Levoglucosenone production by catalytic pyrolysis of cellulose using ionic liquid as catalyst
-
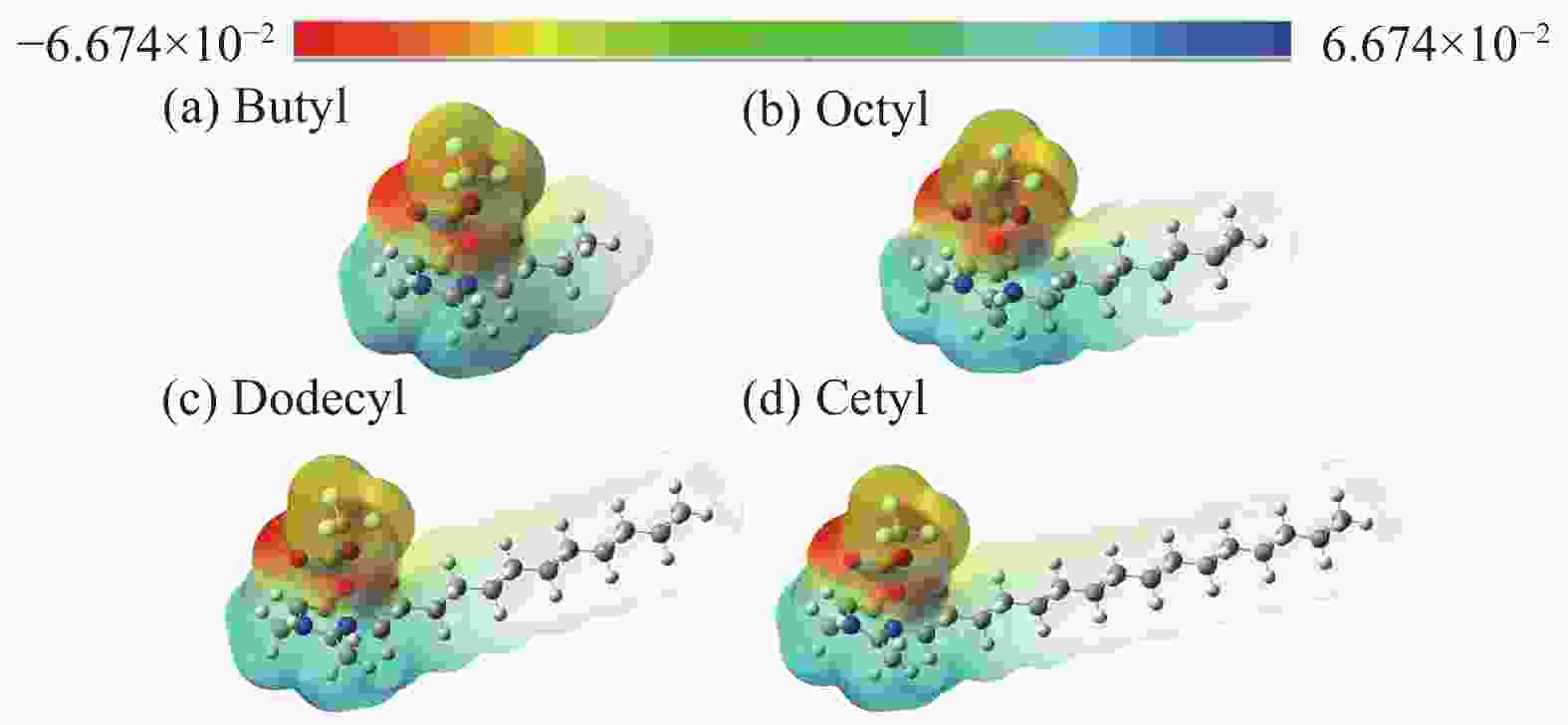
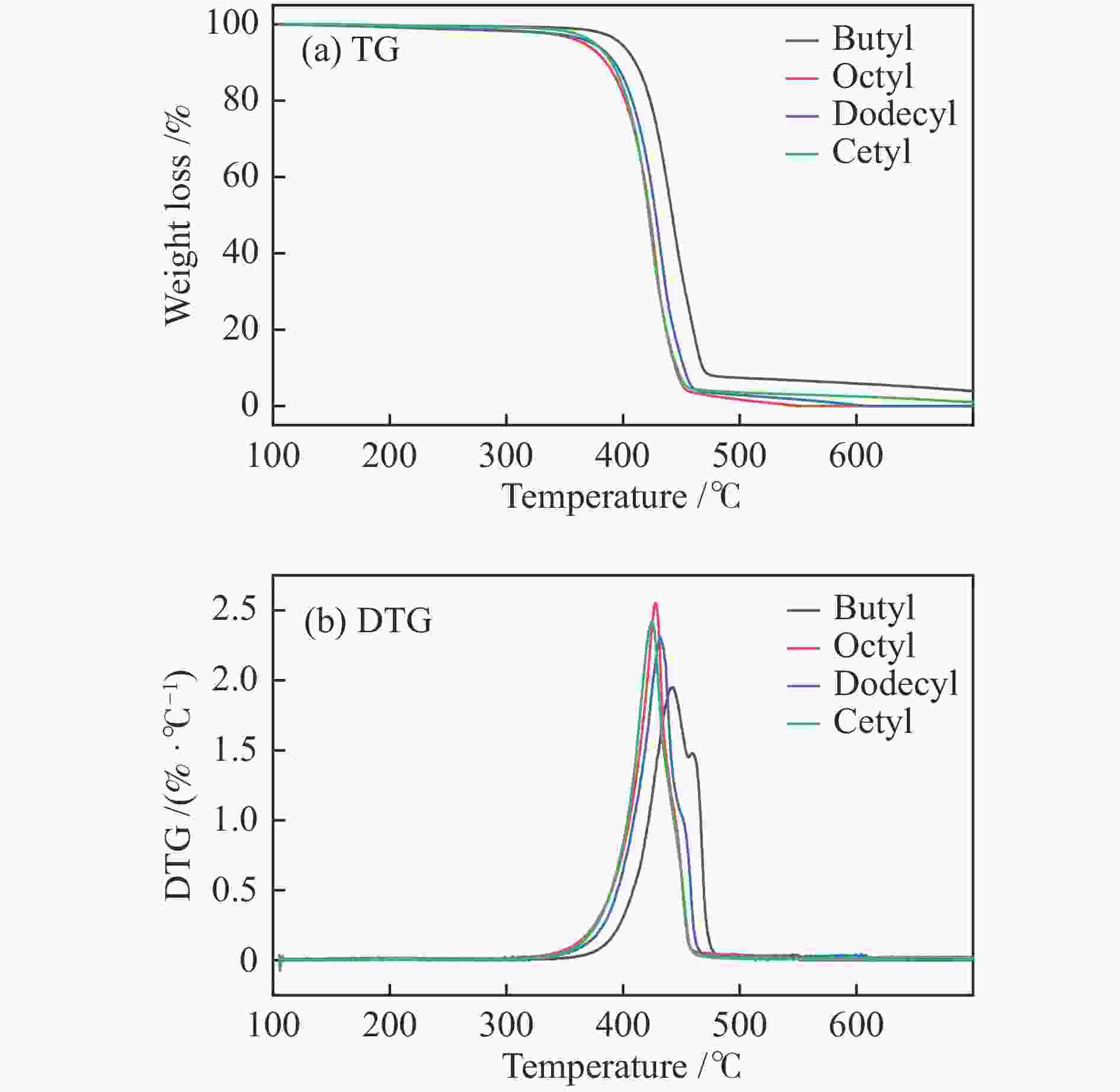
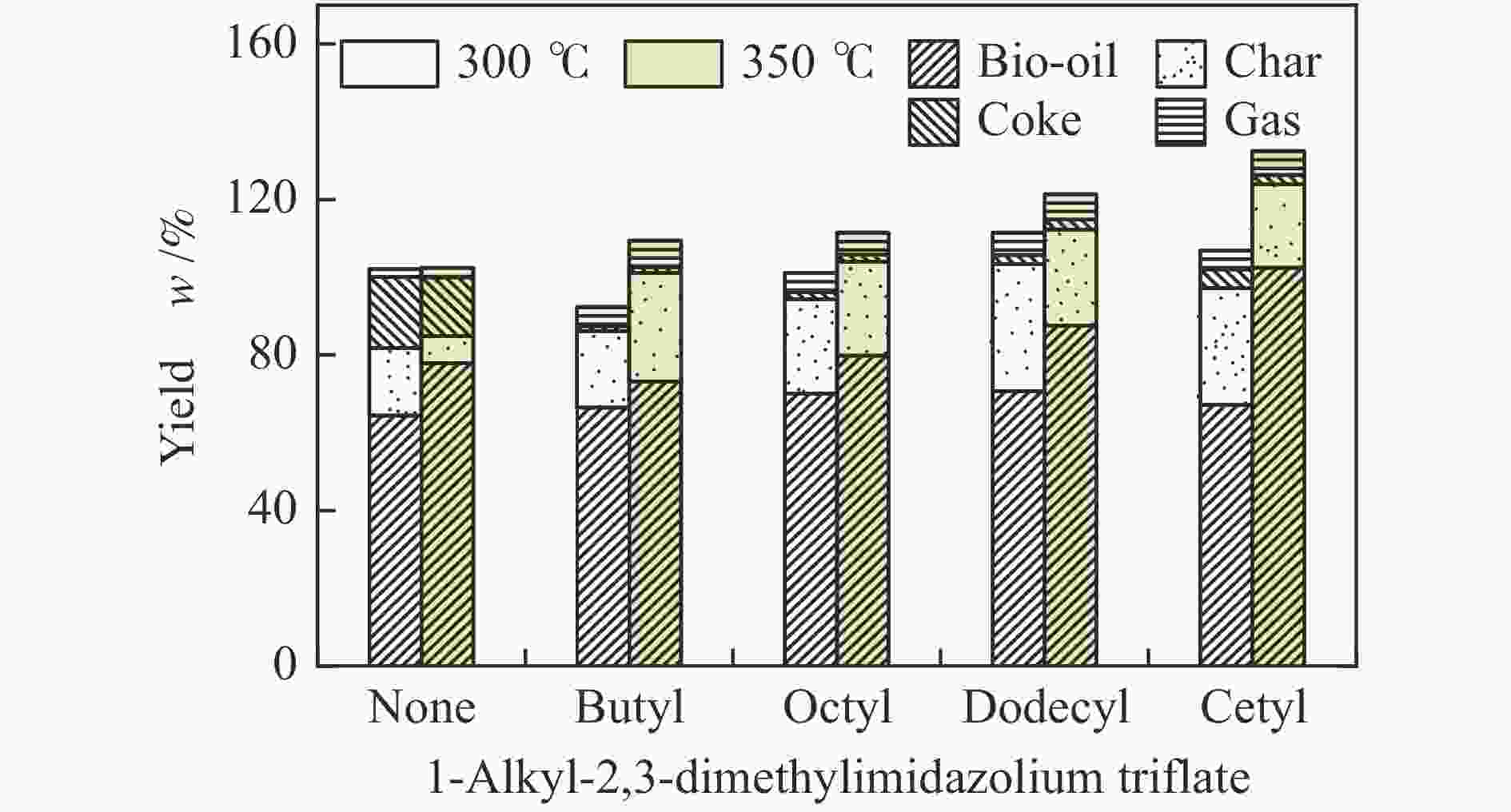
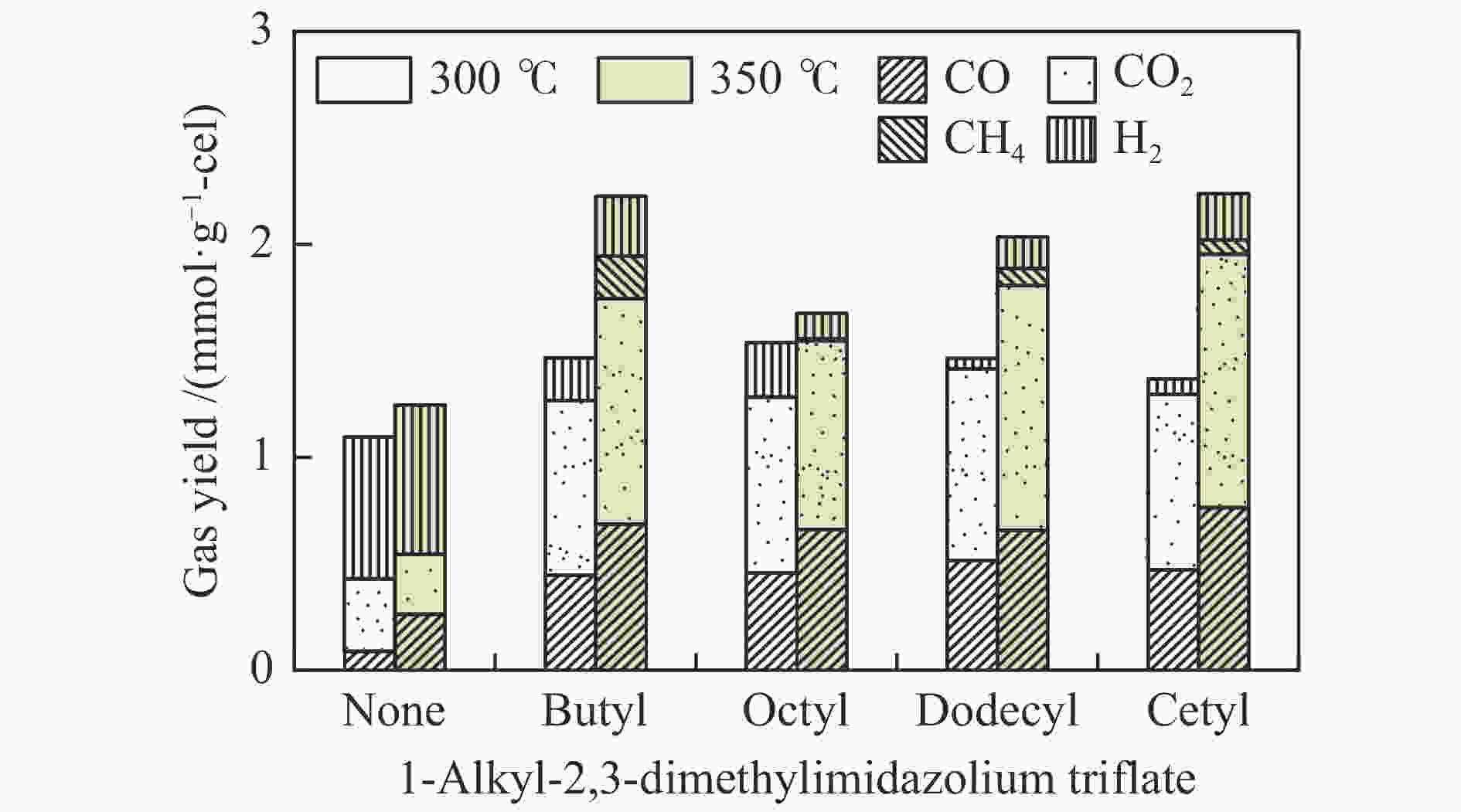
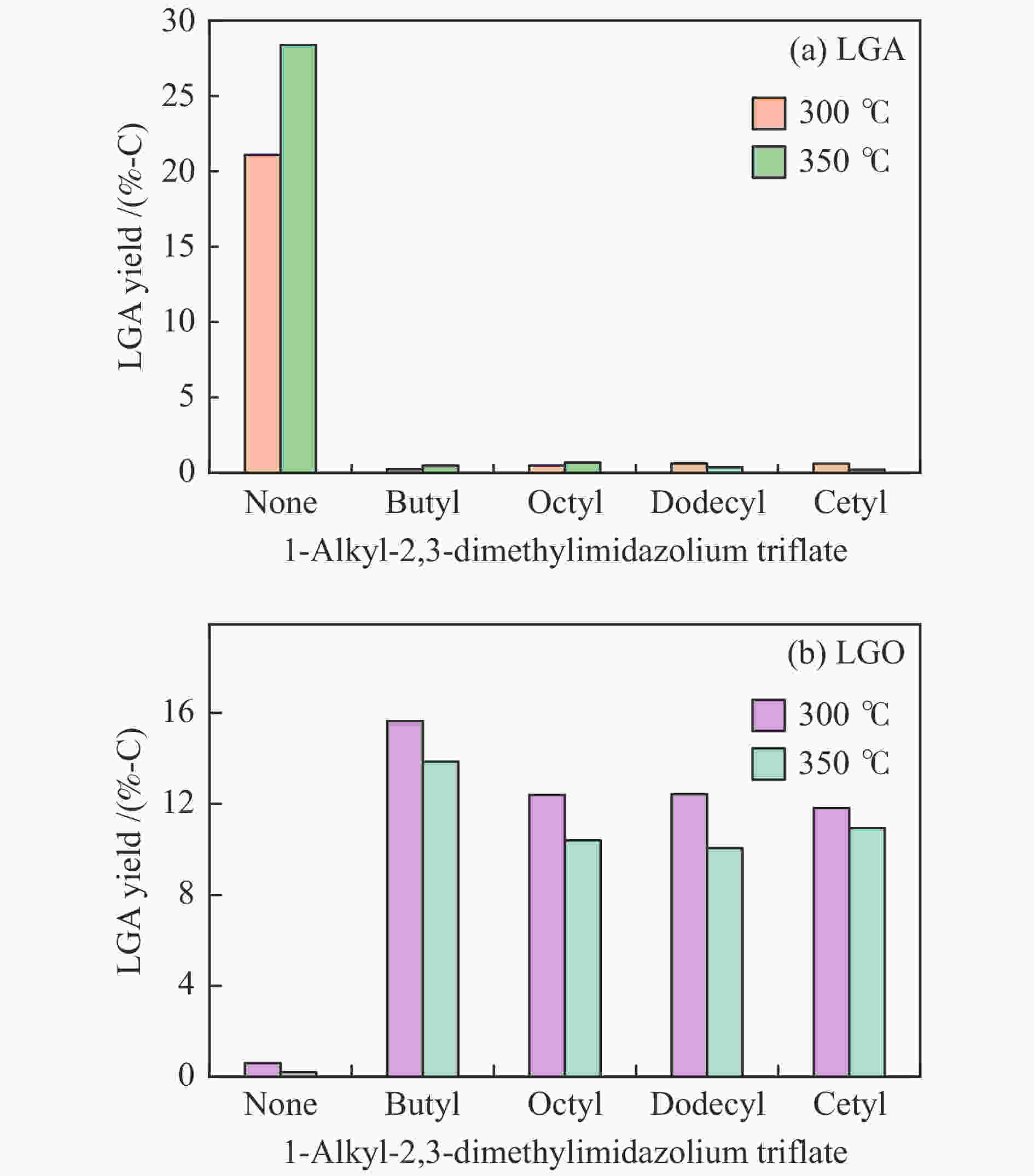
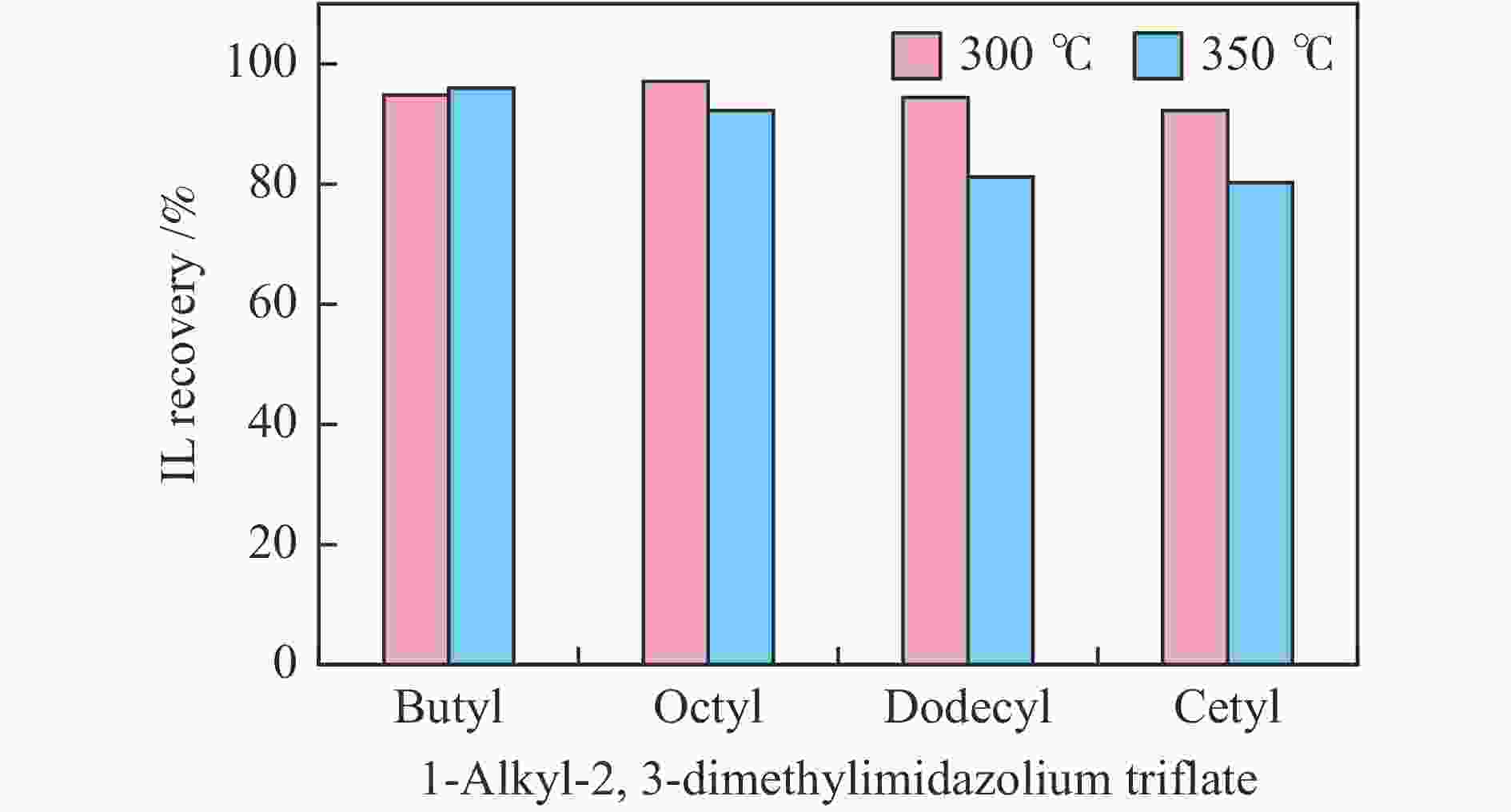
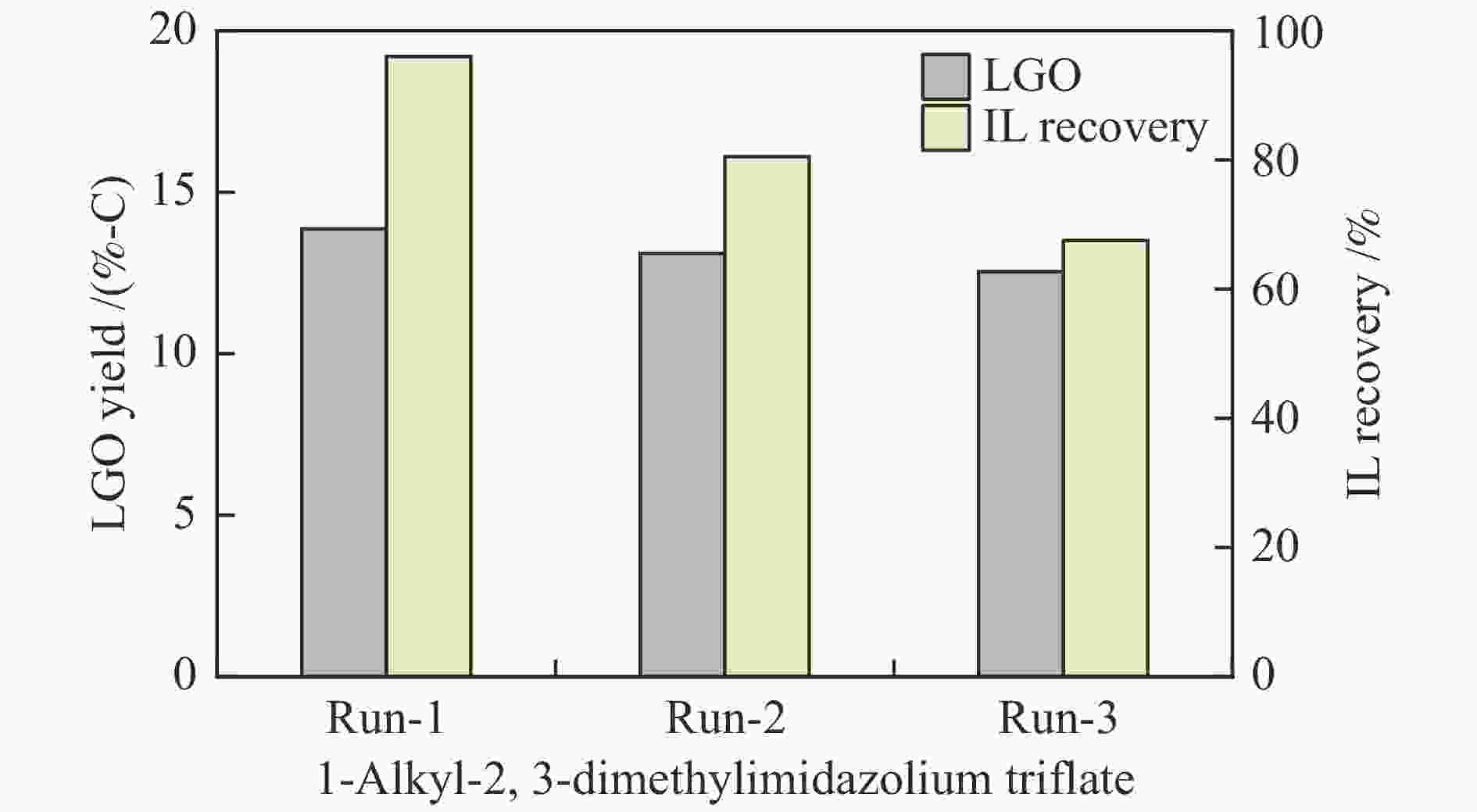
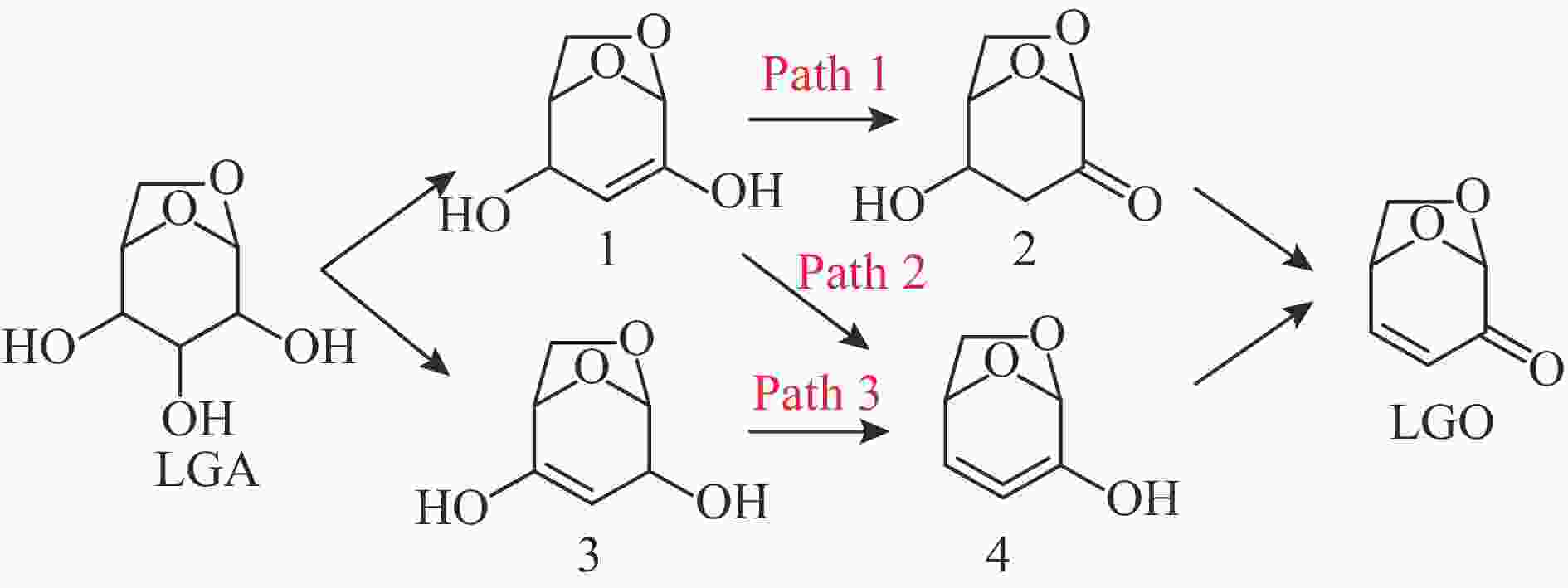
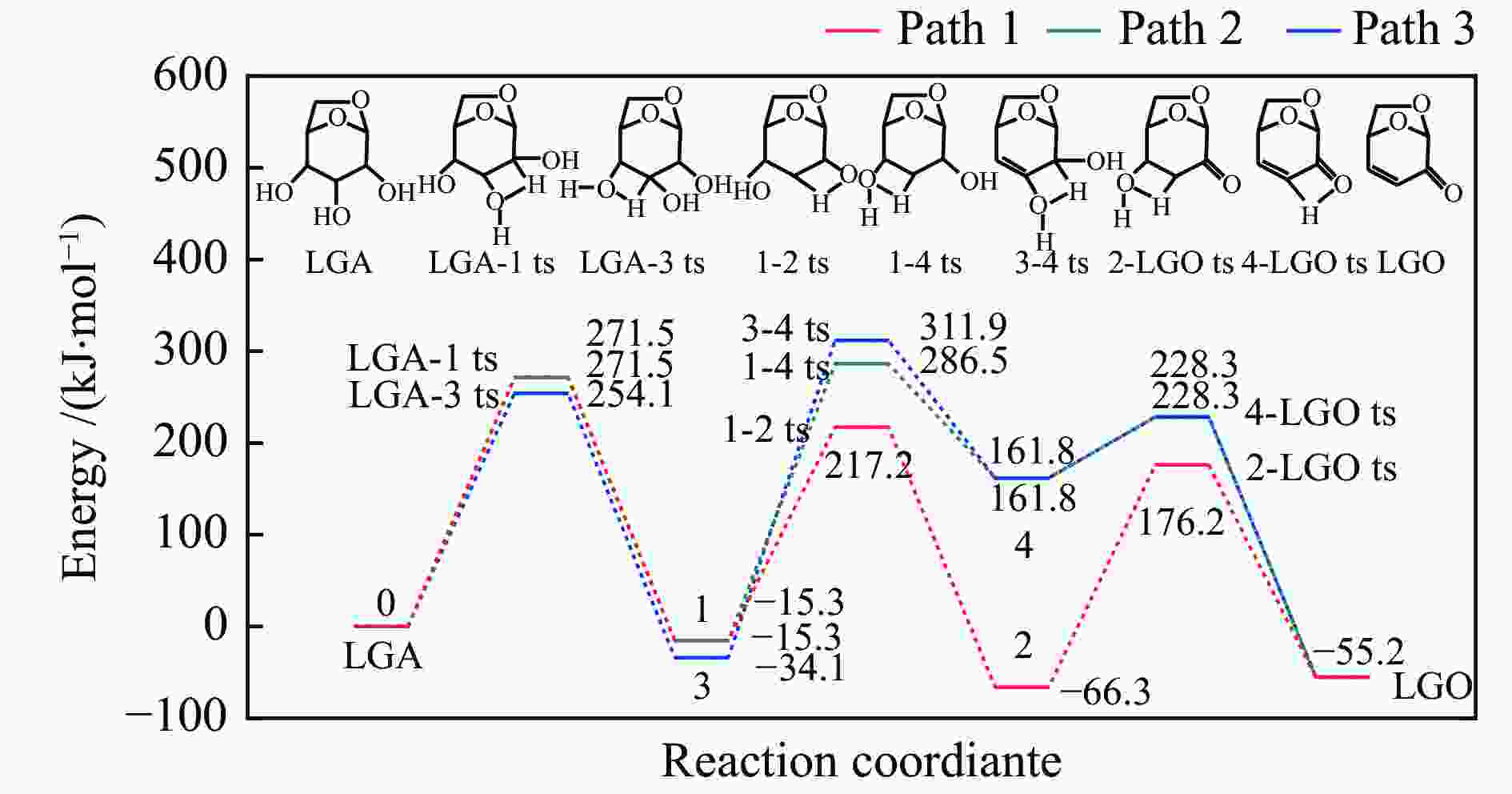
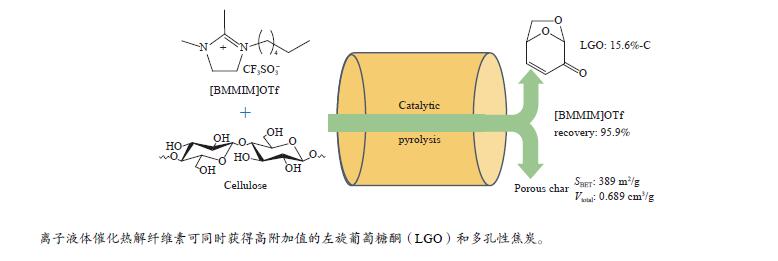
摘要: 纤维素是世界上含量最丰富的可再生有机碳资源之一,左旋葡萄糖酮(LGO)是来源于纤维素热解的一种高附加值平台化合物。本研究考察了离子液体烷基侧链长度对纤维素催化热解制备LGO的影响规律。实验结果表明,最短侧链的1-丁基-2,3-二甲基三氟甲烷磺酸咪唑离子液体对LGO表现了最好的催化效果,其原因是侧链长度减少导致离子液体阴阳离子间相互作用减弱,使离子液体扩散增强。在300 ℃热解时 LGO产率达到15.6%-C,离子液体的回收率为95.9%,其重复利用三次后LGO的产率只有轻微下降。通过密度泛函理论得到了LGO的最佳生成路径,其最低反应活化能为176.2 kJ/mol。此外,本方法也可同时获得多孔性的焦炭,其最高比表面积和孔容分别为389.4 m2/g和0.689 cm3/g。Abstract: Cellulose is one of the most abundant renewable organic carbon resources in the world. Levoglucosenone (LGO) is a high value-added platform chemical derived from cellulose pyrolysis. In this study, the influence of ionic liquid catalyst on the production of LGO by catalytic pyrolysis of cellulose was revealed. The results showed that 1-butyl-2,3-dimethylimidazolium triflate performed best for the LGO formation. The reason was that the decrease in the length of the side chain weakened the interaction between the cation and anion of the ionic liquid, which increased the diffusion of the ionic liquid. LGO reached a yield of 15.6%-C at pyrolysis temperature of 300 ℃, and the recovery rate of ionic liquid attained to 95.9%. Besides, LGO yield only slightly decreased after 3 times re-utilization of the ionic liquid. The formation path of LGO was calculated by density functional theory. The result showed the lowest activation energy was 176.2 kJ/mol. Moreover, this method was effective to obtain porous char at the same time, and the highest specific surface area and pore volume were 389.4 m2/g and 0.689 cm3/g, respectively.
-
Key words:
- cellulose /
- ionic liquid /
- pyrolysis /
- levoglucosenone /
- porous char
-
表 1 GC/MS检测到的化合物的定量分析(mg/g-纤维素)
Table 1 Quantitative analysis of compounds detected by GC/MS (mg/g-cellulose)
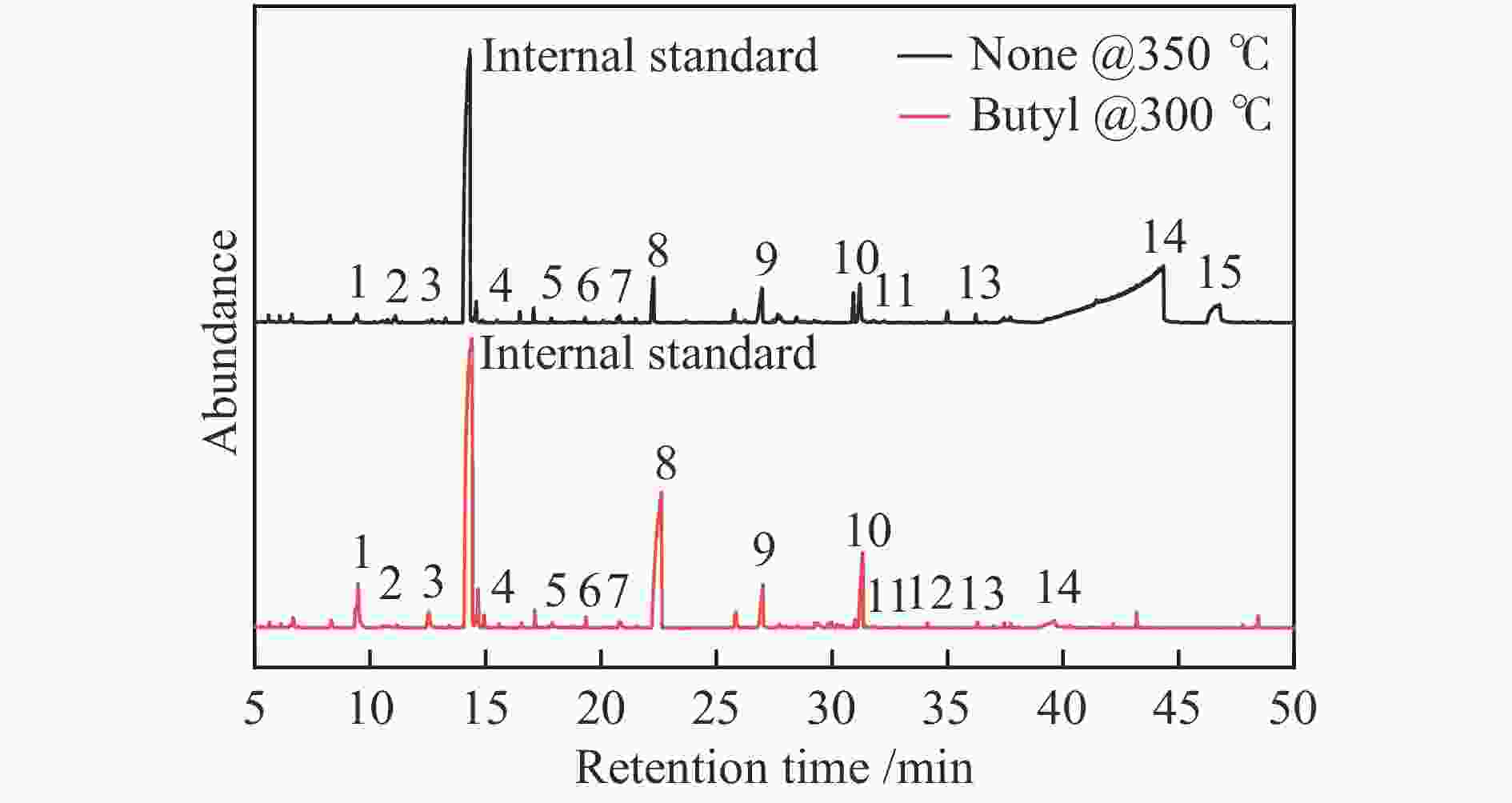
Peak Compound None Butyl Octyl Dodecyl Cetyl 300 ℃ 350 ℃ 300 ℃ 350 ℃ 300 ℃ 350 ℃ 300 ℃ 350 ℃ 300 ℃ 350 ℃ 1 furfural 0.9 1.0 13.8 2.9 6.7 3.3 4.4 5.6 5.9 3.1 2 4-hydroxydihydro
furan-2(3H)-one0.2 0.4 0.3 1.5 0.4 0.4 0.4 0.8 0.7 0.3 3 2-hydroxycyclopent
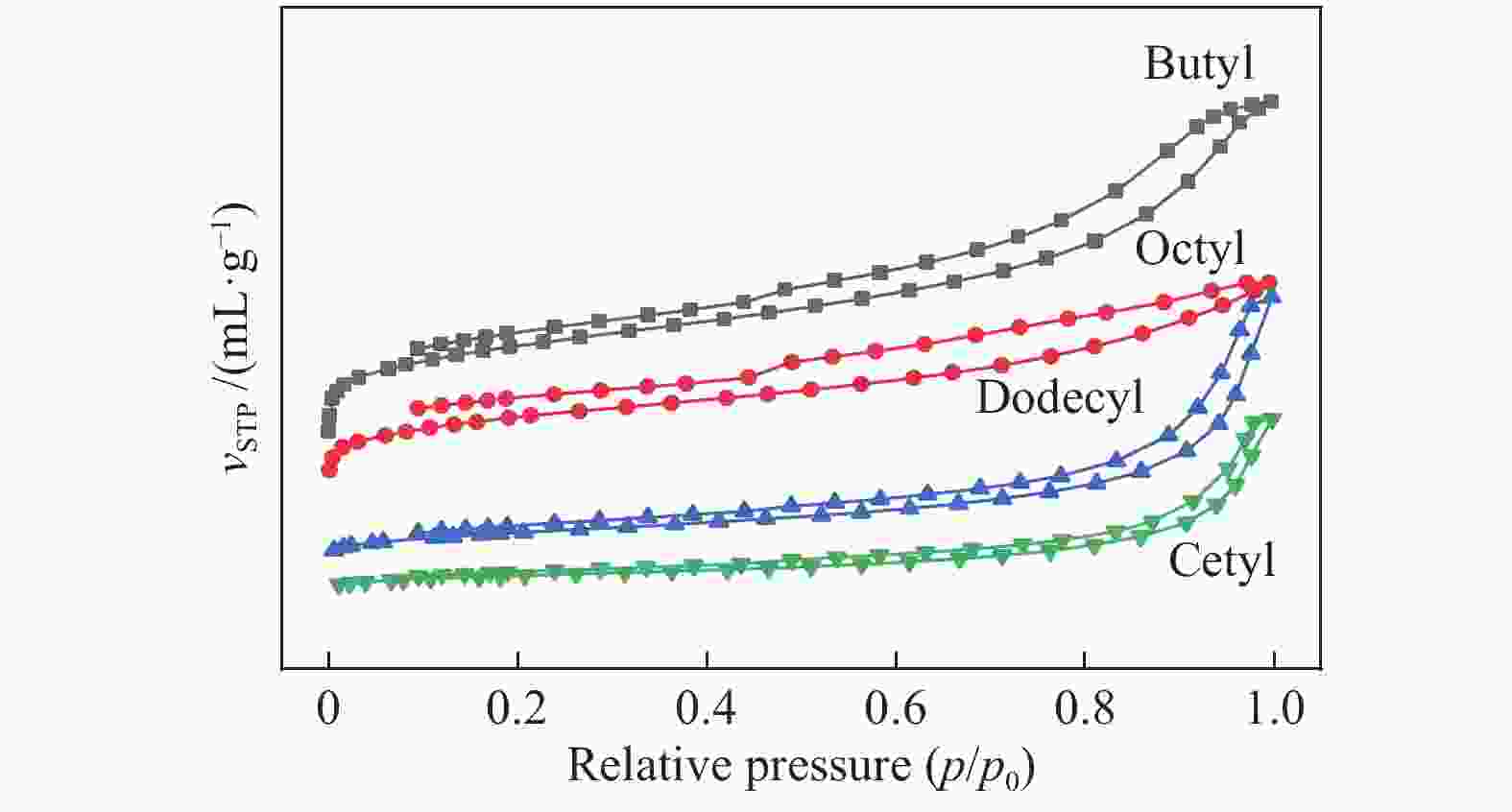
-2-enone0.4 0.6 0.6 − 0.8 1.1 0.5 2.9 0.5 0.8 4 5-methylfurfural 0.2 0.4 1.9 10.9 1.9 1.2 1.0 1.3 1.4 0.8 5 3-methylcyclopent-2-enone 0.3 0.4 1.3 − 1.0 2.8 1.8 2.9 0.6 1.7 6 methyl 2-furoate 0.4 0.4 2.8 − 1.0 1.2 1.6 1.7 1.4 0.5 7 furaneol 0.6 0.7 0.1 − 0.1 0.1 0.1 0.1 − − 8 LGO 6.0 1.4 121.7 107.8 96.5 80.8 96.7 78.2 91.9 85.1 9 LAC 1.9 2.4 5.3 6.6 40.3 21.0 31.7 12.2 4.6 2.6 10 DGP 7.6 5.7 19.8 56.5 17.3 9.2 13.8 11.7 13.4 9.1 11 HMF 1.1 1.2 1.2 − 0.1 − 1.9 − 0.5 − 12 DH − − 0.5 − 0.2 − 0.1 − 0.1 − 13 ADGH 0.9 1.1 1.3 − 1.2 0.9 1.1 0.6 0.9 0.4 14 LGA 211 284 11.1 8.9 9.9 19.5 51.5 18.5 34.5 10.4 15 AGF 14.6 20.1 − − − − − − − − (LAC) 1-hydroxy, (1R)-3,6-dioxabicyclo[3.2.1]octan-2-one, (DGP) 1,4:3,6-Dianhydro-α-D-glucopyranose, (HMF) 5-hydroxymethylfurfural, (DH) 6,8-dioxabicyclo[3.2.1]octane-2,4,4-triol, (ADGH) 1,5-anhydro-4-deoxy-D-glycero-hex-1-en-3-ulose, (AGF) 1,6-Anhydro-β-D-glucofuranose 表 2 焦炭的比表面积、孔容和平均直径
Table 2 Analysis of the specific surface area, pore volume and average diameter of char
Ionic
liquidsSBET/
(m2·g−1)vtotal/
(cm3·g−1)vmicro/
(cm3·g−1)dave Butyl 389.4 0.689 0.067 7.08 Octyl 281.2 0.408 0.045 5.80 Dodecyl 105.3 0.52 − 19.91 Cetyl 58.1 0.340 − 23.40 vmicro was calculated based on t-plot method -
[1] BRUIJNINCX P C A, WECKHUYSEN B M. Lignin up for break-down[J]. Nat Chem,2014,6(12):1035−1036. doi: 10.1038/nchem.2120 [2] HUANG X, REN J, RAN J Y, QIN C L, YANG Z Q, CAO J P. Recent advances in pyrolysis of cellulose to value-added chemicals[J]. Fuel Process Technol,2022,229:107175. doi: 10.1016/j.fuproc.2022.107175 [3] 李承宇, 张军, 袁浩然, 王树荣, 陈勇. 纤维素热解转化研究进展[J]. 燃料化学学报.,2021,49(12):1733−1751. doi: 10.1016/S1872-5813(21)60134-2LI Cheng-yu, ZHANG Jun, YUAN Hao-ran, WANG Shu-rong, CHEN Yong. Advance on the pyrolytic transformation of cellulose[J]. J Fuel Chem Technol,2021,49(12):1733−1751. doi: 10.1016/S1872-5813(21)60134-2 [4] 胡斌, 郭学文, 李洋, 程安帅, 刘吉, 陆强. 山梨醇快速热解特性与反应机理研究[J]. 燃料化学学报,2021,49(12):1821−1831. doi: 10.1016/S1872-5813(21)60150-0HU Bin, GUO Xue-wen, LI Yang, CHENG An-shuai, LIU Ji, LU Qiang. Reaction characteristics and mechanisms of sorbitol fast pyrolysis[J]. J Fuel Chem Technol,2021,49(12):1821−1831. doi: 10.1016/S1872-5813(21)60150-0 [5] HUANG X, KUDO S, SPERRY J. HAYASHI, J. Clean synthesis of 5-hydroxymethylfurfural and levulinic acid by aqueous phase conversion of levoglucosenone over solid acid catalysts[J]. ACS Sustainable Chem Eng,2019,7(6):5892−5899. doi: 10.1021/acssuschemeng.8b05873 [6] ZHONG D, ZENG K, LI J, QIU Y. FLAMANT G, NZIHOU A, VLADIMIROVICH V S, YANG H P, CHEN H P. Characteristics and evolution of heavy components in bio-oil from the pyrolysis of cellulose, hemicellulose and lignin[J]. Renewable Sustainable Energy Rev,2022,157:111989. doi: 10.1016/j.rser.2021.111989 [7] 钱乐, 蒋立群, 岳元茂, 赵增立. 催化热解生物质生成左旋葡聚糖酮的研究进展[J]. 化工学报,2020,71(12):5376−5387.QIAN Le, JIANG Li-qun, YUE Yuan-mao, ZHAO Li-zeng. Research progress of catalytic pyrolysis of biomass to yield levoglucosenone[J]. CIESC J,2020,71(12):5376−5387. [8] KAWAMOTO H, SAITO S, HATANAKA W, SAKA S. Catalytic pyrolysis of cellulose in sulfolane with some acidic catalysts[J]. J Wood Sci,2007,53(2):127−133. doi: 10.1007/s10086-006-0835-y [9] CAO F, SCHWARTZ T J, MCCLELLAND D J. KRISHNA S, DUMESIC J A, HUBER G W. Dehydration of cellulose to levoglucosenone using polar aprotic solvents[J]. Energy Environ Sci,2015,8:1808−1815. doi: 10.1039/C5EE00353A [10] OYOLA-RIVERA O, HE J, HUBER G W, DUMESIC J A, CARDONA-MARTÍNEZ N. Catalytic dehydration of levoglucosan to levoglucosenone using Brønsted solid acid catalysts in tetrahydrofuran[J]. Green Chem,2019,21(18):4988−4999. doi: 10.1039/C9GC01526D [11] COURT G R, LAWRENCE C H, RAVERTY W D, DUNCAN A J. Method for converting lignocellulosic materials into useful chemicals: U. S. Patent 9, 505, 985[P]. 2016–11–29. [12] DOBELE G, ROSSINSKAJA G, TELYSHEVA G, MEIER D, FAIX O. Cellulose dehydration and depolymerization reactions during pyrolysis in the presence of phosphoric acid[J]. J Anal Appl Pyrolysis,1999,49(1/2):307−317. doi: 10.1016/S0165-2370(98)00126-0 [13] DOBELE G, DIZHBITE T, ROSSINSKAJA G, TELYSHEVA G, MEIER D, RADTKE S, FAIX O. Pre-treatment of biomass with phosphoric acid prior to fast pyrolysis: a promising method for obtaining 1, 6-anhydrosaccharides in high yields[J]. J Anal Appl Pyrolysis,2003,68:197−211. [14] ZHANG H, MENG X, LIU C, WANG Y, XIAO R. Selective low-temperature pyrolysis of microcrystalline cellulose to produce levoglucosan and levoglucosenone in a fixed bed reactor[J]. Fuel Process Technol,2017,167:484−490. doi: 10.1016/j.fuproc.2017.08.007 [15] YE X, LU Q, WANG X, GUO H Q, CUI M S, DONG C Q, YANG Y P. Catalytic fast pyrolysis of cellulose and biomass to selectively produce levoglucosenone using activated carbon catalyst[J]. ACS Sustainable Chem Eng,2017,5(11):10815−10825. doi: 10.1021/acssuschemeng.7b02762 [16] FABBRI D, TORRI C, MANCINI I. Pyrolysis of cellulose catalysed by nanopowder metal oxides: Production and characterisation of a chiral hydroxylactone and its role as building block[J]. Green Chem,2007,9(12):1374−1379. doi: 10.1039/b707943e [17] WEI X, WANG Z, WU Y, YU Z, JIN J, WU K. Fast pyrolysis of cellulose with solid acid catalysts for levoglucosenone[J]. J Anal Appl Pyrolysis,2014,107:150−154. doi: 10.1016/j.jaap.2014.02.015 [18] 卫新来, 隋先伟, 俞志敏, 金杰, 吴克, 王文奎. 生物质催化热解制备左旋葡萄糖酮的研究进展[J]. 化工进展,2014,33(4):873−877.WEI Xin-lai, SUI Xian-wei, YU Zhi-min, JIN Jie, WU ke, WANG Wen-kui. Progress in catalytic pyrolysis of biomass for levoglucosenone[J]. Chem Ind Eng Prog,2014,33(4):873−877. [19] WANG Z, LU Q, ZHU X F, ZHANG Y. Catalytic fast pyrolysis of cellulose to prepare levoglucosenone using sulfated zirconia[J]. ChemSusChem,2011,4(1):79−84. doi: 10.1002/cssc.201000210 [20] 陆强, 朱锡锋. 利用固体超强酸催化热解纤维素制备左旋葡萄糖酮[J]. 燃料化学学报,2011,39(6):425−431.LU Qiang ZHU Xi-Feng. Production of levoglucosenone from pyrolysis of cellulose catalyzed by solid superacids[J]. J Fuel Chem Technol,2011,39(6):425−431. [21] KUDO S, HUANG X, ASANO S, HAYASHI J. Catalytic strategies for levoglucosenone production by pyrolysis of cellulose and lignocellulosic biomass[J]. Energy Fuels,2021,35(12):9809−9824. doi: 10.1021/acs.energyfuels.1c01062 [22] KUDO S, ZHOU Z, YAMASAKI K, NORINAGA K, HAYASHI J. Sulfonate ionic liquid as a stable and active catalyst for levoglucosenone production from saccharides via catalytic pyrolysis[J]. Catalysts,2013,3(4):757−773. doi: 10.3390/catal3040757 [23] KUDO S, ZHOU Z, NORINAGA K, HAYASHI J. Efficient levoglucosenone production by catalytic pyrolysis of cellulose mixed with ionic liquid[J]. Green Chem,2011,13(11):3306−3311. doi: 10.1039/c1gc15975e [24] KUDO S, GOTO N, SPERRY J, NORINAGA K, HAYASHI J. Production of levoglucosenone and dihydrolevoglucosenone by catalytic reforming of volatiles from cellulose pyrolysis using supported ionic liquid phase[J]. ACS Sustainable Chem Eng,2017,5(1):1132−1140. doi: 10.1021/acssuschemeng.6b02463 [25] SUN N, RODRIGUEZ H, RAHMAN M, ROGERS, R. D. Where are ionic liquid strategies most suited in the pursuit of chemicals and energy from lignocellulosic biomass?[J]. Chem Commun,2011,47(5):1405−1421. doi: 10.1039/C0CC03990J [26] HUANG X, CAO J P, SHI P, ZHAO X Y, FENG X B, ZHAO Y P, FAN X, WEI X Y, TAKARADA T. Influences of pyrolysis conditions in the production and chemical composition of the bio-oils from fast pyrolysis of sewage sludge[J]. J Anal Appl Pyrolysis,2014,110:353−362. doi: 10.1016/j.jaap.2014.10.003 [27] LU T, CHEN F. Revealing the nature of intermolecular interaction and configurational preference of the nonpolar molecular dimers (H2)2, (N2)2, and (H2)(N2)[J]. J Mol Model,2013,19(12):5387−5395. doi: 10.1007/s00894-013-2034-2 [28] WITTE J, GOLDEY M, NEATON J B, HEAD-GORDON M. Beyond energies: Geometries of nonbonded molecular complexes as metrics for assessing electronic structure approaches[J]. J Chem Theory Comput,2015,11(4):1481−1492. doi: 10.1021/ct501050s [29] FENG S, VOTH G A. Molecular dynamics simulations of imidazolium-based ionic liquid/water mixtures: alkyl side chain length and anion effects[J]. Fluid Ph Equilibria,2010,294(1/2):148−156. doi: 10.1016/j.fluid.2010.02.034 [30] WANG Y, VOTH G A. Tail aggregation and domain diffusion in ionic liquids[J]. J Phys Chem B,2006,110(37):18601−18608. doi: 10.1021/jp063199w [31] YANG X, ZHAO Y, LI R, WU Y, YANG M. A modified kinetic analysis method of cellulose pyrolysis based on TG-FTIR technique[J]. Thermochim Acta,2018,665:20−27. doi: 10.1016/j.tca.2018.05.008 [32] ZHUO K, DU Q, BAI G, WANG C, CHEN Y, WANG J. Hydrolysis of cellulose catalyzed by novel acidic ionic liquids[J]. Carbohydr Polym,2015,115:49−53. doi: 10.1016/j.carbpol.2014.08.078 [33] HUANG X, CAO J P, ZHAO X Y, WANG J X, FAN X, ZHAO Y P, WEI X Y. Pyrolysis kinetics of soybean straw using thermogravimetric analysis[J]. Fuel,2016,169:93−98. doi: 10.1016/j.fuel.2015.12.011 [34] HSU T C, GUO G L, CHEN W H, HWANG W S. Effect of dilute acid pretreatment of rice straw on structural properties and enzymatic hydrolysis[J]. Bioresour Technol,2010,101(13):4907−4913. doi: 10.1016/j.biortech.2009.10.009 [35] KRISHNA S H, WALKER T W, DUMESIC J A, HUBER, G W. Kinetics of levoglucosenone isomerization[J]. ChemSusChem,2016,10(1):129−138. [36] HU B, LU Q, WU Y, XIE W L, CUI M S, LIU J, DONG C Q, YANG Y P. Insight into the formation mechanism of levoglucosenone in phosphoric acid-catalyzed fast pyrolysis of cellulose[J]. J Energy Chem,2020,43:78−89. doi: 10.1016/j.jechem.2019.08.001 -




 下载:
下载: