, JING M, ZHI W, JUNHAO L, XUZHONG G. ORR and OER of Co–N codoped carbon-based electrocatalysts enhanced by boundary layer oxygen molecules transfer[J]. Carbon,2021,172:556−568. doi: 10.1016/j.carbon.2020.10.075 [18] JUNYU Z, GONG Z, SHENGYAO J, YUJUN Z, QINGHUA J, HUACHUN L, HUIJUAN L, JIUHUI Q. Graphitic N in nitrogen-doped carbon promotes hydrogen peroxide synthesis from electrocatalytic oxygen reduction[J]. Carbon,2020,163:154−161. [19] ZHI Q, SOOYEON H, XING L, CHENYU W, WIDITHA S, STAVROS K, DONGGUO L, MENGJIE C, YANGHUA H, MAOYU W, ZHENYU L, GUOFENG W, HUA Z, ZHENXING F, DONG S, JACOB S S, GANG W. 3D porous graphitic nanocarbon for enhancing the performance and durability of Pt catalysts: A balance between graphitization and hierarchical porosity[J]. Energy Environ Sci,2019,12(9):2830−2841. doi: 10.1039/C9EE01899A [20] JOSEPH V G, JAMES N B, EHREN E, MOHAMMAD Z R, MANISH V N, KEITH A F, SHERY S C, DAVID C C, BUDDIE M. Evaluation of two potassium-based activation agents for the production of oxygen- and nitrogen-doped porous carbons[J]. Energy Fuels,2020,34(5):6101−6112. doi: 10.1021/acs.energyfuels.0c00427 [21] ZHANG J, SONG L H, ZHAO C F, YIN X P, ZHAO Y F. Co, N co-doped porous carbons as high-performance oxygen reduction electrocatalysts[J]. New Carbon Mater,2021,36(1):209−218. doi: 10.1016/S1872-5805(21)60016-1 [22] CHAO W, RESHMA R R, PENG J Y, HUANG B T, IFAN E L S, MARCEL R, XU Z C J, YANG S H. Recommended practices and benchmark activity for hydrogen and oxygen electrocatalysis in water splitting and fuel cells[J]. Adv Mater,2019,31(31):1806296. doi: 10.1002/adma.201806296 [23] XIANGLONG W, YIZHAO L, CHAO Y, YALI C, XINTAI S, MUHAMMAD U. Self-template porous carbon by direct activation of high-ash coal liquefaction residue for high-rate supercapacitor electrodes[J]. Int J Energy Res,2021,45(3):4782−4792. doi: 10.1002/er.6096 [24] HE W H, WANG Y, JIANG C H, LU L H. Structural effects of a carbon matrix in non-precious metal O2-reduction electrocatalysts[J]. Chem Soc Rev,2016,45(9):2396−2409. doi: 10.1039/C5CS00665A [25] LIANG J, JIAO Y, JARONIEC M, QIAO S Z. Sulfur and nitrogen dual-doped mesoporous graphene electrocatalyst for oxygen reduction with synergistically enhanced performance[J]. Angew Chem Int Ed,2012,51(46):11496−11500. doi: 10.1002/anie.201206720 [26] CHOI C H, PARK S H, WOO S I. Phosphorus-nitrogen dual doped carbon as an effective catalyst for oxygen reduction reaction in acidic media: effects of the amount of P-doping on the physical and electrochemical properties of carbon[J]. J Mater Chem,2012,22(24):12107−12115. doi: 10.1039/c2jm31079a -





 下载:
下载: