Study on the mercury removal performance and strengthening method of high sulfur and iron content textile dyeing sludge char
-
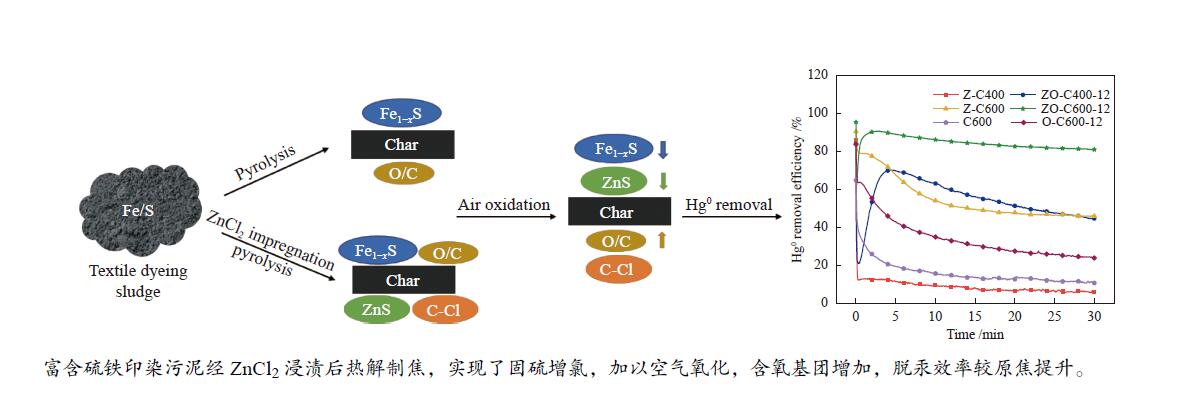
摘要: 以高硫高铁印染污泥为原料制备热解焦,对热解前后样品中的硫和铁结合态进行了分析,并研究了热解焦对单质汞的脱除特性,进一步的,通过空气氧化、ZnCl2浸渍等手段对热解焦进行改性,提升其脱汞性能。研究结果表明,污泥中的硫主要为硫酸盐、硫化物、有机硫三部分,铁以三价和二价化合物存在。热解后,无机硫向有机硫转化,三价铁向二价铁转化,大部分硫和铁保留在热解焦中,部分生成磁黄铁矿 (Fe1−xS)。污泥原焦比表面积较小,具有一定的脱汞能力,以化学吸附为主导。空气氧化时间控制在12 h以内可以使高温(≥ 600 ℃)热解焦的汞吸附量提升46%以上。ZnCl2浸渍污泥热解制焦,可以进一步固硫生成ZnS,600 ℃热解的ZnCl2改性焦30 min内汞吸附量达到了28.71 μg/g,加以空气氧化,脱汞效率进一步提升,氧化12 h后性能最佳,汞吸附量为43.75 μg/g。Abstract: Pyrolysis char was prepared from high sulfur and iron content textile dyeing sludge. The combined states of S and Fe in the samples before and after pyrolysis and the removal characteristics of Hg0 by pyrolysis char were studied. The performance of Hg0 removal was improved by air oxidation and ZnCl2 impregnation. The results showed that S in sludge was divided into sulfate, sulfide, and organic sulfur. Fe existed as Fe3+ and Fe2+ compounds. After pyrolysis, inorganic sulfur was transferred to organic sulfur and Fe3+ was transferred to Fe2+. Most S and Fe were retained in pyrolysis char and some formed pyrrhotite (Fe1−xS). The specific surface area of raw char was small and had a certain Hg0 removal capacity, dominated by chemical adsorption. When the air oxidation time was controlled within 12 h, the Hg0 adsorption capacity of pyrolysis char at high temperature (≥600 ℃) was increased by more than 46%. During pyrolysis of ZnCl2 impregnated sludge, more S was fixed in pyrolysis char to generate ZnS. The Hg0 adsorption capacity of ZnCl2 modified char pyrolyzed at 600 ℃ reached 28.71 μg/g in 30 min. With air oxidation, the Hg0 removal efficiency was further improved. After oxidation for 12 h, the Hg0 adsorption capacity was 43.75 μg/g.
-
Key words:
- textile dyeing sludge /
- pyrolysis char /
- demercuration /
- adsorption
-
表 1 污泥及热解焦特性
Table 1 Characteristics of sludge and pyrolysis char
Sample Char yield/% Ultimate analysis w/% Ash wd/% XRF analysis of ash w/% R(S) R(Fe) S/C C S N H Fe2O3 SO3 ZnO Cl C000 − 32.22 10.19 5.15 4.34 32.93 42.30 16.38 1.41 ND 12.35 9.75 0.38 C400 66.33 31.90 13.47 5.47 2.40 43.97 51.36 14.35 2.16 ND 16.00 15.81 0.50 C500 61.92 32.92 13.75 5.22 1.96 48.60 49.92 16.24 2.38 ND 16.91 16.98 0.51 C600 57.25 33.98 14.10 4.60 1.66 52.57 56.95 15.42 2.24 ND 17.34 20.96 0.51 C700 52.93 33.58 14.54 4.10 1.41 55.54 52.59 15.34 2.48 ND 17.95 20.45 0.53 Z-C600 72.89 17.76 7.23 2.89 1.59 59.41 27.73 16.11 28.42 16.40 11.06 11.53 0.62 d, dry base; ND, not detected; P(X), contents of components (Table 1); R(S), the relative content of S in char, R(S)=0.4×P(SO3)×P(Ash)+P(S), 0.4 is the proportion of S in SO3 molecular weight; R(Fe), the relative content of Fe in char, R(Fe)=0.7×P(Fe2O3)×P(Ash), 0.7 is the proportion of Fe in the molecular weight of Fe2O3; S/C=[R(S)]/P(C) 表 2 不同热解焦孔隙结构
Table 2 Pore structure of different pyrolysis char
Sample SBET/
(m2·g−1)Pore volume/
(10−2 cm3·g−1)Average pore
width/nmC000 3.95 2.40 24.33 C400 12.57 4.62 14.69 C500 18.49 5.26 11.38 C600 28.16 6.55 9.29 C700 17.60 5.43 12.33 Z-C400 1.47 0.87 23.40 Z-C600 7.98 3.34 16.73 SBET: specific surface area 表 3 样品热解前后不同结合态S、Fe相对含量
Table 3 Relative contents of different combined-form S and Fe before and after pyrolysis
Combined-form Before pyrolysis/% After pyrolysis/% Sulfides 55.28 30.01 Organic sulfur 35.88 59.59 Sulfates 8.84 10.40 Fe(Ш) 46.51 36.78 Fe(Ⅱ) 53.49 63.21 -
[1] RAN C, LIU Y, SIDDIQUI A R, SIYAL A A, MAO X, KANG Q, FU J, AO W, DAI J. Pyrolysis of textile dyeing sludge in fluidized bed and microwave- assisted auger reactor: Comparison, migration and distribution of heavy metals[J]. Energy,2019,182:337−348. doi: 10.1016/j.energy.2019.05.219 [2] WANG M, MAO M, ZHANG M, WEN G, YANG Q, SU B, REN Q. Highly efficient treatment of textile dyeing sludge by CO2 thermal plasma gasification[J]. Waste Management,2019,90:29−36. doi: 10.1016/j.wasman.2019.04.025 [3] 邓昌亚. 不同种类污泥与煤混合燃烧的灰熔融特性研究[D]. 武汉: 华中科技大学, 2016.DENG Chang-ya. The melting characteristics during co-combustion of coal with different sludge[D]. Wuhan: Huazhong University of Science and Technology, 2016. [4] BRIDLE T, SKRYPSKI-MANTELE D S. Assessment of sludge reuse options: A life-cycle approach[J]. Water Sci Technol,2000,41(8):131−135. doi: 10.2166/wst.2000.0152 [5] HORTTANAINEN M, KAIKKO J, BERGMAN R, PASILA-LEHTINEN M, NERG J. Performance analysis of power generating sludge combustion plant and comparison against other sludge treatment technologies[J]. Appl Therm Eng,2011,30(2/3):110−118. [6] 李高磊, 郭沂权, 张世博, 王洪跃, 朱彬彬, 常林, 赵永椿, 张军营. 超低排放燃煤电厂SO3生成及控制的试验研究[J]. 中国电机工程学报,2019,39(4):1079−1086.LI Gao-lei, GUO Yi-quan, ZHANG Shi-bo, WANG Hong-yue, ZHU Bin-bin, CHANG Lin, ZHAO Yong-chun, ZHANG Jun-ying. Experimental research on SO3 generation and control in ultra-low emission coal-fired power plant[J]. Proc CSEE,2019,39(4):1079−1086. [7] 孙东晓, 董志强, 刘学明, 张吉琛, 陈钱宝, 雷思聪. 污泥基生物炭的制备技术及环境应用与研究热点[J]. 净水技术,2021,40(8):16−25.SUN Dong-xiao, DONG Zhi-qiang, LIU Xue-ming, ZHANG Ji-chen, CHEN Qian-bao, LEI Si-cong. Research hotspot and preparation technology and environmental application for sludge Biochar[J]. Water Purif Technol,2021,40(8):16−25. [8] RICE K M, WALKER E M, WU M, GILLETTE C, BLOUGH E R. Environmental mercury and its toxic effects[J]. J Prev Med Pub Health,2014,47(2):74−83. doi: 10.3961/jpmph.2014.47.2.74 [9] YANG J, ZHAO Y, MA S, ZHU B, ZHANG J, ZHENG C. Mercury removal by magnetic biochar derived from simultaneous activation and magnetization of sawdust[J]. Environ Sci Technol,2016,50(21):12040−12047. doi: 10.1021/acs.est.6b03743 [10] ZHAO S, PUDASAINEE D, DUAN Y, GUPTA R, LIU M, LU J. A review on mercury in coal combustion process: content and occurrence forms in coal, transformation, sampling methods, emission and control technologies[J]. Prog Energy Combust Sci,2019,73:26−64. doi: 10.1016/j.pecs.2019.02.001 [11] WEN X, HUSSAIN A, LIU Y. A review on modification methods of adsorbents for elemental mercury from flue gas[J]. Chem Eng J,2018,346:692−711. doi: 10.1016/j.cej.2018.03.049 [12] 洪亚光, 段钰锋, 朱纯, 周强, 佘敏, 杜鸿飞. 硫改性椰壳活性炭管道喷射脱汞实验研究[J]. 东南大学学报(自然科学版),2015,45(3):521−525.HONG Ya-guang, DUAN Yu-feng, ZHU Chun, ZHOU Qiang, SHE Min, DU Hong-fei. Experimental study on mercury adsorption of S-impregnated coconut shell activated carbon by duct injection[J]. J Southeast Univ: Nat Sci Ed,2015,45(3):521−525. [13] REDDY K, SHOAIBI A A, SRINIVASAKANNAN C. Mercury removal using metal sulfide porous carbon complex[J]. Process Saf Environ Prot,2018,114:153−158. doi: 10.1016/j.psep.2017.12.022 [14] BHARDWAJ R, CHEN X, VIDIC R D. Impact of fly ash composition on mercury speciation in simulated flue gas[J]. Air Repair,2009,59(11):1331−1338. [15] ZHANG J L, WANG Z Y, XING X D, LIU Z J. Effect of aluminum oxide on the compressive strength of pellets[J]. Int J Min Met Mater,2014,21(4):339−344. doi: 10.1007/s12613-014-0914-9 [16] WANG Y, SHEN B, HE C, YUE S, WANG F. Simultaneous removal of NO and Hg0 from flue gas over Mn-Ce/Ti-PILCs[J]. Environ Sci Technol,2015,49:9355−9363. doi: 10.1021/acs.est.5b01435 [17] YANG J, ZHAO Y, ZHANG J, ZHENG C. Regenerable cobalt oxide loaded magnetosphere catalyst from fly ash for mercury removal in coal combustion flue gas[J]. Environ Sci Technol,2014,48(24):14837−14843. doi: 10.1021/es504419v [18] 卢龙, 王汝成, 薛纪越, 陈骏, 陆建军. 硫化物矿物的表面反应及其在矿山环境研究中的应用[J]. 岩石矿物学杂志,2001,20(4):387−394. doi: 10.3969/j.issn.1000-6524.2001.04.004LU Long, WANG Ru-cheng, XUE Ji-yue, CHEN Jun, LU Jian-jun. The surface reaction of sulfide minerals and its application to the study of mine environment[J]. Acta Petrol Mineral,2001,20(4):387−394. doi: 10.3969/j.issn.1000-6524.2001.04.004 [19] TAN Z, SUN L, XIANG J, ZENG H, LIU Z, SONG H, QIU J. Gas-phase elemental mercury removal by novel carbon-based sorbents[J]. Carbon,2012,50(2):362−371. doi: 10.1016/j.carbon.2011.08.036 [20] WANG H, ZHANG L, DENG T, RUAN H, HOU X, CORT R J, YANG B. ZnCl2 induced catalytic conversion of softwood lignin to aromatics and hydrocarbons[J]. Green Chem,2016,18(9):2802−2810. doi: 10.1039/C5GC02967H [21] XIA M, SHAO X, SUN Z, XU Z. Conversion of cotton textile wastes into porous carbons by chemical activation with ZnCl2, H3PO4, and FeCl3[J]. Environ Sci Pollut Res Int,2020,27(20):25186−25196. doi: 10.1007/s11356-020-08873-3 [22] LI L, LI F. Preparation of carbonaceous adsorbent from straw and its adsorption performance for H2S removal[J]. J Air Waste Manage Assoc,2020,70(6):649−656. doi: 10.1080/10962247.2020.1754306 [23] SONG Y, HU J, LIU J, EVRENDILEK F, BUYUKADA M. CO2-assisted co-pyrolysis of textile dyeing sludge and hyperaccumulator biomass: Dynamic and comparative analyses of evolved gases, bio-oils, biochars, and reaction mechanisms[J]. J Hazard Mater,2020,400:123190. doi: 10.1016/j.jhazmat.2020.123190 [24] LIU J, ZHUO Z, XIE W, KUO J, LU X, BUYUKADA M, EVRENDILEK F. Interaction effects of chlorine and phosphorus on thermochemical behaviors of heavy metals during incineration of sulfur-rich textile dyeing sludge[J]. Chem Eng J,2018,351:897−911. doi: 10.1016/j.cej.2018.06.158 [25] AHMAD M, RAJAPAKSHA A U, LIM J E, MING Z, BOLAN N, MOHAN D, VITHANAGE M, SANG S L, YONG S O. Biochar as a sorbent for contaminant management in soil and water: A review[J]. Chemosphere,2014,99:19−33. doi: 10.1016/j.chemosphere.2013.10.071 [26] YUAN H, TAO L, HUANG H, ZHAO D, KOBAYASHI N, YONG C. Influence of pyrolysis temperature on physical and chemical properties of biochar made from sewage sludge[J]. J Anal Appl Pyrolysis,2015,112:284−289. doi: 10.1016/j.jaap.2015.01.010 [27] 胡二峰, 赵立欣, 吴娟, 孟海波, 姚宗路, 丛宏斌, 吴雨浓. 生物质热解影响因素及技术研究进展[J]. 农业工程学报,2018,34(14):212−220. doi: 10.11975/j.issn.1002-6819.2018.14.027HU Er-feng, ZHAO Li-xin, WU Juan, MENG Hai-bo, YAO Zong-lu, CONG Hong-bin, WU Yu-nong. Research advance on influence factors and technologies of biomass pyrolysis[J]. Trans Chin Soc Agric Eng,2018,34(14):212−220. doi: 10.11975/j.issn.1002-6819.2018.14.027 [28] TANG H, XU M, HU H, YANG F, YANG Y, LIU H, LI X, YAO H. In-situ removal of sulfur from high sulfur solid waste during molten salt pyrolysis[J]. Fuel,2018,231:489−494. doi: 10.1016/j.fuel.2018.05.123 [29] ZHANG D, YANI S. Sulphur transformation during pyrolysis of an australian lignite[J]. Proc Combust Inst,2011,33(2):1747−1753. doi: 10.1016/j.proci.2010.07.074 [30] 王彦, 左宁, 姜媛媛, 陈芳媛. 污泥生物炭中氮硫行为及环境效应研究进展[J]. 化工进展,2020,39(4):1539−1549.WANG Yan, ZUO Ning, JIANG Yuan-yuan, CHEN Fang-yuan. Behavior and environmental effects of nitrogen and sulfur in sludge biochar[J]. Chem Ind Eng Prog,2020,39(4):1539−1549. [31] YAMASHITA T, HAYES P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials[J]. Appl Surf Sci,2008,254(8):2441−2449. doi: 10.1016/j.apsusc.2007.09.063 [32] LIKOSOVA E M, KELLER J, POUSSADE Y, FREGUIA S. A novel electrochemical process for the recovery and recycling of ferric chloride from precipitation sludge[J]. Water Res,2014,51:96−103. doi: 10.1016/j.watres.2013.12.020 [33] 戎宇舟, 葛强, 李清, 吴伏安, 杨树成, 齐随涛. 制浆造纸厂富铁污泥性质及其回用为污泥调理剂研究[J]. 西安交通大学学报,2016,50(9):43−48. doi: 10.7652/xjtuxb201609007RONG Yu-zhou, GE Qiang, LI Qing, WU Fu-an, YANG Shu-cheng, QI Sui-tao. Study on properties of iron-rich sludge in pulp and paper industry and its recycling as sludge conditioning agent[J]. J Xi'an Jiaotong Univ,2016,50(9):43−48. doi: 10.7652/xjtuxb201609007 [34] 郭慧卿. 基于同步辐射与模拟计算研究煤热解过程中硫的迁移行为[D]. 内蒙古: 内蒙古大学, 2021.GUO Hui-qing. The study of sulfur transformation behavior during coal pyrolysis by synchrotron radiation combined with simulation calculation[D]. Inner Mongolia: Inner Mongolia University, 2021. [35] ZHANG Y, LI Q, LIU X, XU B, YANG Y, JIANG T. A thermodynamic analysis on the roasting of pyrite[J]. Minerals,2019,9(4):220−220. doi: 10.3390/min9040220 [36] 廖勇. 磁性可循环吸附剂集中控制燃煤烟气零价汞污染的研究[D]. 南京: 南京理工大学, 2017.LIAO Yong. Centralized control of elemental mercury in fuel gas by magnetic recyclable sorbents[D]. Nanjing: Nanjing University of Science and Technology, 2017. [37] 李娜, 韦红旗, 段钰锋, 王晨平, 佘敏, 胡鹏, 王双群. SO2与O2对硫改性石油焦吸附剂脱汞性能的影响[J]. 化工进展,2018,37(5):1908−1915.LI Na, WEI Hong-qi, DUAN Yu-feng, WANG Chen-ping, SHE Min, HU Peng, WANG Shuang-qun. Effects of SO2 and O2 on Hg0 removal capacity of sulfur modified petrol coke adsorbents[J]. Chem Ind Eng Prog,2018,37(5):1908−1915. [38] TU Z, GUO C, ZHANG T, LU G, WAN J, LIAO C, DANG Z. Investigation of intermediate sulfur species during pyrite oxidation in the presence and absence of Acidithiobacillus ferrooxidans[J]. Hydrometallurgy,2017,167:58−65. doi: 10.1016/j.hydromet.2016.11.001 [39] 王志豪. 间二甲苯氧化制间苯二甲酸反应中催化剂结构与性能的关系研究[D]. 上海: 华东理工大学, 2017.WANG Zhi-hao. Relationship between structure and performance of catalyst in m-xylene oxidized to isophthalic acid[D]. Shanghai: East China University of Science and Technology, 2017. [40] LI H, ZHANG M, ZHU L, YANG J. Stability of mercury on a novel mineral sulfide sorbent used for efficient mercury removal from coal combustion flue gas[J]. Environ Sci Pollut Res,2018,25(28):28583−28593. doi: 10.1007/s11356-018-2896-z -





 下载:
下载: