Research on the 9,10-dihyroanthrancene assisted catalytic pyrolysis of pine over nitrogen-doped activated carbon for preparation of alkoxyphenols
-
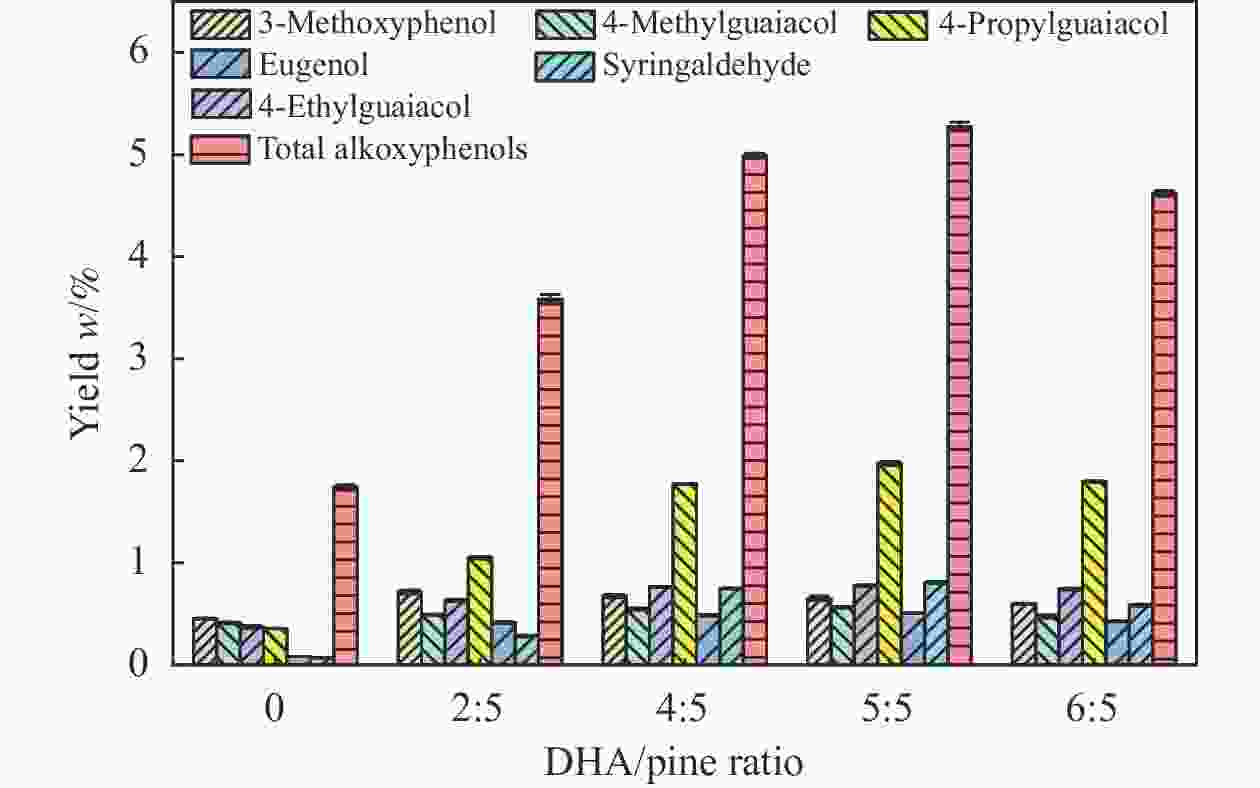
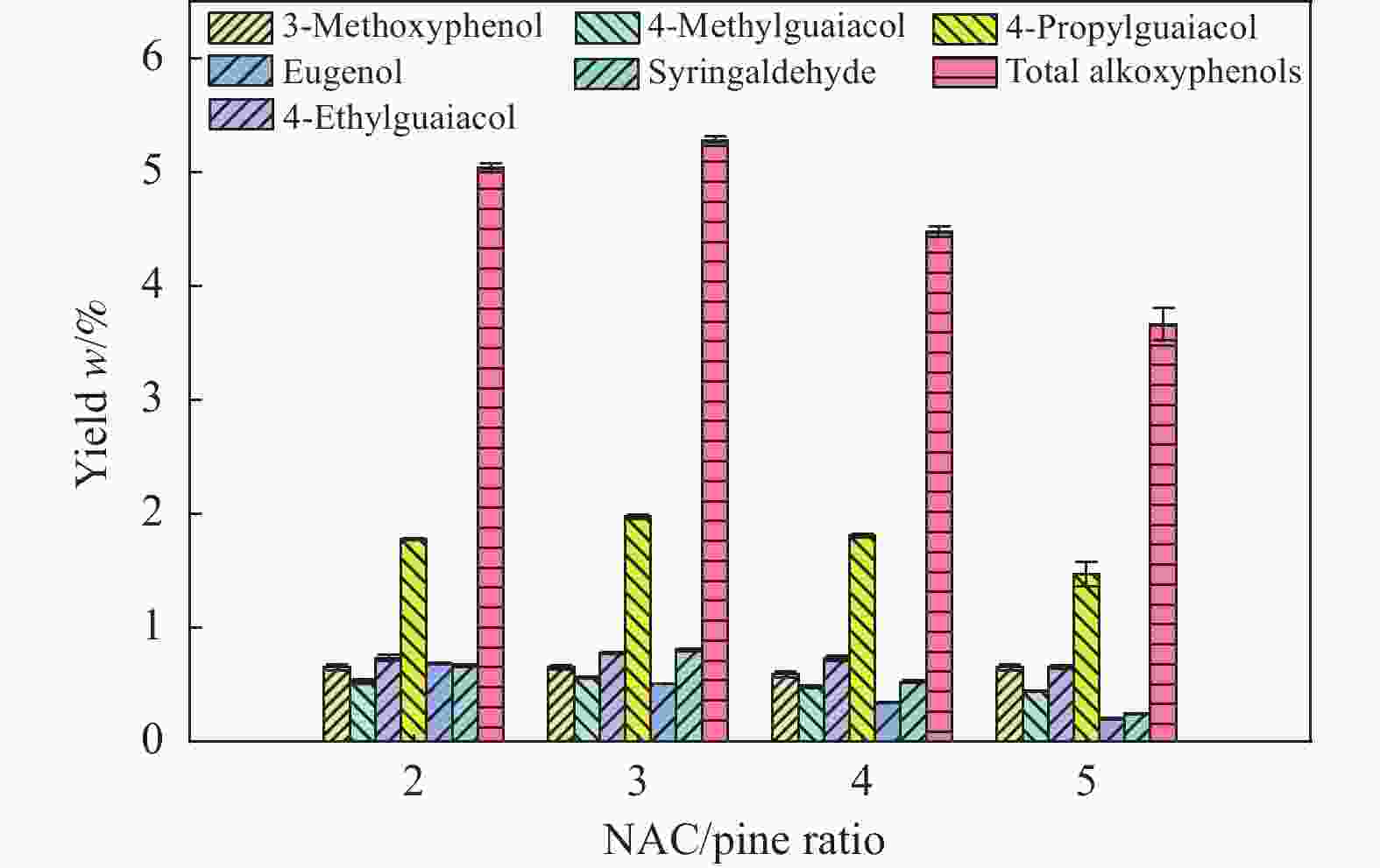
摘要: 本研究以核桃壳基掺氮活性炭(NAC)为催化剂、以9,10-二氢蒽(DHA)为供氢剂,开展了松木选择性热解制备烷氧基酚研究,探究了氨水浓度对NAC理化性能的影响,揭示了DHA/松木比、热解温度、NAC/松木比对烷氧基酚生成的调控机制。结果表明,合适的氨水浓度能够改善NAC孔隙结构及活性位点分布,当氨水浓度为15%时,所制备NAC对烷氧基酚的生成促进效果最佳;当DHA/松木比为3、热解温度为550 ℃、NAC/松木比为3时,烷氧基酚产率最大,为5.27%,明显高于纯松木直接催化热解时烷氧基酚产率(1.74%)。Abstract: In this study, nitrogen-doped activated carbon (NAC) was prepared from walnut shell and applied to the catalytic pyrolysis of pine for selective preparation of alkoxyphenols with 9,10-dihyroanthrancene (DHA) as the hydrogen donor. The effects of ammonia concentration on the physicochemical properties of NAC were investigated. The regulatory functions of DHA/pine ratio, pyrolysis temperature, and NAC/pine ratio on the generation of alkoxyphenols were revealed. The results showed that the pore structure and active sites distribution of NAC could be improved by proper ammonia concentration. The NAC prepared at ammonia concentration of 15% was the best for the production of alkoxylphenols. The yield of alkoxylphenols reached its maximum value of 5.27% at DHA/pine ratio of 3, pyrolysis temperature of 550 ℃, and NAC/pine mass ratio of 3, which was much higher than that from catalytic pyrolysis of pure pine (1.74%).
-
Key words:
- biomass /
- nitrogen-doped activated carbon /
- catalytic pyrolysis /
- 9,10-dihyroanthrancene /
- alkoxyphenols
-
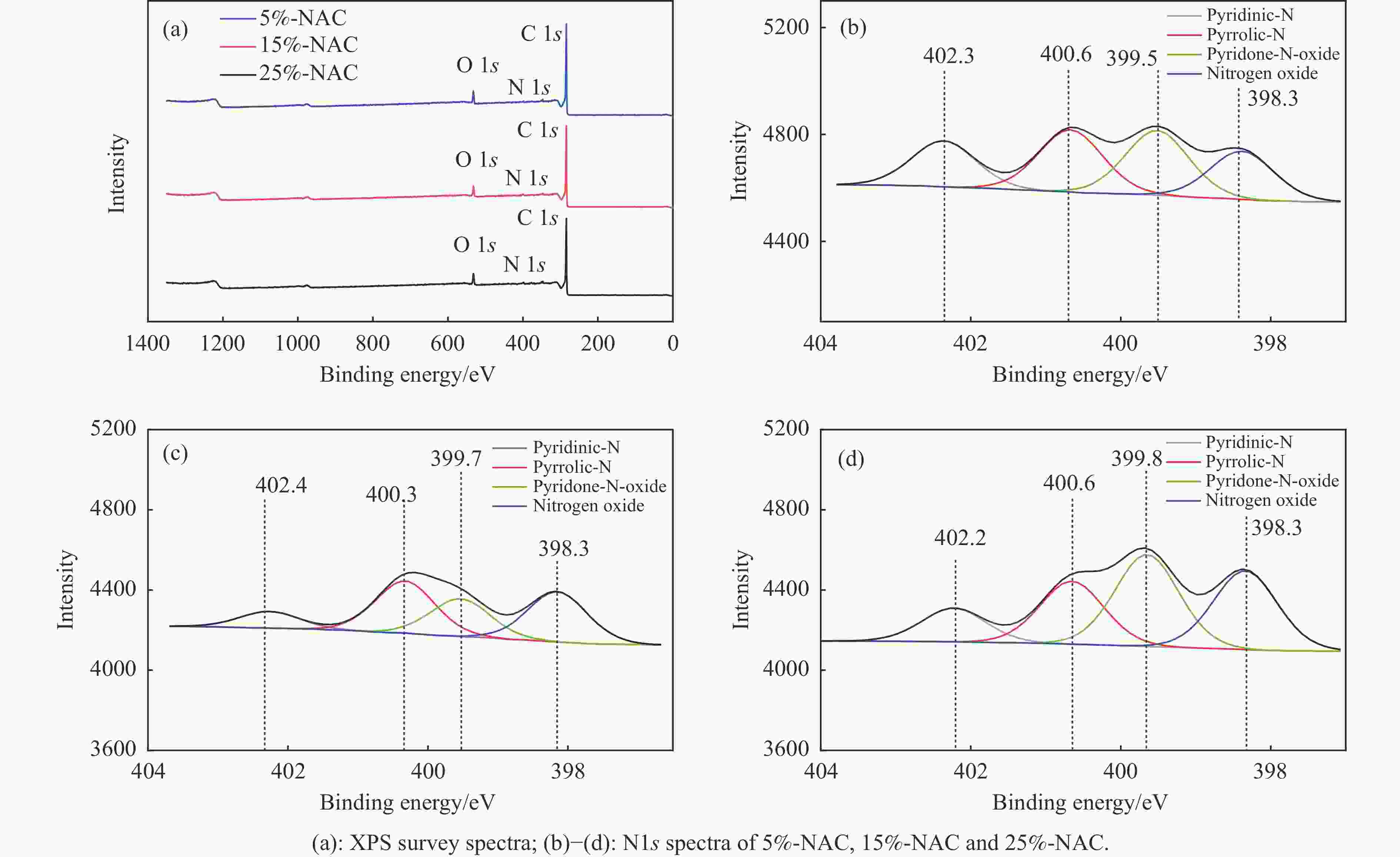
表 1 NAC的孔结构参数
Table 1 Pore structure parameters of NAC
Sample SBET/(m2·g−1) Smica/(m2·g−1) vtotalb/(cm3·g−1) vmicc/(cm3·g−1) vmic/vtotal/% daverage/nm 0%-NAC 748.541 528.310 0.486 0.262 53.91 2.493 5%-NAC 871.653 565.312 0.547 0.298 54.48 2.512 15%-NAC 891.144 749.898 0.503 0.394 78.33 2.258 25%-NAC 856.319 780.752 0.442 0.384 86.88 2.066 a, c: t-plot surface area and pore volume of micropores, b: p/p0= 0.9907. 表 2 NAC中不同种类含氮官能团的相对含量
Table 2 Relative contents of different types of nitrogen-containing functional groups in NAC
Sample Pyridinic-N/% Nitrogen oxide/% Pyrrolic-N/% Pyridone-N-oxide/% 5%-NAC 21.78 29.24 28.19 20.79 15%-NAC 32.03 24.07 33.26 10.64 25%-NAC 29.47 34.41 23.54 12.58 -
[1] CHANG C, LIU Z, LI P, et al. Study on products characteristics from catalytic fast pyrolysis of biomass based on the effects of modified biochars[J]. Energy,2021,229:120818. doi: 10.1016/j.energy.2021.120818 [2] 邓晶晶, 罗泽军, 王储, 等. 疏水性离子液体对生物油水相馏分中酚类物质的萃取研究[J]. 燃料化学学报,2021,49(12):1832−1838. doi: 10.1016/S1872-5813(21)60108-1DENG Jingjing, LUO Zejun, WANG Chu, et al. Extraction of phenols from bio-oil aqueous fraction by hydrophobic ionic liquids[J]. J Fuel Chem Technol,2021,49(12):1832−1838. doi: 10.1016/S1872-5813(21)60108-1 [3] 孙孟超, 袁鑫华, 罗泽军, 等. 蒸馏温度对核桃壳生物油馏分组分分布的影响[J]. 燃料化学学报,2020,48(10):1179−1185. doi: 10.1016/S1872-5813(20)30079-7SUN Mengchao, YUAN Xinhua, LUO Zejun, et al. Influence of heating temperatures on the component distributionof distillates derived from walnut shell bio-oil[J]. J Fuel Chem Technol,2020,48(10):1179−1185. doi: 10.1016/S1872-5813(20)30079-7 [4] WANG S, RU B, LIN H, et al. Pyrolysis behaviors of four lignin polymers isolated from the same pine wood[J]. Bioresour Technol,2015,182:120−127. doi: 10.1016/j.biortech.2015.01.127 [5] TOTONG S, DAORATTANACHAI P, QUITAIN A, et al. Catalytic depolymerization of alkaline lignin into phenolic-based compounds over metal-free carbon-based catalysts[J]. Ind Eng Chem Res,2019,58:13041−13052. [6] SEKHON S S, PARK J-S. Biomass-derived N-doped porous carbon nanosheets for energy technologies[J]. Chem Eng J,2021,425:129017. doi: 10.1016/j.cej.2021.129017 [7] AYIANIA M, WEISS-HORTALA E, SMITH M, et al. Microstructural analysis of nitrogen-doped char by Raman spectroscopy: Raman shift analysis from first principles[J]. Carbon,2020,167:559−574. doi: 10.1016/j.carbon.2020.05.055 [8] WAN Y, HU Y, ZHOU W. Catalytic mechanism of nitrogen-doped biochar under different pyrolysis temperatures: The crucial roles of nitrogen incorporation and carbon configuration[J]. Sci Total Environ,2022,816:151502. doi: 10.1016/j.scitotenv.2021.151502 [9] CHEN W, FANG Y, KAIXU L, et al. Bamboo wastes catalytic pyrolysis with N-doped biochar catalyst for phenols products[J]. Appl Energy,2020,260:114242. doi: 10.1016/j.apenergy.2019.114242 [10] 郭岳. 介孔掺氮炭负载镍基催化剂催化木质素解聚制备单酚类化合物[D]. 广州: 华南理工大学, 2021.GUO Yue. Mesoporous nitrogen-doped carbon supported nickel-based catalyst for catalytic depolymerization of lignin to prepare monophenolic compounds[D]. Guangzhou: South China University of Technology, 2021. [11] MA S-W, LI H, ZHANG G, et al. Catalytic fast pyrolysis of walnut shell for alkylphenols production with nitrogen-doped activated carbon catalyst[J]. Front Environ Sci Eng,2020,15(2):25. [12] LIU X, ZUO S, CUI N, et al. Investigation of ammonia/steam activation for the scalable production of high-surface area nitrogen-containing activated carbons[J]. Carbon,2022,191:581−592. doi: 10.1016/j.carbon.2022.02.014 [13] 陆强, 郭浩强, 叶小宁, 等. 供氢剂作用下生物质快速热解的研究进展[J]. 林产化学与工业,2017,37(6):1−9. doi: 10.3969/j.issn.0253-2417.2017.06.001LU Qiang, GUO Haoqiang, YE Xiaoning, et al. Research progress of hydrogen donor assisted fast pyrolysis of biomass[J]. Chem Ind For Prod,2017,37(6):1−9. doi: 10.3969/j.issn.0253-2417.2017.06.001 [14] ZHANG Z-X, LI K, MA S-W, et al. Fast pyrolysis of biomass catalyzed by magnetic solid base catalyst in a hydrogen atmosphere for selective production of phenol[J]. Ind Crops Prod,2019,137:495−500. doi: 10.1016/j.indcrop.2019.05.066 [15] LU Q, ZHANG G, ZHANG Z-X, et al. Enhanced production of 4-ethyl phenol from activated carbon catalyzed fast pyrolysis of bagasse with 9, 10-dihydroanthracene as a hydrogen donor[J]. J Anal Appl Pyrolysis,2020,150:104880. doi: 10.1016/j.jaap.2020.104880 [16] RYU D-Y, SHIMOHARA T, NAKABAYASHI K, et al. Urea/nitric acid co-impregnated pitch-based activated carbon fiber for the effective removal of formaldehyde[J]. J Ind Eng Chem,2019,80:98−105. doi: 10.1016/j.jiec.2019.07.036 [17] TSAMO C, KAMGA R. Variation of physico-chemical and textural properties of laboratory prepared red mud through acid and thermal activations[J]. Adv Mater,2017,6:11−19. doi: 10.11648/j.am.20170602.12 [18] 林燕. 氨水浓度对果壳活性炭结构及性能的影响[J]. 福建工程学院学报,2017,15(1):9−14. doi: 10.3969/j.issn.1672-4348.2017.01.003LIN Yan. Effect of aqueous ammonia concentration on the structure and property of nut activated carbon[J]. J Fujian Univ Technol,2017,15(1):9−14. doi: 10.3969/j.issn.1672-4348.2017.01.003 [19] 刁瑞, 王储, 朱谢飞, 等. 炭化程度对核桃壳焦孔隙结构和燃烧特性的影响[J]. 燃料化学学报,2019,47(10):1173−1180.DIAO Rui, WANG Chu, ZHU Xiefei, et al. Influence of carbonization de gree of walnut shell char on pore structure and combustion characteristics[J]. J Fuel Chem Technol,2019,47(10):1173−1180. [20] SEKHON S S, KAUR P, PARK J-S. From coconut shell biomass to oxygen reduction reaction catalyst: Tuning porosity and nitrogen doping[J]. Renewable Sustainable Energy Rev,2021,147:111173. doi: 10.1016/j.rser.2021.111173 [21] BAŞER B, YOUSAF B, YETIS U, et al. Formation of nitrogen functionalities in biochar materials and their role in the mitigation of hazardous emerging organic pollutants from wastewater[J]. J Hazardous Mater,2021,416:126131. doi: 10.1016/j.jhazmat.2021.126131 [22] ZOU K, DENG Y, CHEN J, et al. Hierarchically porous nitrogen-doped carbon derived from the activation of agriculture waste by potassium hydroxide and urea for high-performance supercapacitors[J]. J Power Sources,2018,378:579−588. doi: 10.1016/j.jpowsour.2017.12.081 [23] 张德懿, 雷龙艳, 尚永花. 氮掺杂对碳材料性能的影响研究进展[J]. 化工进展,2016,35(3):831−836. doi: 10.16085/j.issn.1000-6613.2016.03.028ZHANG Deyi, LEI Longyan, SHANG Yonghua, et al. Effect of the nitrogen doping on the performance of nano-structure carbon materials: a review[J]. Chem Ind Eng Prog,2016,35(3):831−836. doi: 10.16085/j.issn.1000-6613.2016.03.028 [24] 李宇明, 刘梓烨, 张启扬, 等. 氮掺杂碳材料的制备及其在催化领域中的应用[J]. 化工学报,2021,72(8):3919−3932. doi: 10.11949/0438-1157.20201932LI Yuming, LIU Ziye, ZHANG Qiyang, et al. Preparation of nitrogen-doped carbon materials and their applications in catalysis[J]. CIESC J,2021,72(8):3919−3932. doi: 10.11949/0438-1157.20201932 [25] XUE Y, ZHOU S, BAI X. Role of hydrogen transfer during catalytic copyrolysis of lignin and tetralin over HZSM-5 and HY zeolite catalysts[J]. ACS Sustainable Chem Eng,2016,4(8):4237−4250. [26] HU B, LU Q, ZHANG Z-X, et al. Mechanism insight into the fast pyrolysis of xylose, xylobiose and xylan by combined theoretical and experimental approaches[J]. Combust Flame,2019,206:177−188. doi: 10.1016/j.combustflame.2019.04.052 [27] FAN L, ZHANG Y, LIU S, et al. Bio-oil from fast pyrolysis of lignin: Effects of process and upgrading parameters[J]. Bioresour Technol,2017,241:1118−1126. doi: 10.1016/j.biortech.2017.05.129 [28] 冯亮亮, 黄黎, 严珍奇, 等. 氨水改性活性炭对沼气吸附脱碳的实验研究[J]. 太阳能学报,2021,42(9):400−404. doi: 10.19912/j.0254-0096.tynxb.2019-0957FENG Liangliang, HUANG Li, YAN Zhenqi, et al. Experimental study on ammonia modified activated carbon for methane adsorption and decarbonization[J]. Acta Energ Sol Sin,2021,42(9):400−404. doi: 10.19912/j.0254-0096.tynxb.2019-0957 [29] ZHANG Z-B, LU Q, YE X-N, et al. Production of phenolic-rich bio-oil from catalytic fast pyrolysis of biomass using magnetic solid base catalyst[J]. Energy Convers Manag,2015,106:1309−1317. doi: 10.1016/j.enconman.2015.10.063 [30] WANG K, ZHANG J, SHANKS B H, et al. Catalytic conversion of carbohydrate-derived oxygenates over HZSM-5 in a tandem micro-reactor system[J]. Green Chem,2015,17(1):557−564. doi: 10.1039/C4GC01784F [31] 苏银海, 张书平, 刘凌沁, 等. 活性炭催化热解纤维素协同制备酚类和合成气[J]. 化工学报,2021,72(10):5206−5217. doi: 10.11949/0438-1157.20210416SU Yinhai, ZHANG Shuping, LIU Lingqin, et al. Synergetic production of phenols and syngas from the catalytic pyrolysis of cellulose on activated carbon[J]. CIESC J,2021,72(10):5206−5217. doi: 10.11949/0438-1157.20210416 [32] 邵晴莉, 左宋林, 肖志良, 等. 生物质热裂解反应器对镍/木炭催化剂重复使用过程中积炭的影响研究[J]. 林产化学与工业,2015,35(2):17−24.SHAO Qingli, ZUO Songlin, XIAO Zhiliang, et al. Effect of biomass pyrolysis reactor on carbon deposition of Ni/Char catalysts during repeat experiments[J]. Chem Ind For Prod,2015,35(2):17−24. -





 下载:
下载: