Selective dealumination of ZSM-5 by steaming and its effect on ethanol to propene
-
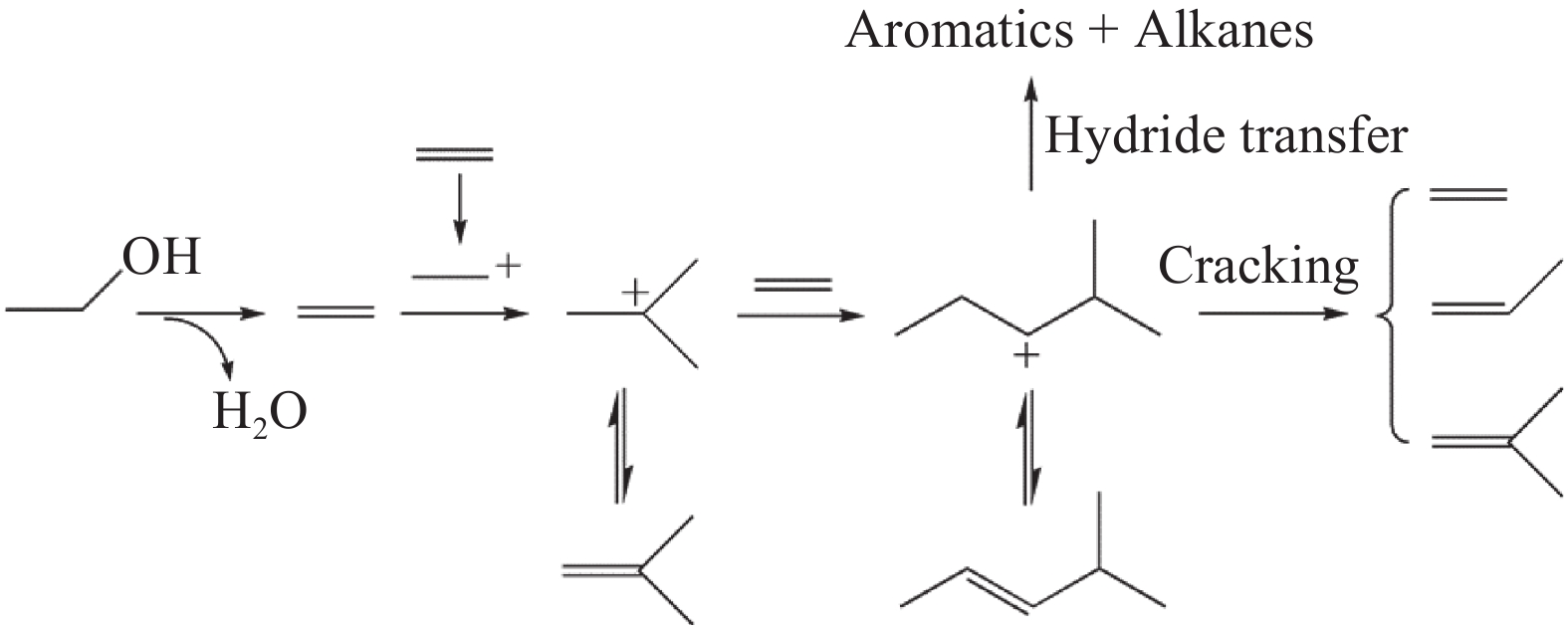
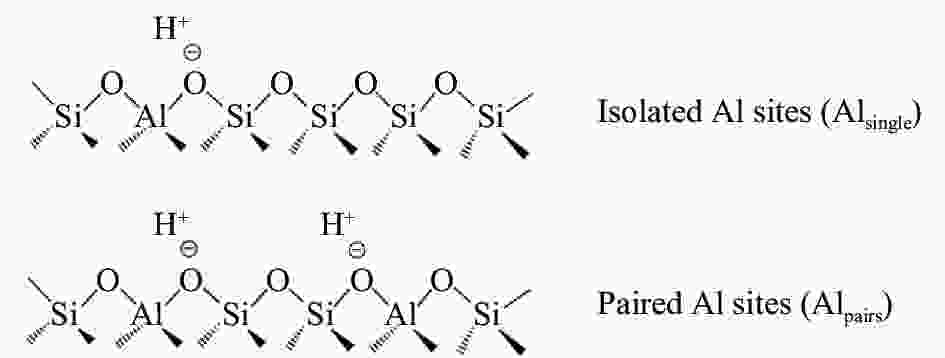
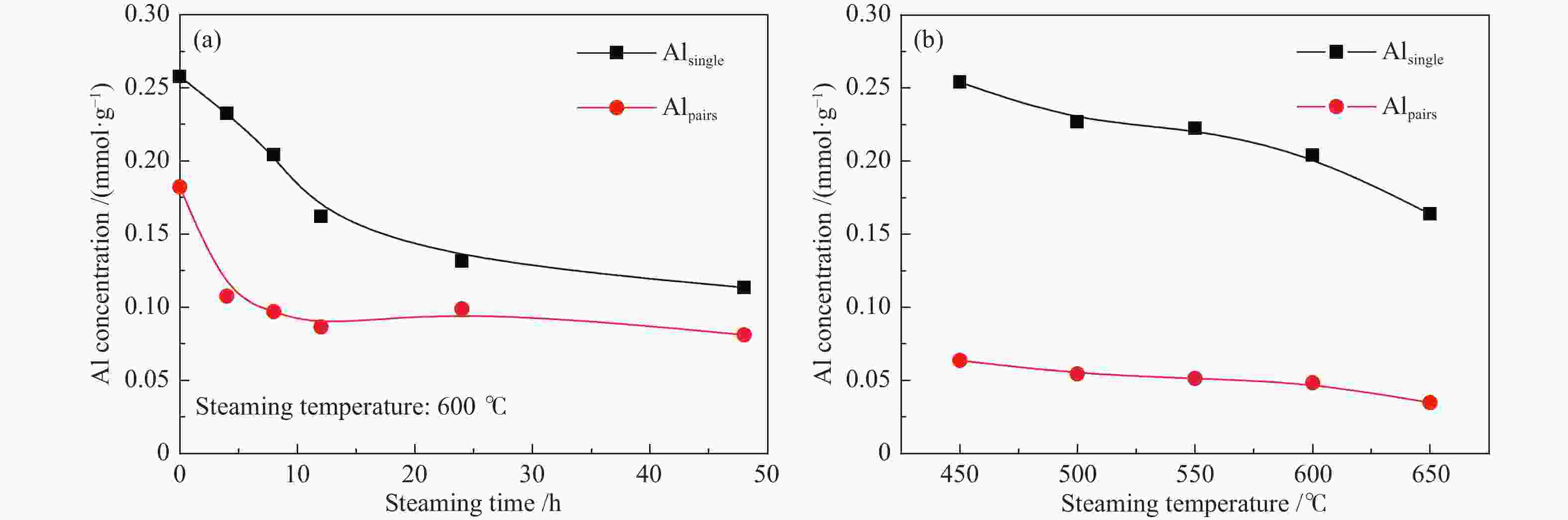
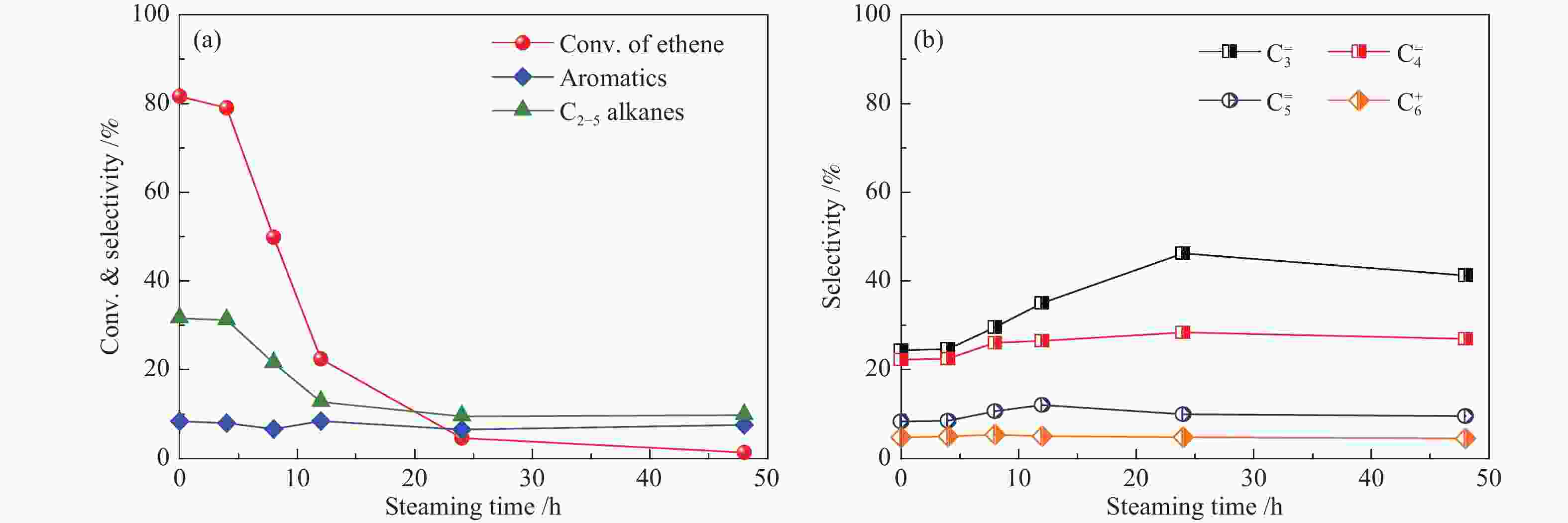
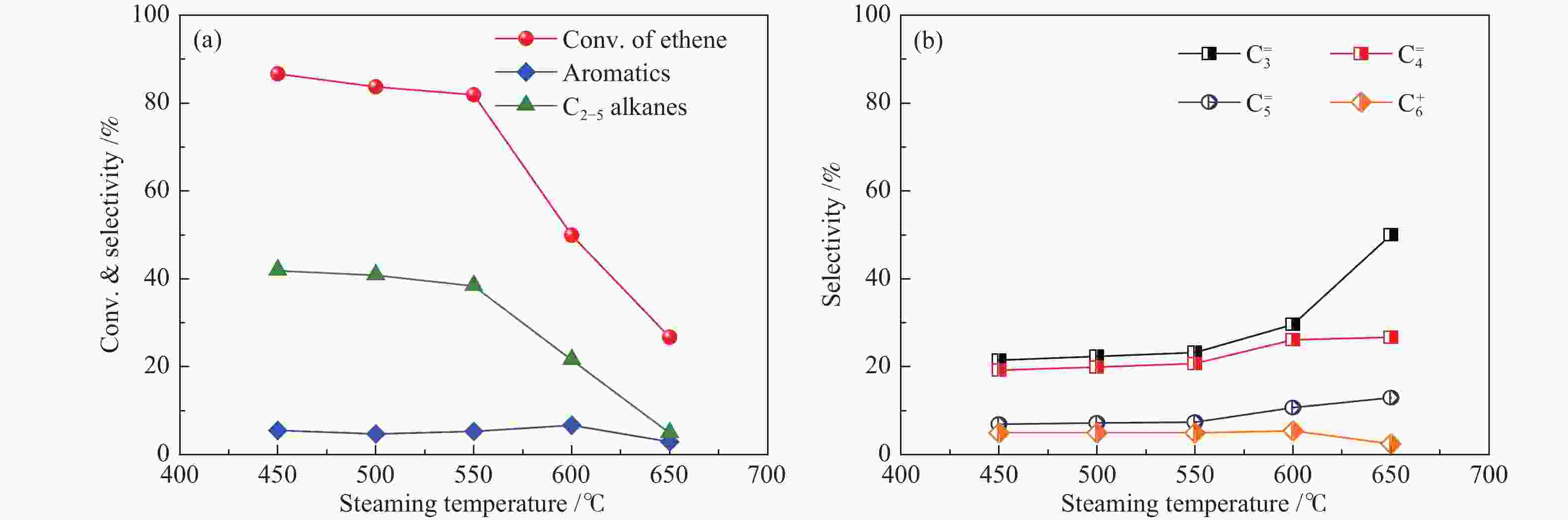
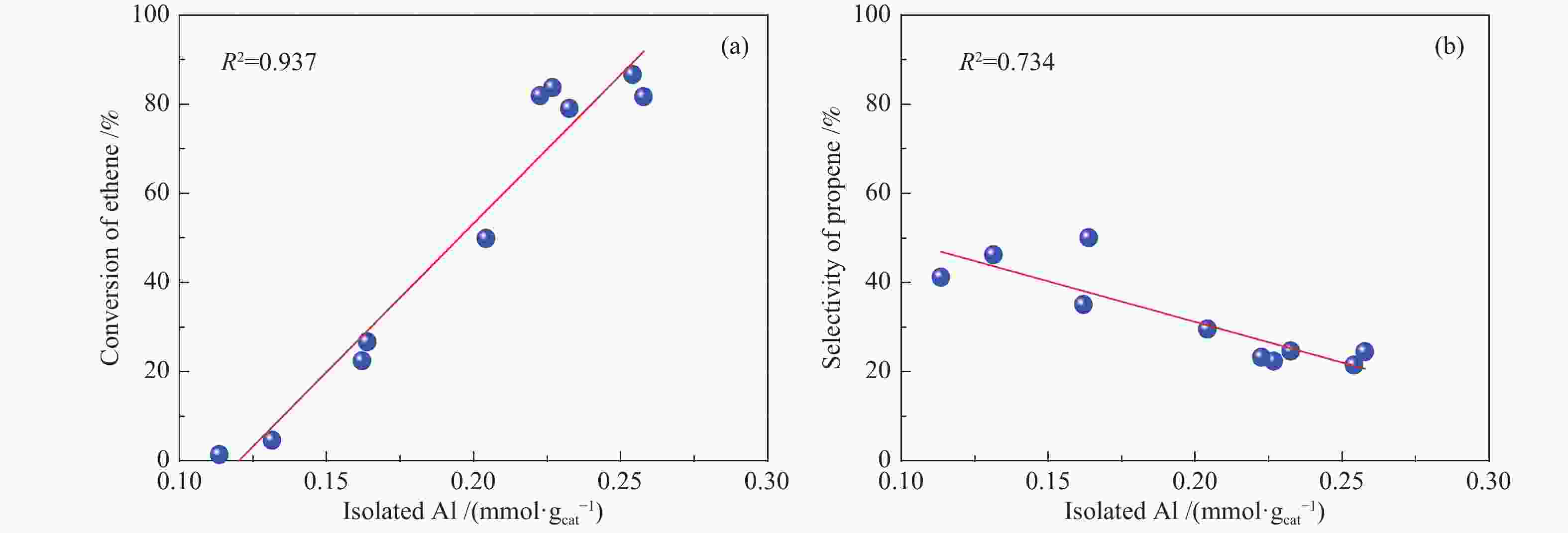
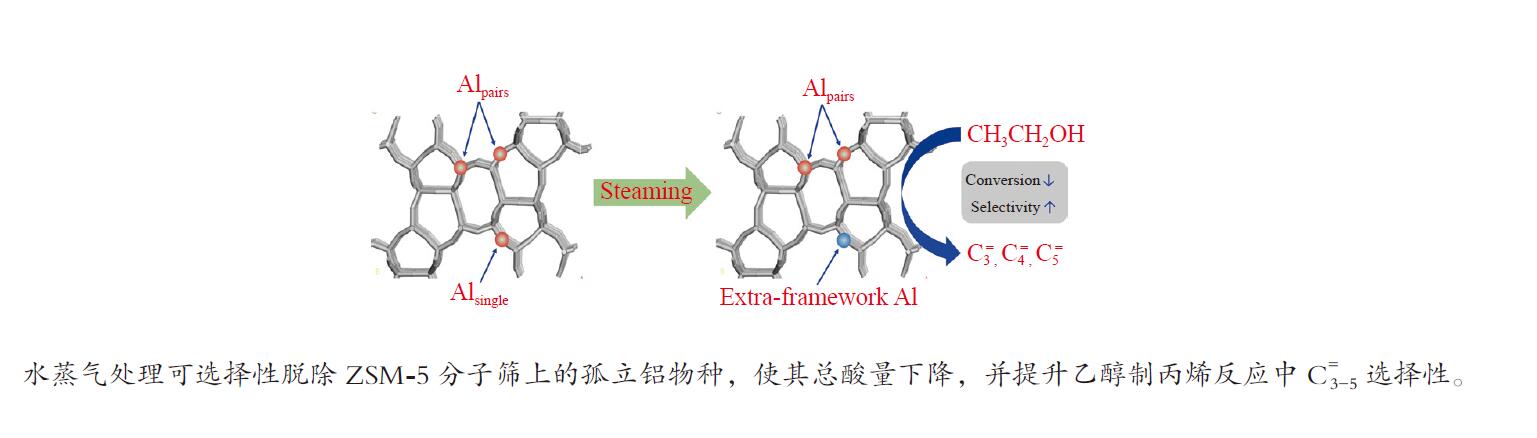
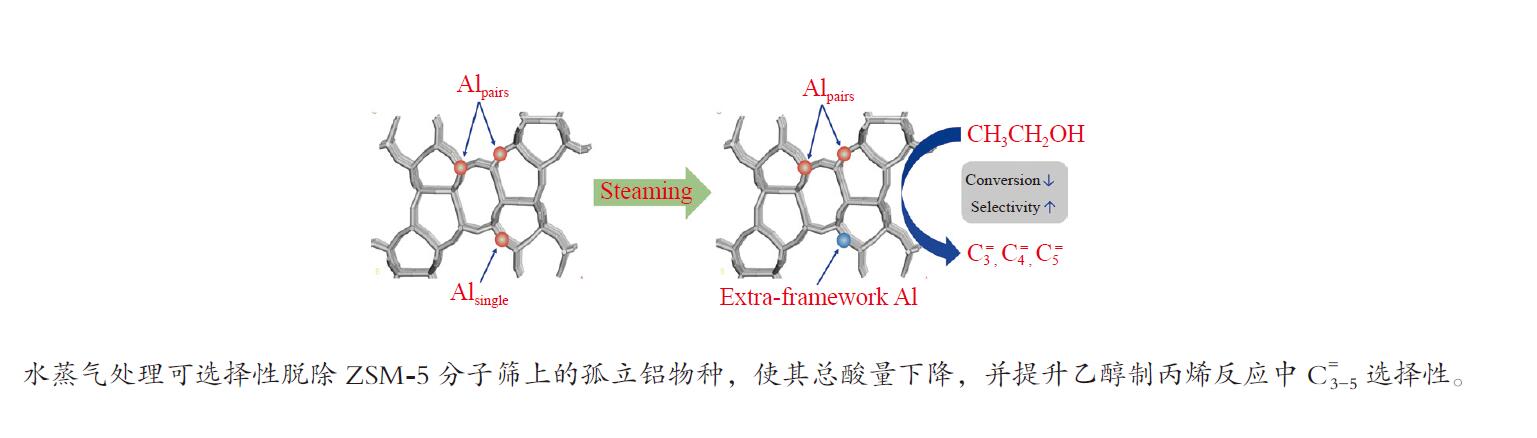
摘要: 采用水蒸气对ZSM-5分子筛进行后处理,系统考察了水蒸气处理时间和处理温度对分子筛孔结构、骨架铝分布、酸性及乙醇转化制丙烯的影响。研究结果表明,随着水蒸气处理时间的延长及处理温度的升高,分子筛结晶度和比表面积明显下降。27Al NMR及Co(Ⅱ)交换-ICP结果显示,在水蒸气作用下,分子筛骨架上孤立铝(Alsingle)物种被优先脱除,但临近铝(Alpairs)物种较为稳定。同时,吡啶吸附红外光谱结果显示,在水蒸气作用下,分子筛酸量和酸强度均显著降低。乙醇催化转化结果显示,随着水蒸气处理时间的延长和处理温度的升高,乙烯转化率和烷烃选择性迅速下降,但丙烯和丁烯等高碳烯烃的选择性明显提高。同时发现,乙烯转化率和孤立铝含量之间存在较好的正相关线性关系,而丙烯的生成则与孤立铝和邻近铝的共同作用有关。Abstract: The effects of steaming at varying times and temperatures on ZSM-5 pore structures, framework Al distribution, acid properties and ethanol to propene (ETP) catalytic performance were systematically studied in this study. The results show that the crystallinity and specific surface areas of steam treated ZSM-5 samples decrease with the increase treatment time and temperature. 27Al MAS NMR and Co(Ⅱ) exchange-ICP results show that the isolated framework Al species (Alsingle) can be preferentially removed from the zeolite framework, while the paired Al sites (Alpairs) remain relatively stable after steam treatment. The characterization of Py-IR reveals that the concentration of B acid sites and the acid strength are all declined with the steaming time or temperature increase. The catalytic results of ETP at 450 ℃ show that the sample after steaming gives improved selectivities to propene and butene at the expense of ethene conversion and alkanes selectivity relative to the unmodified zeolite. Besides, a good positive correlation between ethylene conversion and Alsingle concentration is found, whereas the propene formation is influenced by the combination effect of Alsingle and Alpairs sites.
-
Key words:
- ZSM-5 zeolite /
- steaming /
- dealumination /
- ethanol to propene /
- Al distribution
-
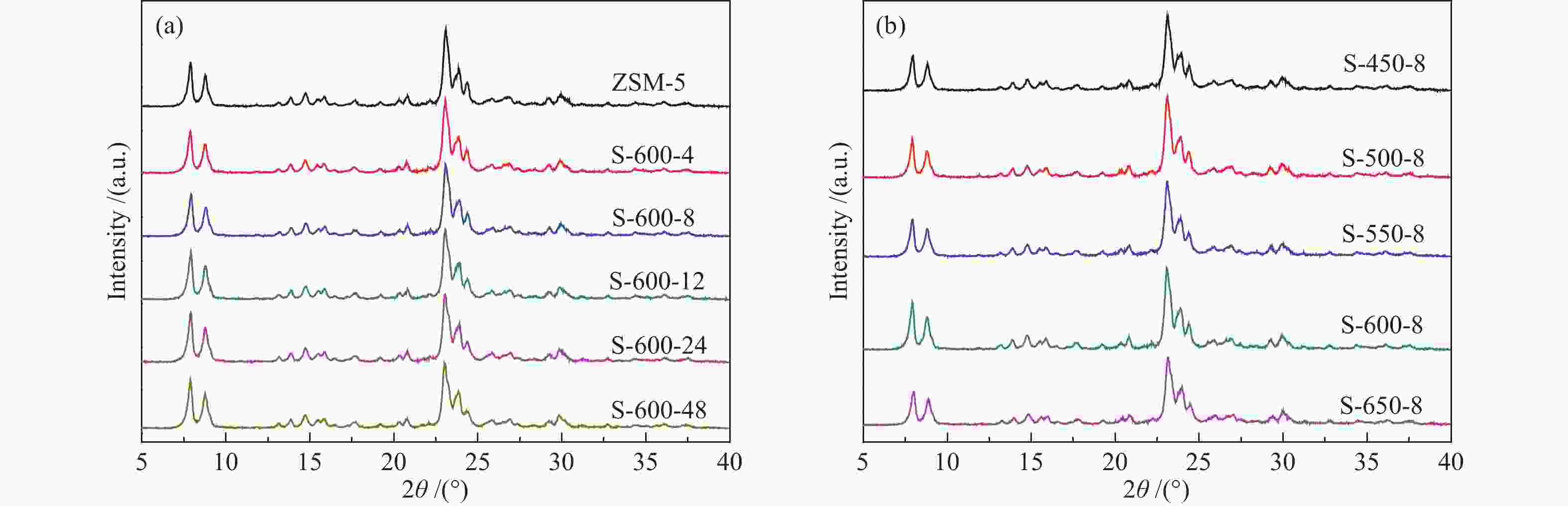
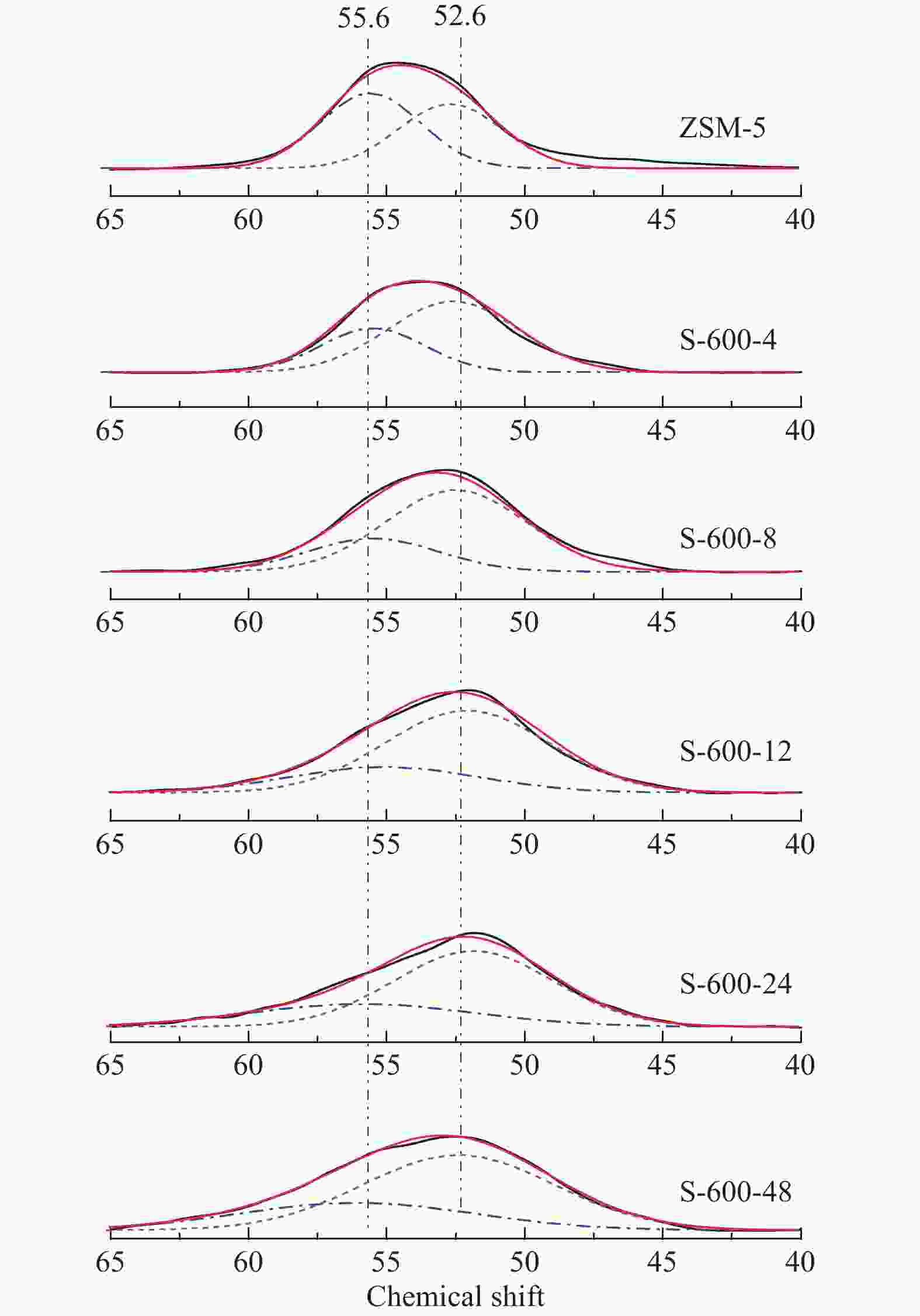
表 1 水蒸气处理时间及温度对ZSM-5分子筛织构性质的影响
Table 1 The effects of steaming time and temperature on textural properties of ZSM-5 samples
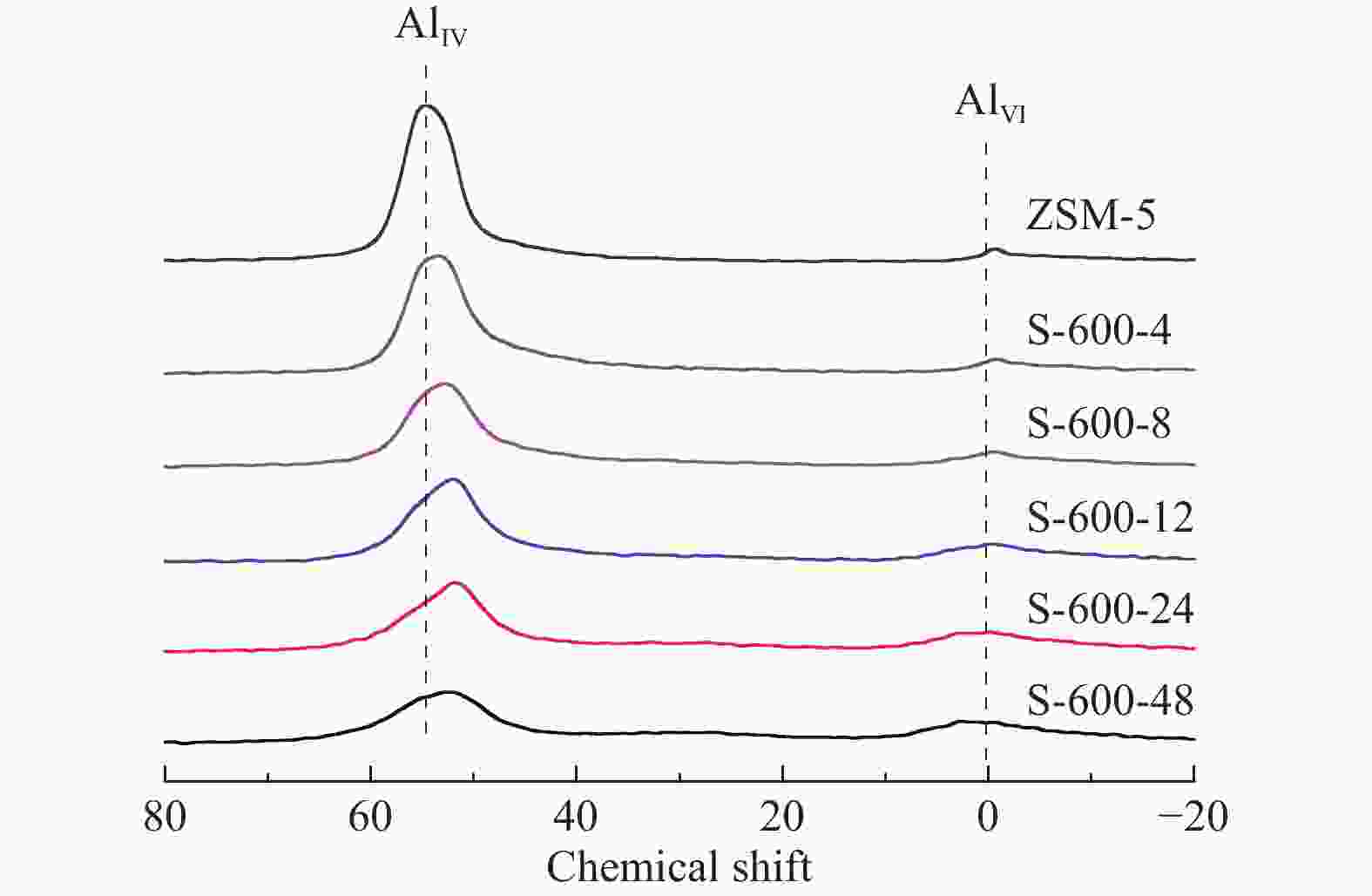
Sample Si/Ala Crystallinity/%b Surface area/(m2·g−1) Pore volume/(cm3·g−1) total micropore external total micropore ZSM-5 25 100 387 269 118 0.32 0.13 S-600-4 25 94 358 254 104 0.30 0.12 S-600-8 25 94 348 257 91 0.29 0.11 S-600-12 25 91 333 259 74 0.28 0.11 S-600-24 25 88 337 275 62 0.29 0.12 S-600-48 25 83 329 276 53 0.29 0.12 S-450-8 25 95 365 240 125 0.33 0.11 S-500-8 25 92 369 246 123 0.31 0.11 S-550-8 25 86 381 259 122 0.32 0.12 S-650-8 25 77 354 274 80 0.29 0.12 a: Determined by ICP-AES; b: The relative crystallinity of a sample determined by XRD was calculated by comparing its peak area in the 2θ range of 22°−25° to that of ZSM-5 (ZSM-5, assuming that it had a crystallinity of 100%) 表 2 27Al MAS NMR谱图拟合
Table 2 Results of deconvolution of 27Al MAS NMR spectra
Sample Relative peak areas/% 55.6
(chemical shift)52.6
(chemical shift)0
(chemical shift)ZSM-5 51 46 3 S-600-4 31 63 6 S-600-8 24 63 14 S-600-12 22 61 17 S-600-24 23 53 24 S-600-48 21 48 31 表 3 水蒸气处理后分子筛的酸性质
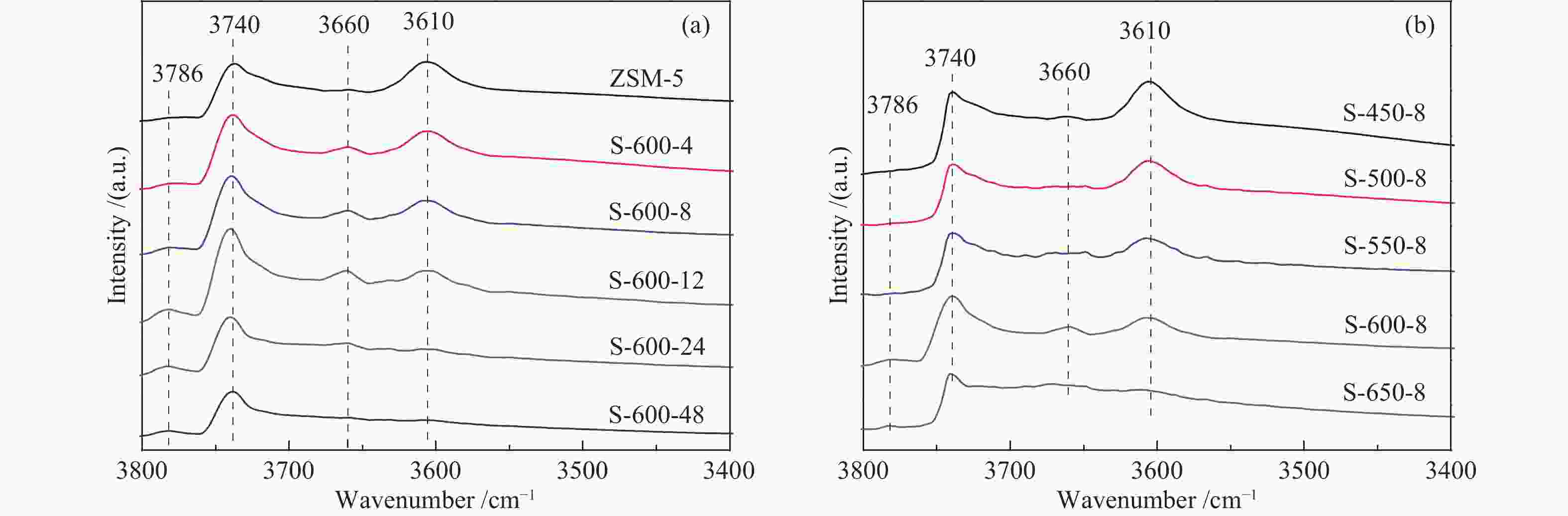
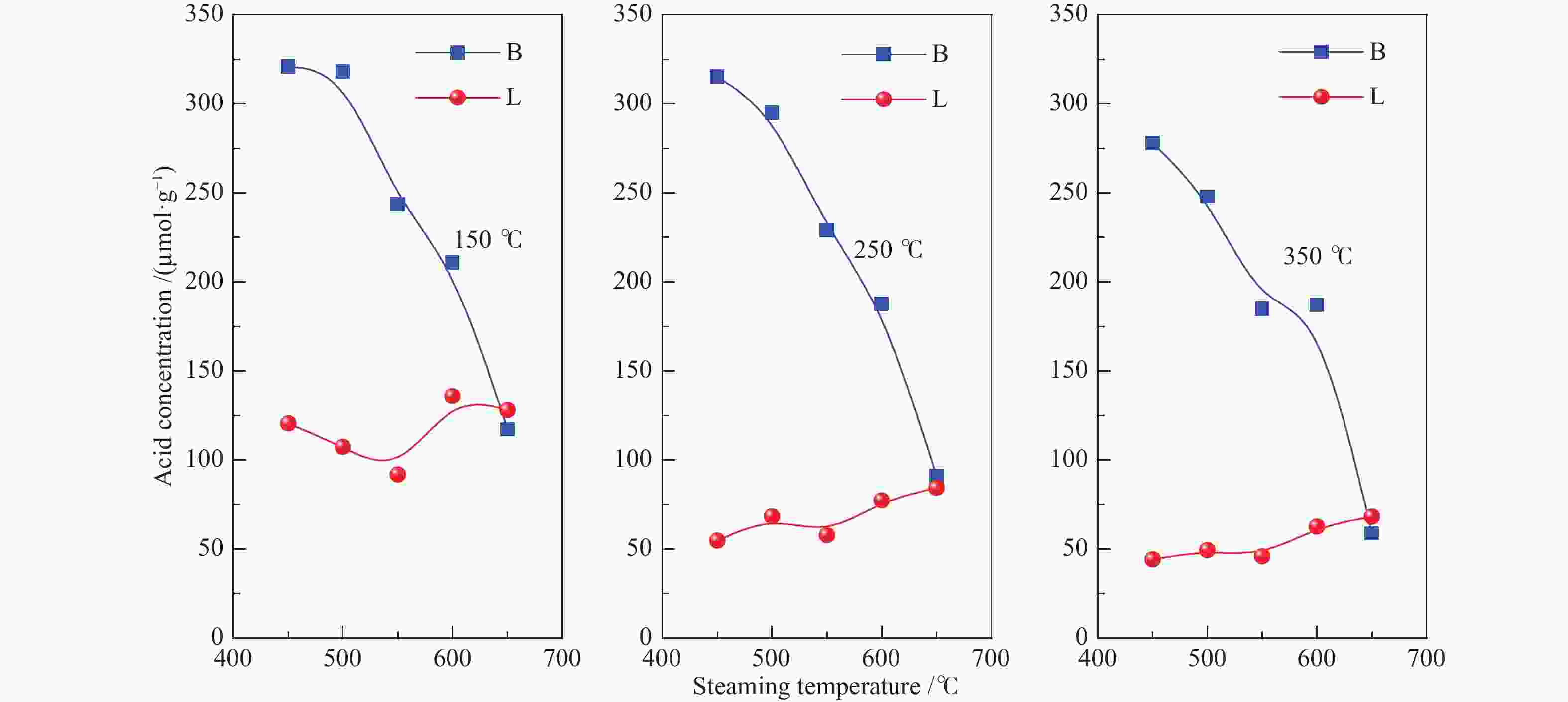
Table 3 Acidic properties of ZSM-5 samples after steaming
Sample Acidity by type/(μmol·g−1) Brønsted acid distributiona External Acidity/(μmol·g−1) B L B/L strong medium weak B proportion/% ZSM-5 327 88 3.7 82 10 8 28 8 S-600-4 230 118 2.0 83 16 1 31 14 S-600-8 211 136 1.6 89 0 11 38 18 S-600-12 176 149 1.2 66 15 19 39 22 S-600-24 115 139 0.8 49 45 5 29 25 S-600-48 78 78 1.0 49 26 25 30 39 S-450-8 321 121 2.7 87 12 2 22 7 S-500-8 318 107 3.0 78 15 7 24 8 S-550-8 244 92 2.7 76 18 6 38 16 S-650-8 117 128 0.9 50 28 22 9 8 a: Brønsted acidic sites (BAS) distribution were calculated from the amounts of BAS determined by Py-FTIR at different temperatures (weak, pyridine desorbed at 150−250 ℃; medium, pyridine desorbed at 250−350 ℃; strong, pyridine remained at 350 ℃) -
[1] 翟岩亮, 张少龙, 张络明, 尚蕴山, 王文轩, 宋宇, 姜彩彤, 巩雁军. 不同B, Al分布对ZSM-5分子筛的甲醇制丙烯反应性能的影响[J]. 物理化学学报,2019,35(11):1248−1258. doi: 10.3866/PKU.WHXB201901062ZHAI Yan-liang, ZHANG Shao-long, ZHANG Luo-ming, SHANG Yun-shan, WANG Wen-xuan, SONG Yu, JIANG Cai-tong, GONG Yan-jun. Effect of B and Al distribution in ZSM-5 zeolite on methanol to propylene reaction performance[J]. Acta Phys-Chim Sin,2019,35(11):1248−1258. doi: 10.3866/PKU.WHXB201901062 [2] WANG S, ZHANG L, LI S Y, QIN Z F, SHI D Z, HE S P, YUAN K, WANG P F, ZHAO T S, FAN S B, DONG M, LI J F, FAN W B, WANG J G. Tuning the siting of aluminum in ZSM-11 zeolite and regulating its catalytic performance in the conversion of methanol to olefins[J]. J Catal,2019,377:81−97. doi: 10.1016/j.jcat.2019.07.028 [3] 申文杰. ZSM-5分子筛强弱酸梯度分布增强其MTP反应的转化效率[J]. 物理化学学报,2019,35(11):1179−1182. doi: 10.3866/PKU.WHXB201906068SHENG Wen-jie. The strong and weak acid distributions of ZSM-5 zeolite promote its MTP reaction performance[J]. Acta Phys-Chim Sin,2019,35(11):1179−1182. doi: 10.3866/PKU.WHXB201906068 [4] BECERRA J, FIGUEREDO M, COBO M. Thermodynamic and economic assessment of the production of light olefins from bioethanol[J]. J Environ Chem Eng,2017,5:1554−1564. doi: 10.1016/j.jece.2017.02.035 [5] ZHANG L, WANG S, SHI D Z, QIN Z F, WANG P F, WANG G F, LI J F, DONG M, FAN W B, WANG J G. Methanol to olefins over H-RUB-13 zeolite: Regulation of framework aluminum siting and acid density and their relationship to the catalytic performance[J]. Catal Sci Technol,2020,10:1835−1847. doi: 10.1039/C9CY02419K [6] OIKAWA H, SHIBATA Y, INAZU K, IWASE Y, MURAI K, HYODO S, KOBAYASHI G, BABA T. Highly selective conversion of ethene to propene over SAPO-34 as a solid acid catalyst[J]. Appl Catal A: Gen,2006,312:181−185. doi: 10.1016/j.apcata.2006.06.045 [7] DAI W L, SUN X M, TANG B, WU G J, LI L D, GUAN N J, HUNGER M. Verifying the mechanism of the ethene-to-propene conversion on zeolite H-SSZ-13[J]. J Catal,2014,314:10−20. doi: 10.1016/j.jcat.2014.03.006 [8] RODEMERCK U, KONDRATENKO E V, STOYANOVA M, LINKE D. Study of reaction network of the ethylene-to-propene reaction by means of isotopically labelled reactants[J]. J Catal,2020,389:317−327. doi: 10.1016/j.jcat.2020.06.009 [9] RAMASAMY K K, WANG Y. Ethanol conversion to hydrocarbons on HZSM-5: Effect of reaction conditions and Si/Al ratio on the product distributions[J]. Catal Today,2014,237:89−99. doi: 10.1016/j.cattod.2014.02.044 [10] FURUMOTO Y, HARADA Y, TSUNOJI N, TAKAHASHI A, FUJITANI T, IDE Y, SADAKANE M, SANO T. Effect of acidity of ZSM-5 zeolite on conversion of ethanol to propylene[J]. Appl Catal A: Gen,2011,399:262−267. doi: 10.1016/j.apcata.2011.04.009 [11] TAKAHASHI A, XIA W, NAKAMURA I, SHIMADA H, FUJITANI T. Effects of added phosphorus on conversion of ethanol to propylene over ZSM-5 catalysts[J]. Appl Catal A: Gen,2012,423−424:162−167. doi: 10.1016/j.apcata.2012.02.029 [12] HUANGFU J J, MAO D S, ZHAI X L, GUO Q S. Remarkably enhanced stability of HZSM-5 zeolite co-modified with alkaline and phosphorous for the selective conversion of bio-ethanol to propylene[J]. Appl Catal A: Gen,2016,520:99−104. doi: 10.1016/j.apcata.2016.04.016 [13] ZHANG S L, GONG Y J, ZHANG L L, LIU Y S, DOU T, XU J, DENG F. Hydrothermal treatment on ZSM-5 extrudates catalyst for methanol to propylene reaction: Finely tuning the acidic property[J]. Fuel Process Technol,2015,129:130−138. doi: 10.1016/j.fuproc.2014.09.006 [14] ARAMBURO L R, RUIZ-MARTINEZ J, SOMMER L, ARSTAD B, BUITRAGO-SIERRA R, SEPULVEDA-ESCRIBANO A, ZANDBERGEN H W, OLSBYE U, DE GROOT F M F, WECKHUYSEN B M. X-Ray Imaging of SAPO-34 molecular sieves at the nanoscale: Influence of steaming on the methanol-to-hydrocarbons reaction[J]. ChemCatChem,2013,5:1386−1394. doi: 10.1002/cctc.201200670 [15] NIU X J, GAO J, WANG K, MIAO Q, DONG M, WANG G F, FAN W B, QIN Z F, WANG J G. Influence of crystal size on the catalytic performance of H-ZSM-5 and Zn/H-ZSM-5 in the conversion of methanol to aromatics[J]. Fuel Process Technol,2017,157:99−107. doi: 10.1016/j.fuproc.2016.12.006 [16] WANG S, LI S Y, ZHANG L, QIN Z F, DONG M, LI J F, WANG J G, FAN W B. Insight into the effect of incorporation of boron into ZSM-11 on its catalytic performance for conversion of methanol to olefins[J]. Catal Sci Technol,2017,7:4766−4779. doi: 10.1039/C7CY01428G [17] DEDECEK J, TABOR E, SKLENAK S. Tuning the Aluminum Distribution in Zeolites to Increase their Performance in Acid-Catalyzed Reactions[J]. ChemSusChem,2018,11:1−22. doi: 10.1002/cssc.201702374 [18] PASHKOVA V, KLEIN P, DEDECEK J, TOKAROVA V, WICHTERLOVA B. Incorporation of Al at ZSM-5 hydrothermal synthesis. Tuning of Al pairs in the framework[J]. Microporous Mesoporous Mater,2015,202:138−146. doi: 10.1016/j.micromeso.2014.09.056 [19] ONG L H, DOMOK M, OLINDO R, VAN VEEN A C, LERCHER J A. Dealumination of HZSM-5 via steam-treatment[J]. Microporous Mesoporous Mater,2012,164:9−20. doi: 10.1016/j.micromeso.2012.07.033 [20] TEKETEL S, OLSBYE U, LILLERUD K P, BEATO P, SVELLE S. Co-conversion of methanol and light alkenes over acidic zeolite catalyst H-ZSM-22: Simulated recycle of non-gasoline range products[J]. Appl Catal A: Gen,2015,494:68−76. doi: 10.1016/j.apcata.2015.01.035 [21] WU W Z, GUO W Y, XIAO W D, LUO M. Dominant reaction pathway for methanol conversion to propene over high silicon H-ZSM-5[J]. Chem Eng Sci,2011,66:4722−4732. doi: 10.1016/j.ces.2011.06.036 [22] ARAMBURO L R, DE SMIT E, ARSTAD B, VAN SCHOONEVELD M M, SOMMER L, JUHIN A, YOKOSAWA T, ZANDBERGEN H W, OLSBYE U, DE GROOT F M, WECKHUYSEN B M. X-ray imaging of zeolite particles at the nanoscale: Influence of steaming on the state of aluminum and the methanol-to-olefin reaction[J]. Angew Chem Int Ed,2012,51:3616−3619. doi: 10.1002/anie.201109026 [23] NIU X J, GAO J, MIAO Q, DONG M, WANG G F, FAN W B, QIN Z F, WANG J G. Influence of preparation method on the performance of Zn-containing HZSM-5 catalysts in methanol-to-aromatics[J]. Microporous Mesoporous Mater,2014,197:252−261. doi: 10.1016/j.micromeso.2014.06.027 [24] LI C G, VIDAL-MOYA A, MIGUEL P J, DEDECEK J, BORONAT M, CORMA A. Selective introduction of acid sites in different confined positions in ZSM-5 and its catalytic implications[J]. ACS Catal,2018,8:7688−7697. doi: 10.1021/acscatal.8b02112 [25] MAIER S M, JENTYS A, LERCHER J A. Steaming of zeolite BEA and its effect on acidity: A comparative NMR and IR spectroscopic study[J]. J Phys Chem C,2011,115:8005−8013. doi: 10.1021/jp108338g [26] BARBERA K, BONINO F, BORDIGA S, JANSSENS T V W, BEATO P. Structure-deactivation relationship for ZSM-5 catalysts governed by framework defects[J]. J Catal,2011,280:196−205. doi: 10.1016/j.jcat.2011.03.016 [27] SAZAMA P, WICHTERLOVA B, DEDECEK J, TVARUZKOVA Z, MUSILOVA Z, PALUMBO L, SKLENAK S, GONSIOROVA O. FTIR and 27Al MAS NMR analysis of the effect of framework Al- and Si-defects in micro- and micro-mesoporous H-ZSM-5 on conversion of methanol to hydrocarbons[J]. Microporous Mesoporous Mater,2011,143:87−96. doi: 10.1016/j.micromeso.2011.02.013 [28] LIANG T Y, CHEN J L, QIN Z F, LI J F, WANG P F, WANG S, WANG G F, DONG M, FAN W B, WANG J G. Conversion of methanol to olefins over H-ZSM-5 zeolite: Reaction pathway is related to the framework aluminum siting[J]. ACS Catal,2016,6:7311−7325. doi: 10.1021/acscatal.6b01771 [29] ALMUTAIRI S M T, MEZARI B, PIDKO E A, MAGUSIN P C M M, HENSEN E J M. Influence of steaming on the acidity and the methanol conversion reaction of HZSM-5 zeolite[J]. J Catal,2013,307:194−203. doi: 10.1016/j.jcat.2013.07.021 -




 下载:
下载: