Study on the transformation behavior of Fe and Ca compounds in residue at different reaction space velocities
-
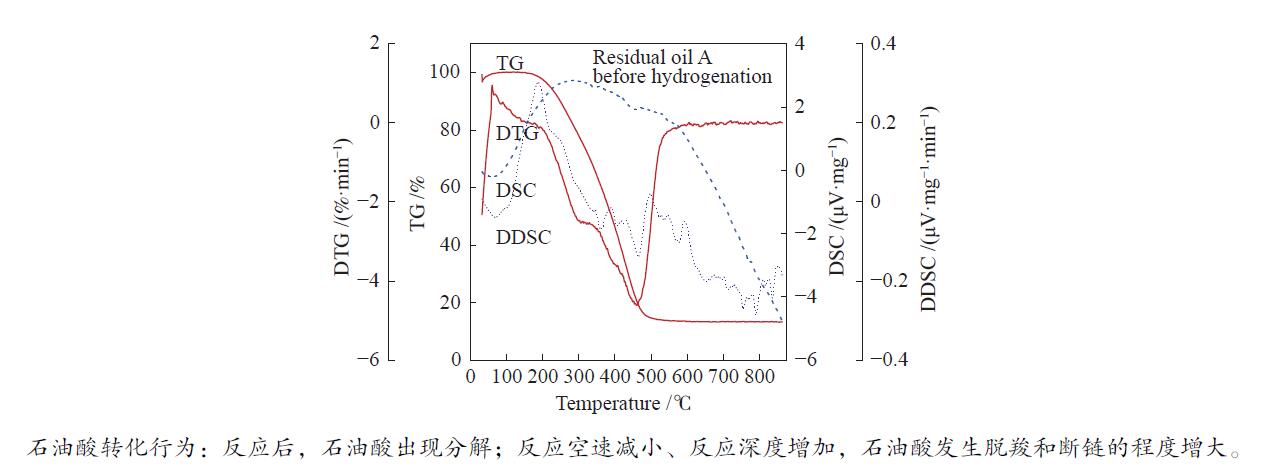
摘要: 以不同空速下固定床加氢反应前后的油样为研究对象,考察了渣油中铁、钙化合物在固定床加氢反应过程中赋存形态的转化行为。使用酸解-醇碱法将石油酸盐中的石油酸部分萃取出来。通过红外光谱、元素分析、核磁共振氢谱、核磁共振碳谱、高分辨率质谱等方法对石油酸结构进行表征。为了研究石油酸在受热条件下的转化,将所得到的石油酸进行热重-质谱表征,分析石油酸在热反应中的转化行为,进而推断得到铁、钙化合物在热反应中的转化行为。结果表明,首先,相对于原料样品,反应样品酸值呈现显著下降,由原料的1.01 mg/g下降到0.08 mg/g以下,表明酸性物质形态发生了反应。固定床加氢反应前后渣油中石油酸的主要成分为环烷酸,且以多聚体的形式存在。渣油在固定床加氢反应前后,石油酸出现较为明显的分解,主要产物为CO2、C3H8等物质;随着反应深度的增加,石油酸发生脱羧反应和断链反应的程度增大,使石油酸分解。Abstract: For the oil samples before and after fixed bed hydrogenation reaction at different space speeds, transformation behavior of iron and calcium compounds were studied. The oil samples were decomposed with acid and then extracted with aqueous alcoholic solution of sodium hydroxide. The corresponding petroleum acids derived from petroleum acid salts were obtained. The structure of petroleum acid was characterized by infrared spectrometry, element analysis, hydrogen nuclear magnetic spectrum, nuclear magnetic carbon and high resolution mass spectrometry. In order to study the transformation of petroleum acid in thermal reaction, the obtained petroleum acid was characterized by thermogravimetric mass spectrometry. The transformation process of petroleum acid in thermal reaction was analyzed, and then the transformation process of iron and calcium compounds in thermal reaction was obtained. The results show that the main components of oil acid in residue before and after the hydrogenation reaction are naphthenic acid existing as the form of polymer. Before and after the thermal modification process, petroleum acid is decomposed obviously, and the main products are CO2, C3H8 and other substances. With the increase of reaction depth, the degree of decarboxylation and chain breaking reaction of petroleum acid increases, which further makes petroleum acid decomposition.
-
表 1 渣油加氢前后油样的基础物性参数
Table 1 Basic physical parameters of oil samples before and after residue hydrogenation
Analysis item Residual oil A before hydrogenation Residual oil B after hydrogenation Residual oil C after hydrogenation Residual oil D after hydrogenation Residual oil E after hydrogenation Density
(20 ℃) /(g·cm−3)0.992 0.978 0.971 0.973 0.958 Colloid stability parameter /(g·g−1) 1.76 1.81 2.37 2.05 2.57 Acid number /(mg·g−1) 1.01 0.079 0.046 0.042 0.041 Relative molecular mass /(g·mol−1) 864 696 677 665 633 Temperature /°C 375 375 375 375 Pressure /MPa 18.0 18.0 18.0 18.0 Airspeed /h−1 1.70 1.40 1.10 0.60 C /% 83.57 84.49 84.88 84.9 85.35 H /% 10.74 10.95 11.39 11.37 11.57 N /% 0.32 0.31 0.32 0.31 0.29 S /% 4.74 4.18 3.38 3.34 2.78 Fe /(μg∙g−1) 32.3 20.0 15.4 9.38 7.36 Ca /(μg∙g−1) 8.92 10.7 8.56 7.54 5.38 表 2 加氢前后石油酸的元素含量
Table 2 Element content of petroleum acid before and after hydrogenation
Oil sample C/% H/% S/% N/% O*/% H/C (atom ratio) Residual oil A before hydrogenation 76.0 10.1 3.28 0.83 9.79 1.59 Residual oil B after hydrogenation (1.70 h−1) 65.8 9.38 2.19 0.68 21.95 1.71 Residual oil C after hydrogenation (1.40 h−1) 62.8 9.71 1.38 0.47 25.64 1.86 Residual oil D after hydrogenation (1.10 h−1) 62.6 9.39 1.64 0.52 25.84 1.80 Residual oil E after hydrogenation (0.60 h−1) 56.9 9.59 0.86 0.44 32.21 2.02 *: The content of oxygen element was obtained by reduction method 表 3 加氢前后渣油中石油酸盐结构中各类氢的相对含量
Table 3 Relative contents of various kinds of hydrogen in petroleum acid structure of residue before and after hydrogenation
Hydrogen type Symbol Chemical
shift δRelative amounts of each type of hydrogen /% residue A petroleum acid residue B petroleum acid residue C petroleum acid residue D petroleum acid residue E petroleum acid Saturated hydrogen 0.5−4 86.7 80.3 80.3 80.4 77.1 The γ site of the aromatic ring and the far CH3 hydrogen Hγ 0.5−1.05 18.2 12.1 11.6 8.82 8.50 The β site of the aromatic ring and the far CH2 and CH hydrogen Hβ 1.05−1.95 43.9 44.7 44.6 44.8 45.8 The hydrogen attached to the α carbon of the aromatic ring Hα 1.95−4.0 24.6 23.5 24.1 26.8 22.8 Phenyl cyclic hydrogen HA 6.0−9.0 13.3 19.7 19.7 19.6 22.9 表 4 加氢前后渣油中石油酸结构中各类碳的相对含量
Table 4 Relative contents of various kinds of carbon in the petroleum acid structure of residue before and after hydrogenation
Carbon type Chemical shift Relative amounts of each type of carbon/% residue A petroleum acid residue B petroleum acid residue C petroleum acid residue D petroleum acid residue E petroleum acid Carboxyl carbon 178−184 3.91 3.01 1.92 1.15 0.94 Aromatic carbon 100−150 54.9 52.4 51.12 32.92 26.4 Proton aromatic carbon 100−127.1 22.1 6.58 12.96 10.31 4.94 Bridge head aromatic carbon 127.1−133 25.7 22.4 17.51 20.38 19.6 CH3 replaces aryl carbon 133−135 2.18 2.38 1.42 1.34 1.26 Naphthenic aryl carbon 135−137 1.73 3.01 0.58 0.36 0.27 ≥C2 alkyl substituted aryl carbon 137−150 3.24 18.0 0.04 0.53 0.36 Saturated carbon 10−66 41.2 44.6 46.96 65.93 72.7 γ-CH3 10−15 3.02 2.62 4.91 5.12 5.20 β-CH3 15−18 1.60 0.40 1.28 1.05 0.01 α-CH3, CH3 replaced by naphthenes 18−22.5 8.57 5.76 9.03 8.03 7.84 α-CH2, β-CH2, CH2 substituted by naphthenes 22.5−37 28.4 36.4 30.57 44.69 57.3 α-CH, CH substituted by naphthenes 37−66 0.36 0.59 1.17 7.04 2.24 表 5 加氢反应前后石油酸的类型和相对含量
Table 5 Types of petroleum acids before and after hydrogenation
Unsaturation Ω Carboxylic acid type Relative amounts /% residue A petroleum acid residue C petroleum acid residue D petroleum acid residue E petroleum acid 1 fatty acid 9.1 11.4 15.8 24.6 2 monocyclic acid 20.6 22.7 23.6 22.2 3 dicyclic naphthenic acid 29.1 28.9 28.8 25.9 4 tricyclic carboxylic acid 19.6 17.4 17.4 14.6 5 tetracyclic naphthenic acid/aryclic carboxylic acid 8.09 5.71 6.41 5.6 6 pentacyclic naphthenic acid/aromatic cyclic monocyclic carboxylic acid 6.09 6.87 5.41 3.79 7 hexacyclic naphthenic acid/aryclic dicyclic carboxylic acid 7.55 6.97 3.56 3.21 -
[1] QI L Q, LI J T, YAO Y, ZHANG Y J. Heavy metal poisoned and regeneration of selective catalytic reduction catalysts[J]. J Hazard Mater,2019,366:492−500. doi: 10.1016/j.jhazmat.2018.11.112 [2] JIANG H, KENNETH J L, SHANKHAMALA K, CHENG W C. Characterization of iron contamination on equilibrium fluid catalytic cracking catalyst particles[J]. J Catal,2018,361:126−134. doi: 10.1016/j.jcat.2018.02.025 [3] MARAFI A, ALBAZZAZ H, RANA M S. Hydroprocessing of heavy residual oil: Opportunities and challenges[J]. Catal Today,2019,329:125−134. doi: 10.1016/j.cattod.2018.10.067 [4] ETIM U J, BAI P, ULLAH R, SUBHAN F, YAN Z F. Vanadium contamination of FCC catalyst: Understanding the destruction and passivation mechanisms[J]. Appl Catal A: Gen,2018,555:108−117. doi: 10.1016/j.apcata.2018.02.011 [5] PSARRAS A C, ILIOPOULOU E F, NALBANDIAN L, LAPPAS A A, POUWELS C. Study of the accessibility effect on the irreversible deactivation of FCC catalysts from contaminant feed metals[J]. Catal Today,2007,127(1-4):44−53. doi: 10.1016/j.cattod.2007.05.021 [6] CERQUEIRA H S, CAEIRO G, COSTA L, RAMOA R F. Deactivation of FCC catalysts[J]. J Mol Catal A: Chem,2008,292(1-2):1−13. doi: 10.1016/j.molcata.2008.06.014 [7] ORTIGUEIRA J, ALVES L, GOUVEIA L, MOURA P. Third generation biohydrogen production by clostridium butyricum and adapted mixed cultures from scenedesmus obliquus microalga biomass[J]. Fuel,2015,153:128−134. doi: 10.1016/j.fuel.2015.02.093 [8] 侯典国, 汪燮卿. 我国一些原油中钙化合物分布及形态的研究[J]. 石油学报(石油加工),2000,16(1):54−59.HOU Dian-guo, WANG Xie-qing. Study on distribution and morphology of calcium compounds in some crude oil in our country[J]. Acta Pet Sin(Pet Proc Sect),2000,16(1):54−59. [9] 王现元, 张涛, 张龙力, 赵愉生, 边钰清, 杨朝合. 渣油加氢反应样品中含铁和含钙化合物溶解性能研究[J]. 燃料化学学报,2021,49(6):771−779. doi: 10.1016/S1872-5813(21)60053-1WANG Xian-yuan, ZHANG Tao, ZHANG Long-li, ZHAO Yu-sheng, BIAN Yu-qing, YANG Chao-he. Study on dissolution properties of iron and calcium containing compounds in residual oil hydroreaction samples[J]. J Fuel Chem Technol,2021,49(6):771−779. doi: 10.1016/S1872-5813(21)60053-1 [10] 邹滢, 宋丽, 翁惠新. 辽河减压渣油中铁的赋存与分布特征[J]. 石油炼制与化工,2007,38(11):60−64. doi: 10.3969/j.issn.1005-2399.2007.11.014ZOU Ying, SONG Li, WENG Hui-xin. The occurrence and distribution characteristics of Liaohe vacuum residuum railway[J]. Pet Process Petrochem,2007,38(11):60−64. doi: 10.3969/j.issn.1005-2399.2007.11.014 [11] 赵元生, 赵愉生, 夏恩冬, 张龙力, 张志国, 宋元栋. 上流式反应器用于劣质渣油加氢处理的初步探索[J]. 石油化工,2016,45(11):1363−1368. doi: 10.3969/j.issn.1000-8144.2016.11.013ZHAO Yuan-sheng, ZHAO Yu-sheng, XIA Dong-en, ZHANG Long-li, ZHANG Zhi-guo, SONG Yuan-dong. Preliminary study on the upflow reactor for the hydrotreating of inferior residual oil[J]. Chin Petrochem Technol,2016,45(11):1363−1368. doi: 10.3969/j.issn.1000-8144.2016.11.013 [12] 蒋迪, 王大鸷, 徐同宽, 张绍印. 辽河油田减二线柴油中环烷酸的脱除[J]. 大连工业大学学报,2012,31(2):123−126.JIANG Di, WANG Da-zhi, XU Tong-kuan, ZHANG Shao-yin. Removal of naphthenic acid from reduced second line diesel oil in Liaohe Oilfield[J]. J Dalian Inst Technol,2012,31(2):123−126. [13] 郑盟主, 戴俊峰, 袁宏强, 马永清, 王亚波, 赵永平. 高酸原油脱酸工艺条件优化[J]. 石油化工应用,2016,35(5):135−138.ZHENG Meng-zhu, DAI Jun-feng, YUAN Hong-qiang, MA Yong-qing, WANG Ya-bo, ZHAO Yong-ping. Optimization of deacidification process of high acid crude oil[J]. Petrochem Inst Appl,2016,35(5):135−138. [14] WANG X Y, ZHANG T, ZHANG L L, ZHAO Y S, YANG C H, CUI R L. Distribution and form of iron and calcium compounds before and after residue hydrogenation under different space velocities[J]. China Pet Process Petrochem Technol,2022,24(3):86−94. [15] 龚剑洪, 陆善祥, 崔建, 贺彩霞. 国产重油组成的表征[J]. 石油炼制与化工,2000,31(10):48−53.GONG Hong-jian, LU Shan-xiang, CUI Jian, HE Cai-xia. Characterization of domestic heavy oil composition[J]. Pet Process Petrochem,2000,31(10):48−53. [16] 鲁阿信, 肖光, 付新梅, 胡侠. 达尔原油中石油酸的结构组成分析[J]. 石油化工,2010,39(10):48−53.LU A-xin, XIAO Guang, FU Xin-mei, HU Xia. Structure and composition analysis of oleic acid in Dahl crude oil[J]. Chin Petrochem Technol,2010,39(10):48−53. [17] 刘泽龙, 田松柏, 樊雪志, 杨明彪. 蓬莱原油初馏点 ~ 350 ℃馏分中石油羧酸的结构组成[J]. 石油学报(石油加工),2003,19(6):40−45.LIU Ze-long, TIAN Song-bai, FAN Xue-zhi, YANG Ming-biao. Structural composition of petroleum carboxylic acids in the initial distillate point ~ 350 ℃ fraction of Penglai Crude Oil[J]. Acta Pet Sin(Pet Proc Sect),2003,19(6):40−45. -





 下载:
下载: