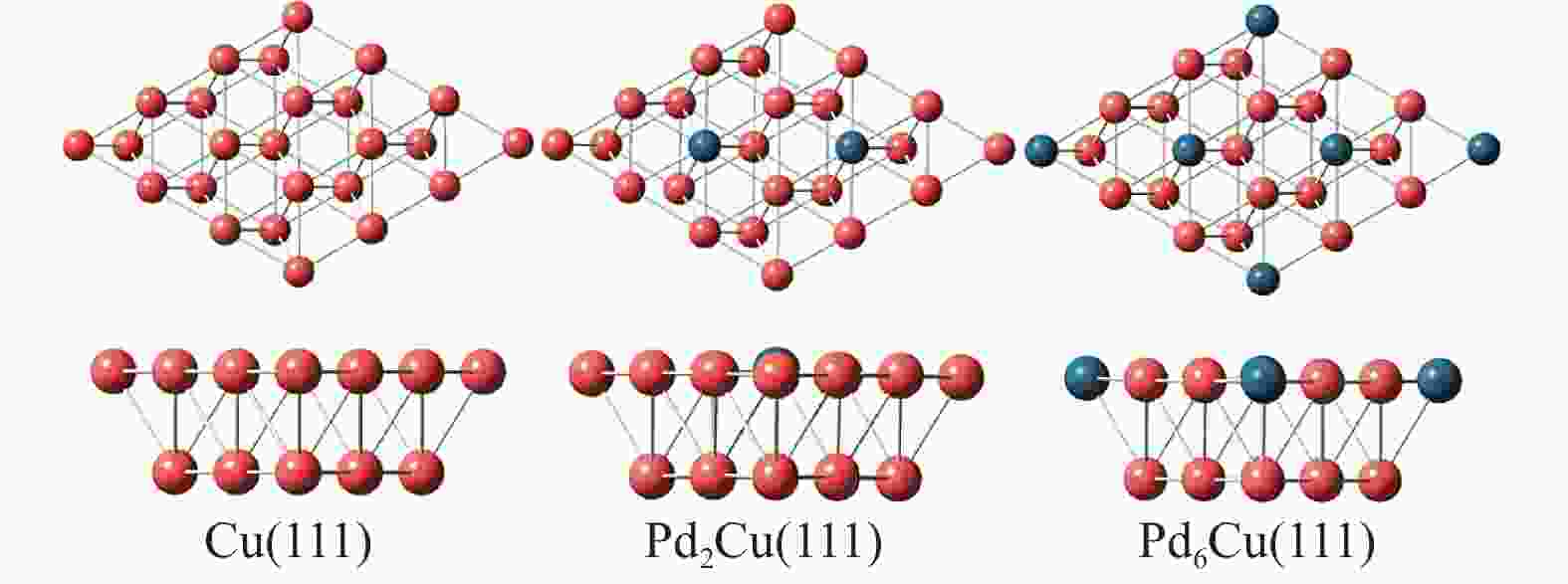
Study on the activation mechanism of O-enhanced methane adsorbed on Pd-Cu catalyst
-
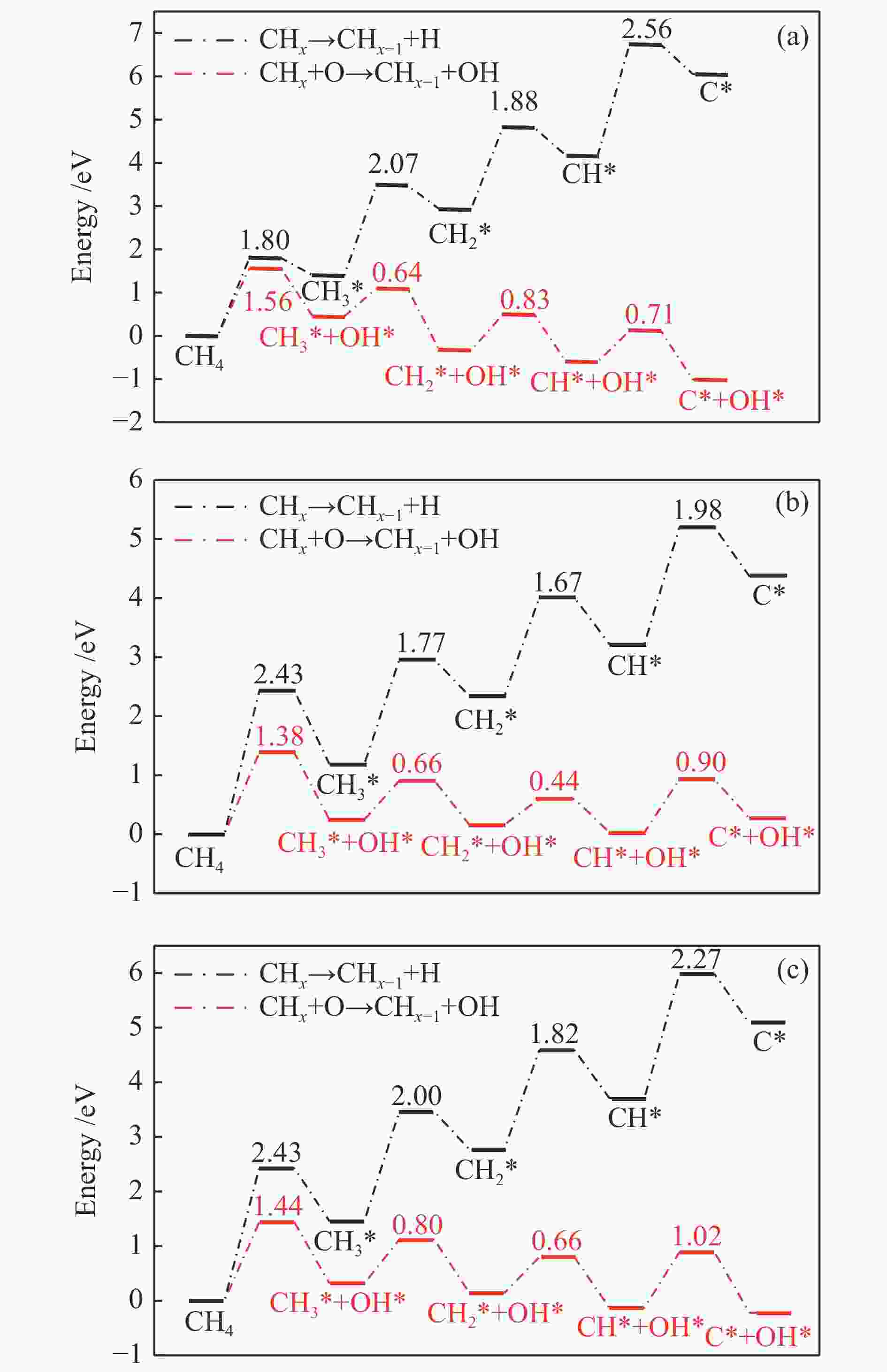
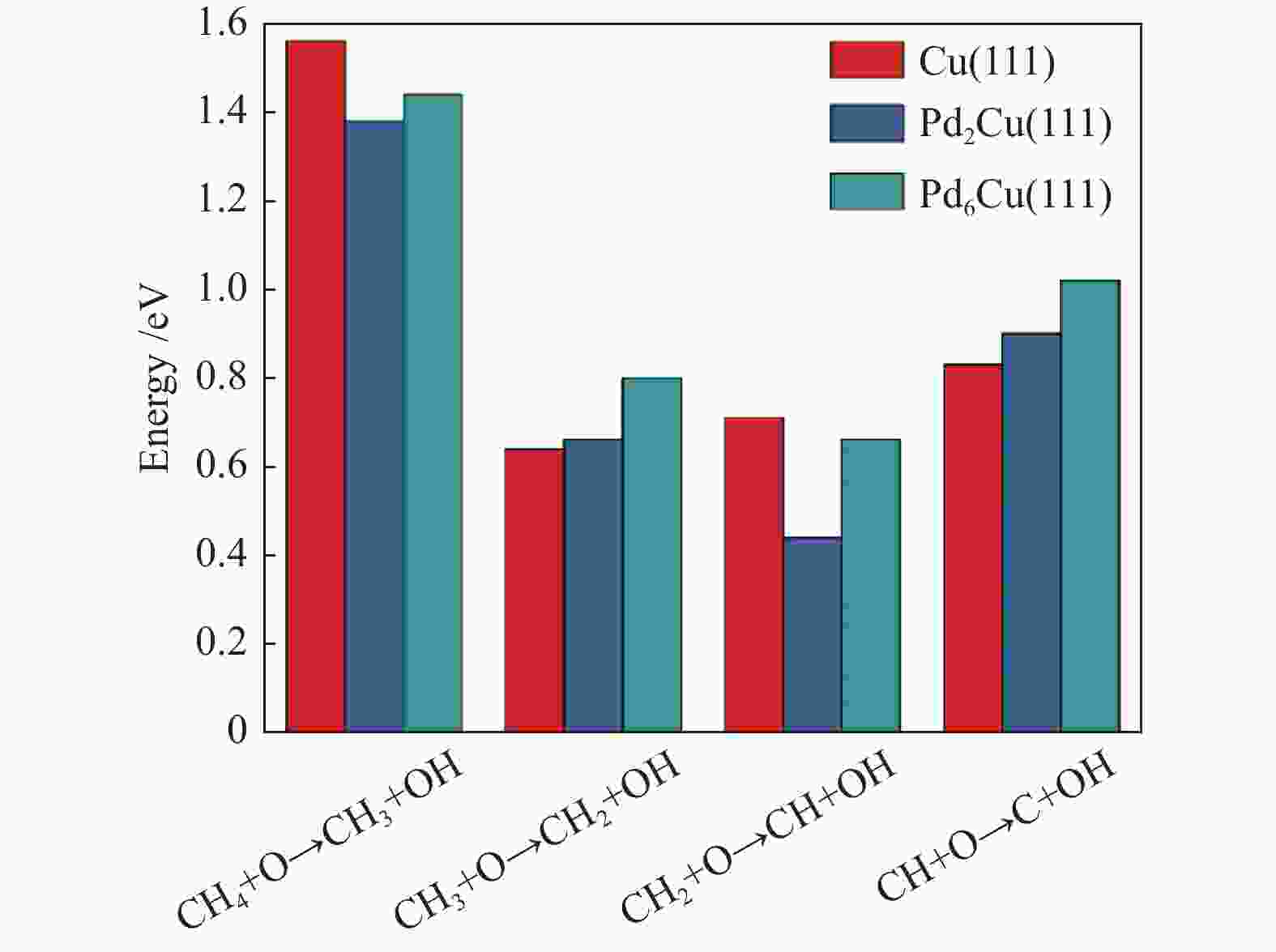
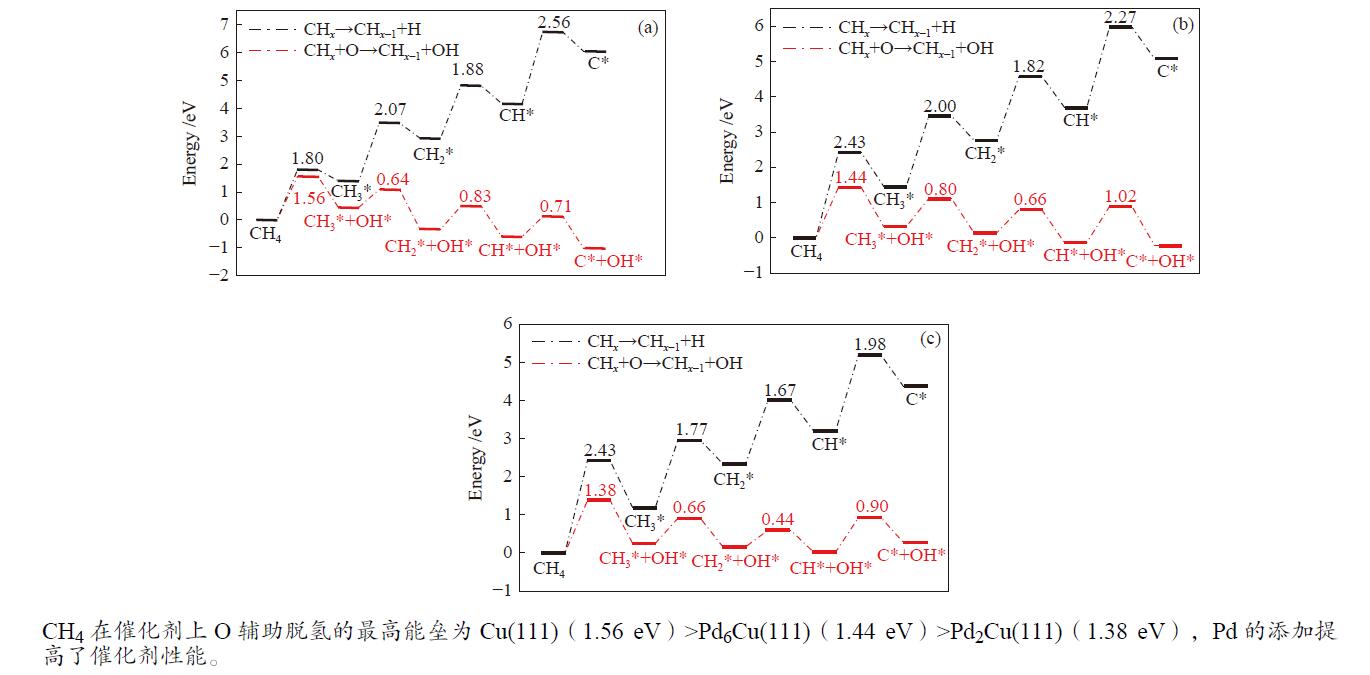
摘要: 甲烷催化燃烧相比较传统燃烧有燃烧温度低,清洁以及高效的优点,在天然气汽车、固体氧化物燃料电池等多个领域具有较好的应用前景。为了揭示甲烷在不同掺杂比的Pd-Cu团簇上的脱氢机理,本研究采用密度泛函理论(DFT)对CH4*在不同团簇上的直接脱氢和O辅助脱氢进行计算。计算结果表明,Pd原子的掺杂提高了Cu(111)表面的吸附能力,在直接脱氢过程中,Pd的掺杂不仅使能垒由2.56 eV降低到2.43 eV,而且使速率控制步骤由CH*+*→C* + H*变为CH4*+*→CH3* + H*。预吸附O能够显著降低甲烷脱氢的能垒,速率控制步骤均为CH4* + O*→CH3* + OH*,甲烷在团簇上O辅助脱氢的最高能垒的大小为Cu(111)(1.56 eV)>Pd6Cu(111)(1.44 eV)>Pd2Cu(111)(1.38 eV),Pd的添加对于直接脱氢和O辅助脱氢的性能都有所提升。Abstract: Compared with traditional combustion, methane catalytic combustion has the advantages of low combustion temperature, clean and high efficiency, and it has good application prospects in natural gas vehicles, solid oxide fuel cell and other fields. In order to reveal the mechanism of dehydrogenation of methane on Pd-Cu clusters with different doping ratios, the density functional theory (DFT) is used to calculate the direct dehydrogenation and O-assisted dehydrogenation of CH4* in different clusters. The calculation results show that the doping of Pd atoms increases the adsorption capacity of Cu(111) surface, and in the process of direct dehydrogenation, the doping of Pd not only reduces the energy barrier from 2.56 to 2.43 eV, but also changes the rate determining step from CH*+*→C* + H* to CH4*+*→CH3* + H*. Pre-adsorbed O can significantly reduce the energy barrier of methane dehydrogenation, and the rate determining steps are CH4* + O*→CH3* + OH*. The highest energy barrier of O-assisted dehydrogenation of CH4* is Cu(111)(1.56 eV)>Pd6Cu(111)(1.44 eV)>Pd2Cu(111)(1.38 eV) on three clusters, which indicates that the addition of Pd has improved the performance of direct dehydrogenation and O-assisted dehydrogenation.
-
Key words:
- CH4 activation /
- Pd-Cu catalyst /
- direct dehydrogenation /
- O-assisted dehydrogenation
-
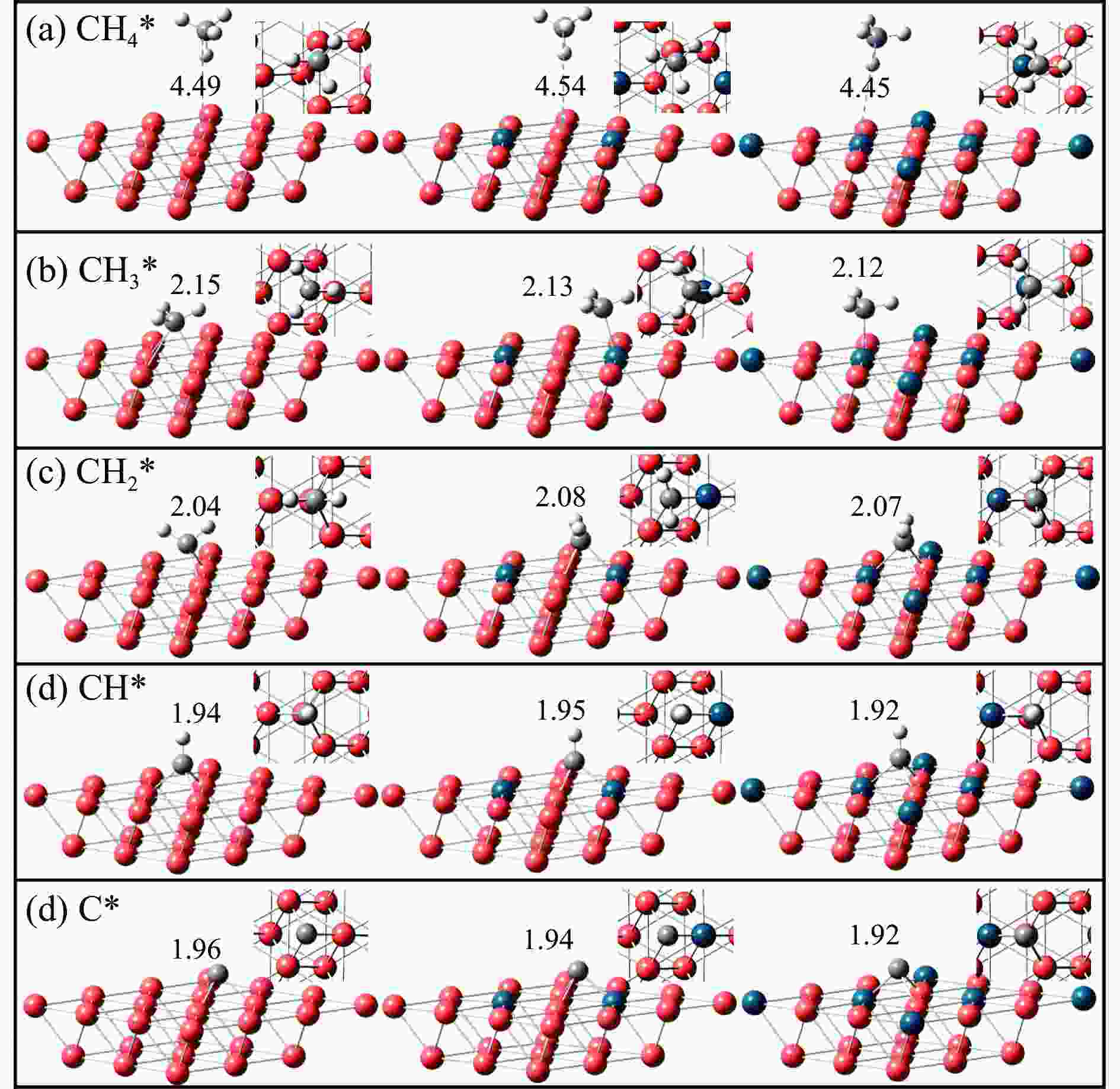
表 1 CHx*(x=0–4)在三种催化剂表面的吸附位点和吸附能(eV)
Table 1 Adsorption sites and adsorption energy (eV) of CHx*(x=0–4) over the surface of three catalysts
Species Cu(111) Pd2Cu(111) Pd6Cu(111) site energy site energy site energy CH4* top −0.02 top −0.03 top −0.02 CH3* hollow −0.98 top −1.41 top −1.12 CH2* bridge −2.79 hollow −3.13 hollow −2.93 CH* hollow −4.08 hollow −4.65 hollow −4.51 C* hollow −4.54 hollow −5.11 hollow −5.17 表 2 甲烷在三种催化剂上直接脱氢的能垒和反应热(eV)
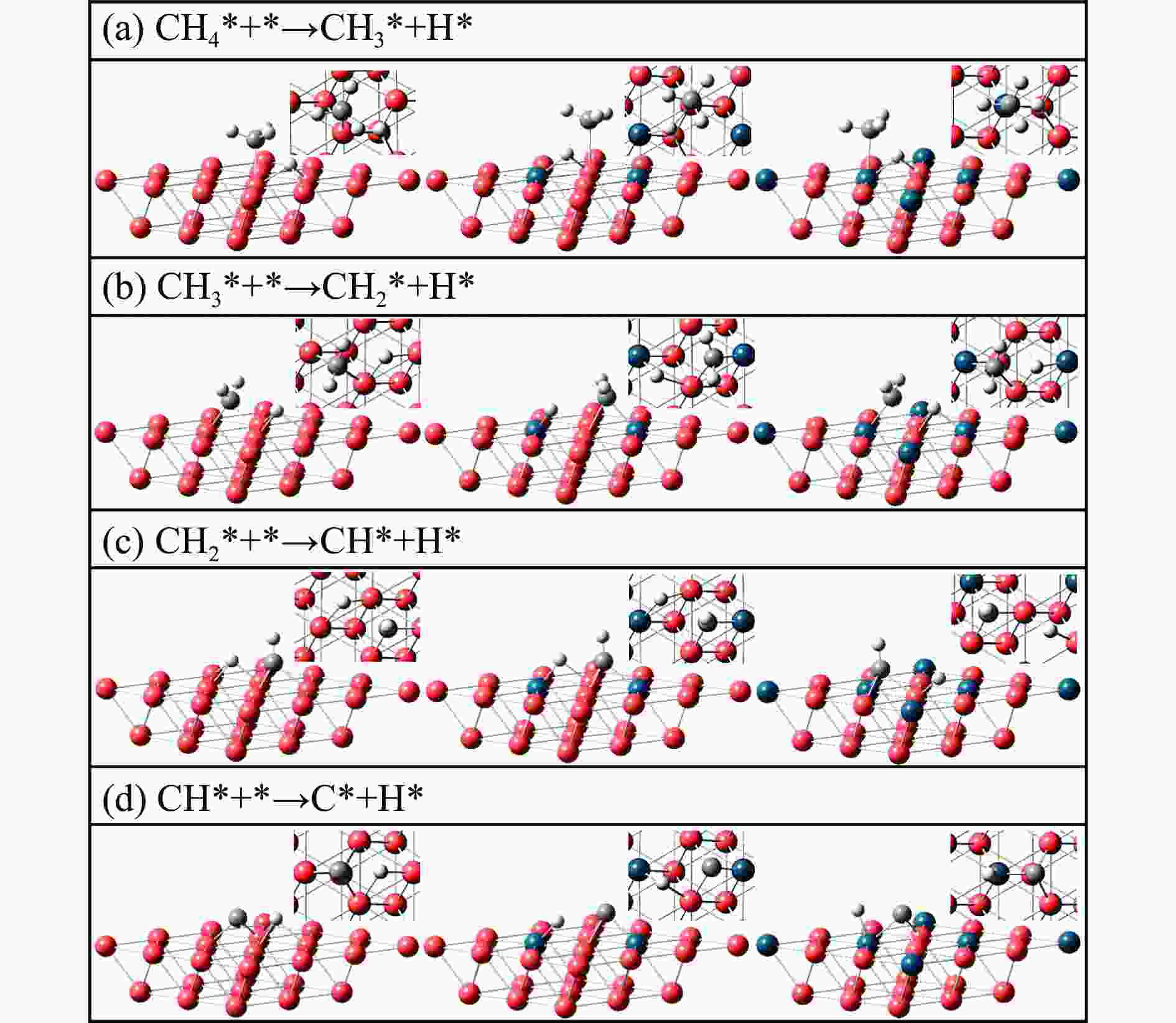
Table 2 Energy barrier and heat of methane direct dehydrogenation on three catalysts (eV)
Elementary step Cu(111) Pd2Cu(111) Pd6Cu(111) barrier heat barrier heat barrier heat CH4*+*→CH3* + H* 1.80 1.40 2.43 1.19 2.43 1.46 CH3*+*→CH2* + H* 2.07 1.53 1.77 1.15 2.00 1.31 CH2*+*→CH* + H* 1.88 1.24 1.67 0.88 1.82 0.94 CH*+*→C* + H* 2.56 1.88 1.98 1.15 2.27 1.37 表 3 O辅助甲烷脱氢在三种催化剂表面上的能垒和反应热(eV)
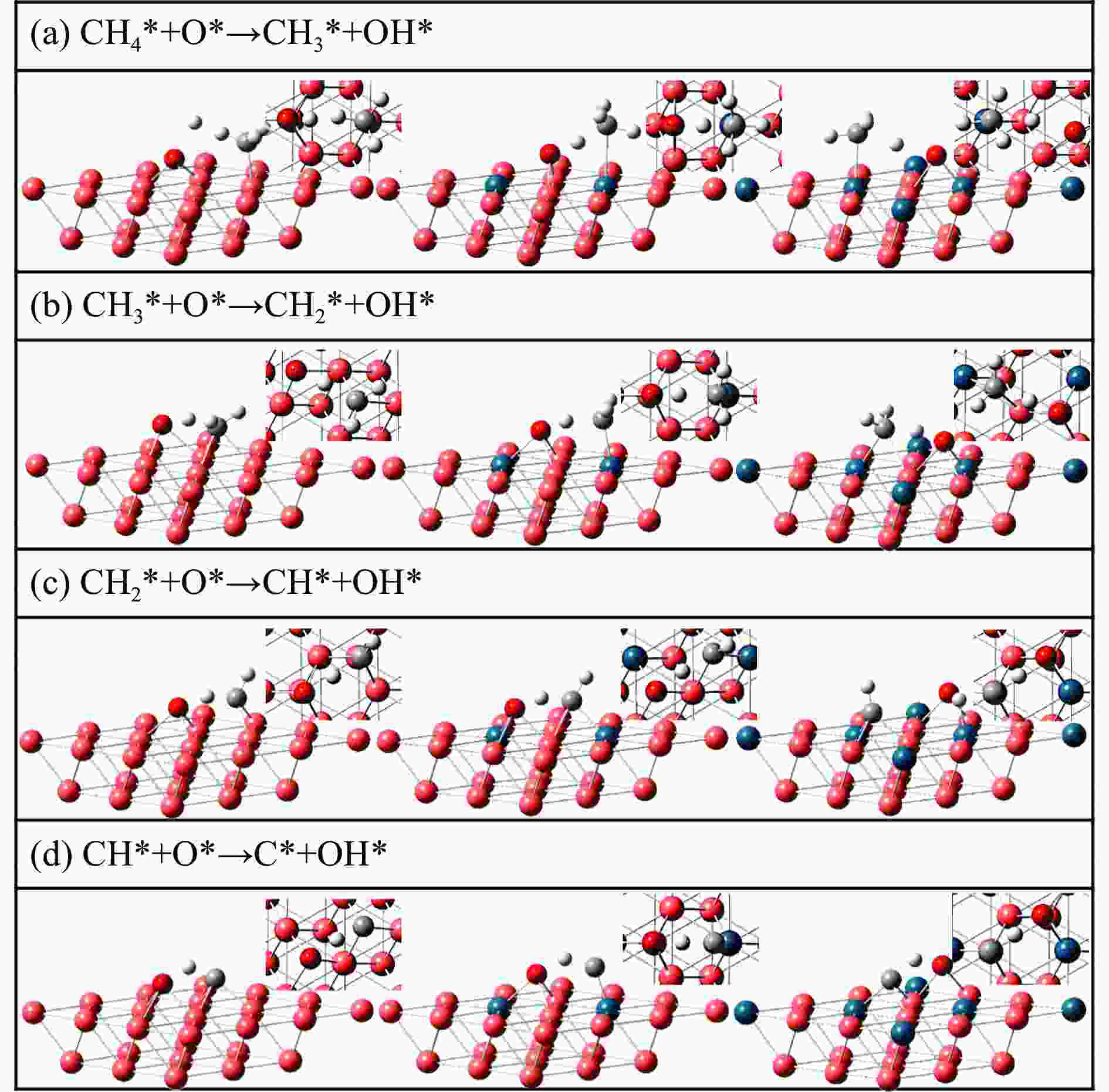
Table 3 Energy barrier and heat of reaction of O-assisted methane dehydrogenation over three catalyst surfaces (eV)
Elementary step Cu(111) Pd2Cu(111) Pd6Cu(111) barrier heat barrier heat barrier heat CH4* + O* → CH3* + OH* 1.56 0.45 1.38 0.25 1.44 0.32 CH3* + O*→ CH2* + OH* 0.64 −0.79 0.66 −0.09 0.80 −0.18 CH2* + O*→ CH* + OH* 0.71 −0.41 0.44 −0.13 0.66 −0.27 CH* + O*→ C* + OH* 0.83 −0.26 0.90 0.24 1.02 −0.09 -
[1] 天工. 《中国天然气发展报告(2021)》发布[J]. 天然气工业,2021,41(8):68.TIAN Gong. China Natural Gas Development Report (2021) released[J]. NGI,2021,41(8):68. [2] DAVID A. Attributing atmospheric methane to anthropogenic emission sources[J]. Acc Chem Res,2016,49(7):1344−1350. doi: 10.1021/acs.accounts.6b00081 [3] REAY D S, SMITH P, CHRISTENSEN T R, JAMES R H, CLARK H. Methane and global environmental change[J]. Annu Rev Env Resour,2018,43(1):165−92. doi: 10.1146/annurev-environ-102017-030154 [4] 楚培齐, 王赛飞, 赵世广, 张依, 邓积光, 刘雨溪, 郭萌, 段二红. 甲烷催化燃烧反应机理及催化剂研究进展[J]. 燃料化学学报,2022,50(2):180−191. doi: 10.19906/j.cnki.jfct.2021077CHU Pei-qi, WANG Sai-fei, ZHAO Shi-guang, ZHANG Yi, DENG Ji-guang, LIU Yu-xi, GUO Meng, DUAN Er-hong. Research progress of reaction mechanism and catalysts on catalytic methane combustion[J]. J Fuel Chem Technol,2022,50(2):180−191. doi: 10.19906/j.cnki.jfct.2021077 [5] HOU M L, ZHANG X, FU C, CEN W L, CHEN J X. Effects of Pd/Pt bimetal supported by γ-Al2O3 surface on methane activation[J]. Phys Chem Chem Phys,2020,22(8):1−21. [6] DIANAT A, SERIANI N, CIACCHI L C, BOBETH M, CUNIBERTI G. DFT study of reaction processes of methane combustion on PdO(100)[J]. Chem Phys,2014,443:53−60. doi: 10.1016/j.chemphys.2014.08.006 [7] JØRGENSEN M, GRÖNBECK H. First-principles microkinetic modeling of methane oxidation over Pd(100) and Pd(111)[J]. ACS Catal,2016,6(10):6730−6738. doi: 10.1021/acscatal.6b01752 [8] 齐大彬, 罗旭东, 姚君, 姚玉龙, 芦晓军. CO在Pd平板与Pd38团簇表面上的催化氧化机理研究[J]. 燃料化学学报,2020,48(4):432−439. doi: 10.1016/S1872-5813(20)30017-7QI Da-bin, LUO Xu-dong, YAO Jun, YAO Yu-long, LU Xiao-jun. Catalytic oxidation of CO on Pd38 cluster and Pd slab, a computational study[J]. J Fuel Chem Technol,2020,48(4):432−439. doi: 10.1016/S1872-5813(20)30017-7 [9] CHEN L X, MCCANN J P, TAIT S L. A re-examination of the catalyst activation and temperature hysteresis in methane combustion on Pt/Al2O3[J]. Appl Catat A: Gen,2018,549:19−30. doi: 10.1016/j.apcata.2017.09.008 [10] ZHAO C C, ZHAO Y H, LI S G, SUN Y H. Effect of Pd doping on CH4 reactivity over Co3O4 catalysts from density-functional theory calculations[J]. Chin J Catal,2017,38(5):813−820. doi: 10.1016/S1872-2067(17)62817-1 [11] LIU W G, GUO D Y, XU X. Research progress of palladium catalysts for methane combustion[J]. China Pet Process,2012,14(3):1−9. [12] SOLYMOSI F, ERDOHELYI A, CSERENYI J. Decemposition of CH4 over supported Pd catalysts[J]. J Catal,1994,147(1):272−278. doi: 10.1006/jcat.1994.1138 [13] QI W J, RAN J Y, WANG R R, DU X S, SHI J, RAN M C. Kinetic mechanism of effects of hydrogen addition on methane catalytic combustion over Pt(111) surface: A DFT study with cluster modeling[J]. Comput Mater Sci,2016,111:430−442. doi: 10.1016/j.commatsci.2015.09.002 [14] CHEN Y, VLACHOS D G. Density functional theory study of methane oxidation and reforming on Pt(111) and Pt(211)[J]. Ind Eng Chem Res,2012,51(38):12244−12252. [15] LIU H Y, ZHANG R G, YAN R X, WANG B J, XIE K C. CH4 dissociation on NiCo(111) surface: A first-principles study[J]. Appl Surf Sci,2011,257(21):8955−8964. doi: 10.1016/j.apsusc.2011.05.073 [16] HE J, YANG Z Q, DING C L, ZHANG L, YAN Y F, DU X S. Methane dehydrogenation and oxidation process over Ni-based bimetallic catalysts[J]. Fuel,2018,226:400−409. doi: 10.1016/j.fuel.2018.04.031 [17] NIU J T, WANG Y L, LILAND S E, SAMUEL K R, YANG J, ROUT K R, LUO J, RØNNING M, RAN J Y, CHEN D. Unraveling enhanced activity, selectivity, and coke-resistance of Pt-Ni bimetallic clusters in dry reforming[J]. ACS Catal,2021,11(4):2398−2411. doi: 10.1021/acscatal.0c04429 [18] NIU J T, LIU H Y, JIN Y, FAN B G, QI W J, RAN J Y. A density functional theory study of methane activation on MgO supported Ni9M1 cluster: Role of M on C–H activation[J]. Front Chem Sci Eng,2022,16(10):1485−1492. doi: 10.1007/s11705-022-2169-8 [19] JIANG Z, WU Z Q, FANG T, YI C H. Enhancement C–H bond activation of methane via doping Pd, Pt, Rh and Ni on Cu(111) surface: A DFT study[J]. Chem Phys Lett,2019,715:323−329. doi: 10.1016/j.cplett.2018.12.001 [20] MENG Y Y, DING C M, GAO X F, MA L C, ZHANG K, WANG J W, LI Z. Adsorption of Pd on the Cu(111) surface and its catalysis of methane partial oxidation: A density functional theory study[J]. Appl Surf Sci,2020,513:145724. doi: 10.1016/j.apsusc.2020.145724 [21] WANG J, WANG G C. Promotion effect of methane activation on Cu(111) by the surface-active oxygen species: A combination of DFT and ReaxFF study[J]. J Phys Chem C,2018,122(30):17338−17346. doi: 10.1021/acs.jpcc.8b05294 [22] 钱梦丹, 薛继龙, 夏盛杰, 倪哲明, 蒋军辉, 曹勇勇. Pd/Cu(111)双金属表面催化糠醛脱碳及加氢的反应机理[J]. 燃料化学学报,2017,45(1):34−42. doi: 10.1016/S1872-5813(17)30008-7QIAN Meng-dan, XUE Ji-long, XIA Sheng-ji, NI Zhe-ming, JIANG Jun-hui, CAO Yong-yong. Decarbonylation and hydrogenation reaction of furfural on Pd/Cu(111) surface[J]. J Fuel Chem Technol,2017,45(1):34−42. doi: 10.1016/S1872-5813(17)30008-7 [23] 康建东. 铜基催化剂氧化低浓度甲烷的反应动力学及性能调控[D]. 重庆: 重庆大学, 2020.KANG Jian-dong. Reaction kinetics and performance regulation of copper-based catalysts for oxidation of low-concentration methane[D]. Chongqing, Chongqing University, 2020. [24] GONZALEZ C, SCHLEGEL H B. An improved algorithm for reaction path following[J]. J Chem Phys,1989,90(4):2154. doi: 10.1063/1.456010 [25] BU X X, RAN J Y, NIU J T, OU Z L, TANG L, HUANG X. Reaction mechanism insights into CH4 catalytic oxidation on Pt13 cluster: A DFT study[J]. Mol Catal,2021,515:111891. doi: 10.1016/j.mcat.2021.111891 [26] HAMMER B, NØRSKOV J K. Theoretical surface science and catalysis-calculations and concepts[J]. Adv Catal,2000,(45):1−71. [27] LU T, CHEN F W. Multiwfn: A multifunctional wavefunction analyzer[J]. J Comput Chem,2012,(33):580−592. -




 下载:
下载: