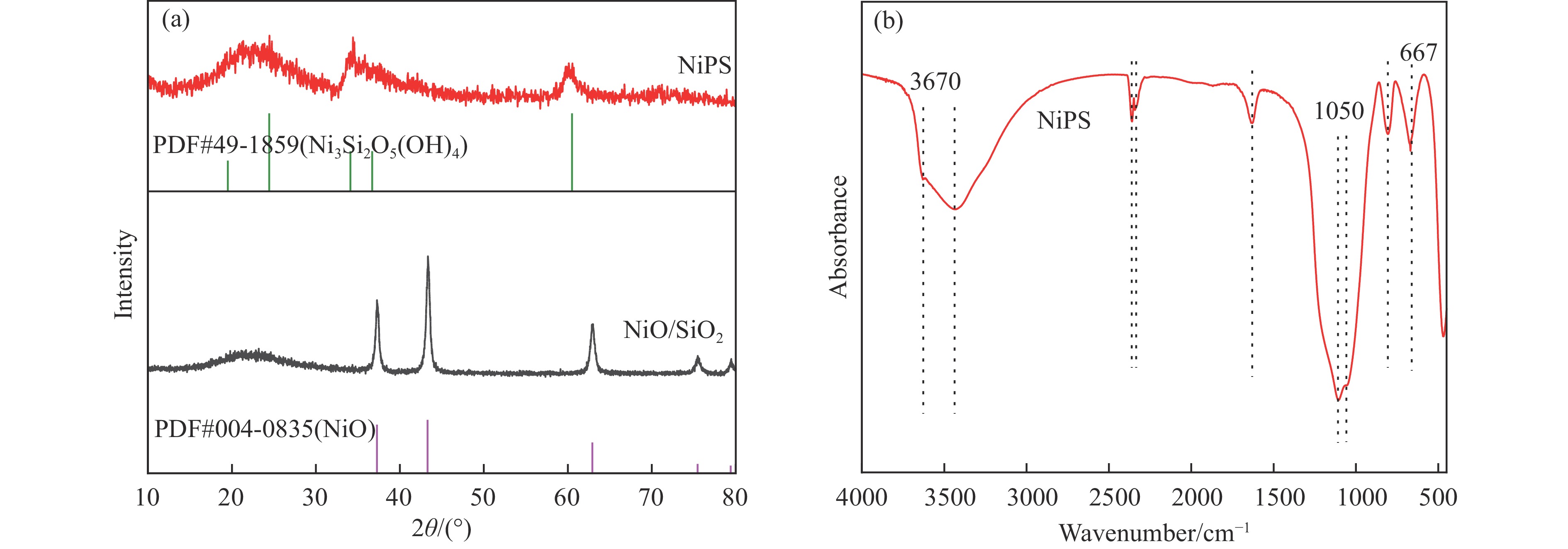
优先发表栏目展示本刊经同行评议确定正式录用的文章,这些文章目前处在编校过程,尚未确定卷期及页码,但可以根据DOI进行引用。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60477-9
摘要:
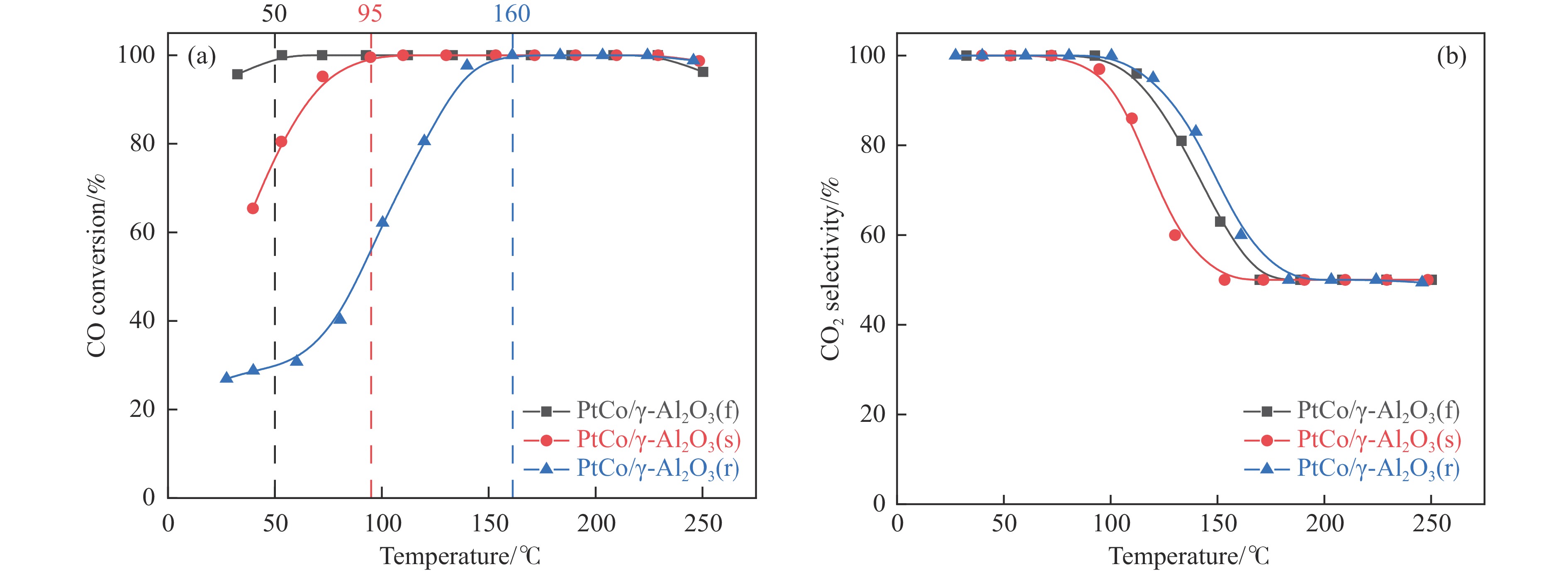
高效加氢脱氧催化剂的开发是生物油提质升级的关键。以介孔SiO2为载体,在Ni2P活性相基础上,通过Co掺杂制备了CoxP-Ni2P双金属位点与酸性位点并存的CoxP-Ni2P/SiO2-y催化剂(y为初始P/(Ni+Co)物质的量比),采用XRD、BET、XPS、H2-TPR、NH3-TPD、Py-FTIR和TEM技术表征了催化剂的结构和化学性质,并以间甲酚为模型化合物,考察了Co掺杂以及P/M物质的量比对Ni2P/SiO2催化剂加氢脱氧性能的影响。结果表明,Co的掺杂不仅新增了活性位点CoxP,还优化了Ni2P的电子结构,进而提高了催化剂的加氢脱氧(HDO)活性。在CoxP-Ni2P/SiO2-y催化剂中,P/M物质的量比为0.5的CoxP-Ni2P/SiO2-0.5催化性能最好,在275 ℃,2 MPa,1 h的反应条件下,间甲酚转化率达到98.7%,对脱氧产物甲基环己烷(MCH)的选择性达到95.6%,且CoxP-Ni2P/SiO2-y催化剂上的HDO反应过程以先加氢后脱氧(HYD)路径为主。
高效加氢脱氧催化剂的开发是生物油提质升级的关键。以介孔SiO2为载体,在Ni2P活性相基础上,通过Co掺杂制备了CoxP-Ni2P双金属位点与酸性位点并存的CoxP-Ni2P/SiO2-y催化剂(y为初始P/(Ni+Co)物质的量比),采用XRD、BET、XPS、H2-TPR、NH3-TPD、Py-FTIR和TEM技术表征了催化剂的结构和化学性质,并以间甲酚为模型化合物,考察了Co掺杂以及P/M物质的量比对Ni2P/SiO2催化剂加氢脱氧性能的影响。结果表明,Co的掺杂不仅新增了活性位点CoxP,还优化了Ni2P的电子结构,进而提高了催化剂的加氢脱氧(HDO)活性。在CoxP-Ni2P/SiO2-y催化剂中,P/M物质的量比为0.5的CoxP-Ni2P/SiO2-0.5催化性能最好,在275 ℃,2 MPa,1 h的反应条件下,间甲酚转化率达到98.7%,对脱氧产物甲基环己烷(MCH)的选择性达到95.6%,且CoxP-Ni2P/SiO2-y催化剂上的HDO反应过程以先加氢后脱氧(HYD)路径为主。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60474-3
摘要:
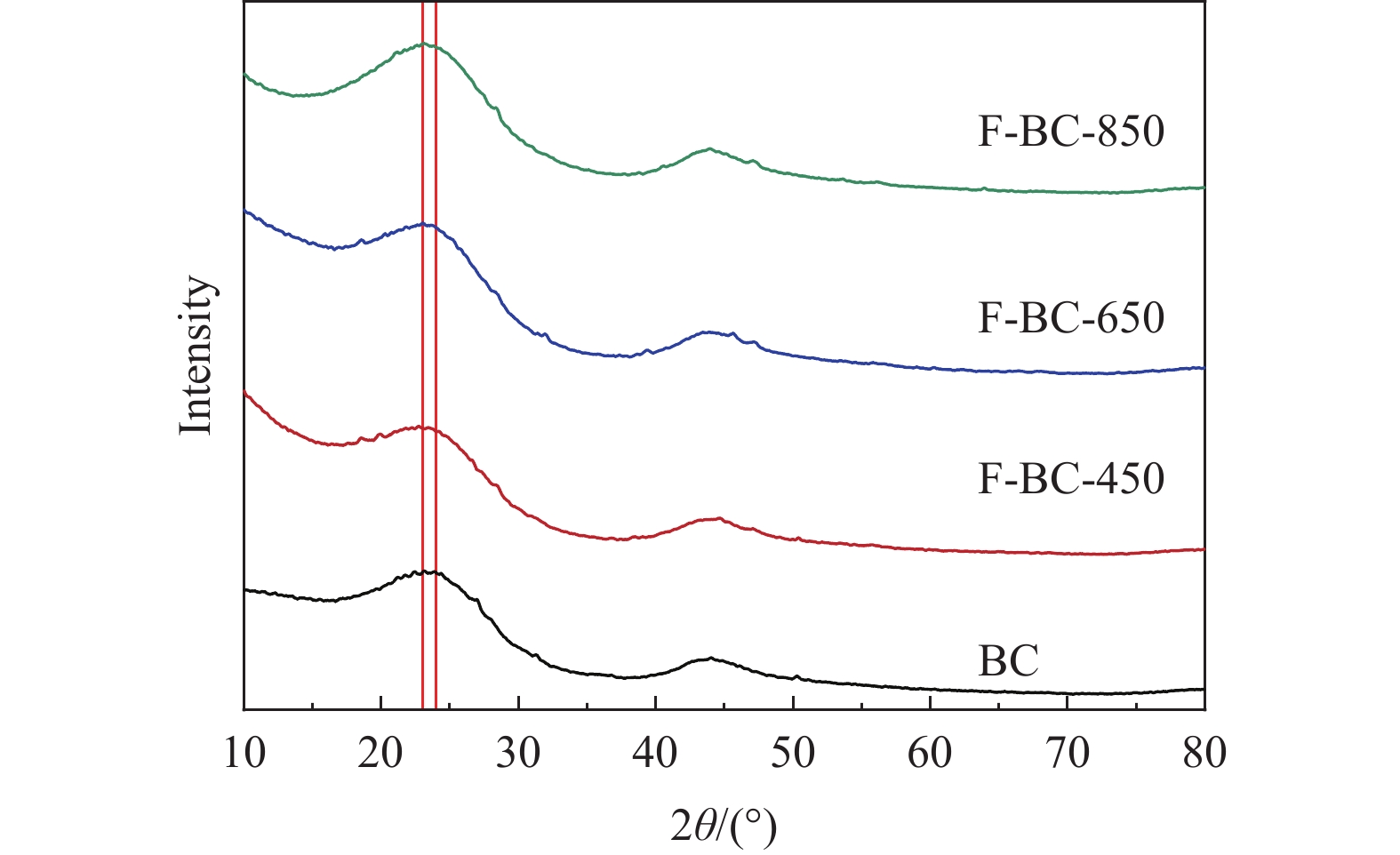
面临着“双碳”目标的约束,氢能,尤其是电解水制氢技术受到了广泛的关注。然而,高能耗是制约该技术产业化的关键问题之一。基于此,提出“牺牲阳极碳辅助电解水制氢”的策略,利用碳氧化反应(COR)替代析氧反应(OER),实现能耗的大幅降低。具体是利用两步碳化法制备出F掺杂生物炭(记作F-BC-850),采用XRD、SEM-EDS、热重分析、XPS等表征手段解析F-BC-850的结构特征;通过电化学方法阐明F-BC-850与制氢性能间的构效关系。结果表明,在0.5 mol/L H2SO4溶液中,10 mA/cm2下,其能耗是传统Pt片电极的57.9%;表征结果发现,氟化铵热解产生的HF刻蚀生物炭,能够形成活性较强的C−F键,进而提升了碳氧化性能。F物种的引入改变了碳结构,促进H2O分子产生*OH,并强化了碳原子对*OH的吸附能力。本工作可为碳辅助电解水制氢高效产H2和生物质高值化利用提供科学依据。
面临着“双碳”目标的约束,氢能,尤其是电解水制氢技术受到了广泛的关注。然而,高能耗是制约该技术产业化的关键问题之一。基于此,提出“牺牲阳极碳辅助电解水制氢”的策略,利用碳氧化反应(COR)替代析氧反应(OER),实现能耗的大幅降低。具体是利用两步碳化法制备出F掺杂生物炭(记作F-BC-850),采用XRD、SEM-EDS、热重分析、XPS等表征手段解析F-BC-850的结构特征;通过电化学方法阐明F-BC-850与制氢性能间的构效关系。结果表明,在0.5 mol/L H2SO4溶液中,10 mA/cm2下,其能耗是传统Pt片电极的57.9%;表征结果发现,氟化铵热解产生的HF刻蚀生物炭,能够形成活性较强的C−F键,进而提升了碳氧化性能。F物种的引入改变了碳结构,促进H2O分子产生*OH,并强化了碳原子对*OH的吸附能力。本工作可为碳辅助电解水制氢高效产H2和生物质高值化利用提供科学依据。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0006
摘要:
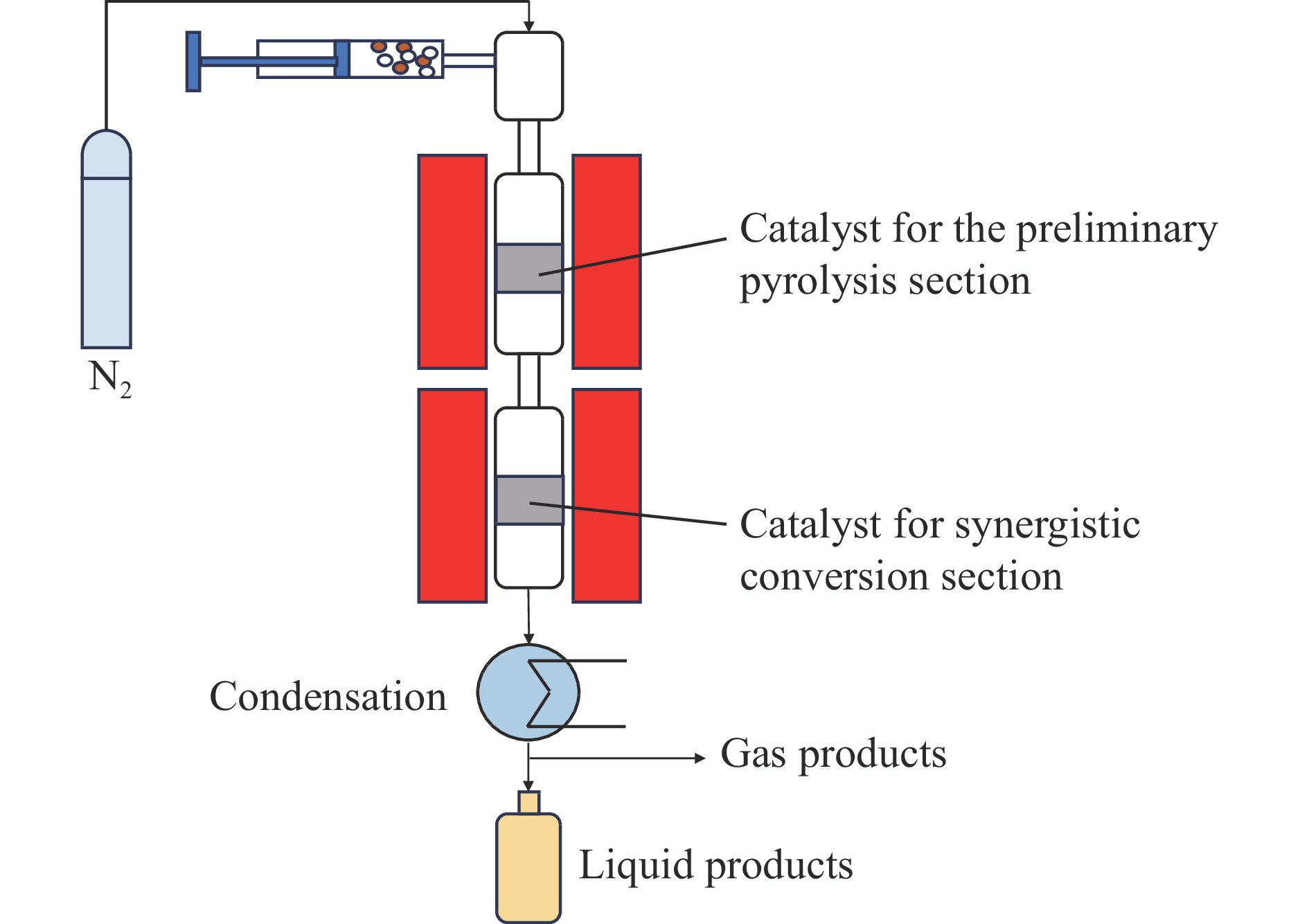
本研究设计了两段式的热解过程,以松木木屑和低密度聚乙烯(LDPE)协同转化制备液体芳烃。催化热解过程所得液体产物自然分层为有机相和水相,当使用两段式热解体系时,其有机相产率比传统单段热解提高11.4%。通过木屑与LDPE单独热解与共热解对比发现,木屑与LDPE共热解过程中存在基于氢转移的协同作用,能够促进液体芳烃的形成,且该协同作用在两段式过程中更显著。原料中LDPE所占比例会影响协同效应强度;在木屑和LDPE质量比为1∶1时协同效应最显著,所得有机相产率达到47.2%,其中,芳烃含量为93.8%。提高两段温度可以促进木屑和LDPE挥发分的析出并强化芳构化反应,但过高的温度会促进气体生成;当初步热解段温度在600 ℃,协同转化段温度在500 ℃时,共热解芳烃产率最高。碱性氧化物MgO可以促进木屑与LDPE的初步热解,使用MgO作为初步热解段催化剂对比ZrO2有机相产率提高5.2%,对协同转化段HZSM-5催化剂通过Ga2O3进行负载能够进一步提升协同转化效果,最终有机相产率达到51.5%,其中,芳烃含量达到98.9%。
本研究设计了两段式的热解过程,以松木木屑和低密度聚乙烯(LDPE)协同转化制备液体芳烃。催化热解过程所得液体产物自然分层为有机相和水相,当使用两段式热解体系时,其有机相产率比传统单段热解提高11.4%。通过木屑与LDPE单独热解与共热解对比发现,木屑与LDPE共热解过程中存在基于氢转移的协同作用,能够促进液体芳烃的形成,且该协同作用在两段式过程中更显著。原料中LDPE所占比例会影响协同效应强度;在木屑和LDPE质量比为1∶1时协同效应最显著,所得有机相产率达到47.2%,其中,芳烃含量为93.8%。提高两段温度可以促进木屑和LDPE挥发分的析出并强化芳构化反应,但过高的温度会促进气体生成;当初步热解段温度在600 ℃,协同转化段温度在500 ℃时,共热解芳烃产率最高。碱性氧化物MgO可以促进木屑与LDPE的初步热解,使用MgO作为初步热解段催化剂对比ZrO2有机相产率提高5.2%,对协同转化段HZSM-5催化剂通过Ga2O3进行负载能够进一步提升协同转化效果,最终有机相产率达到51.5%,其中,芳烃含量达到98.9%。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60473-1
摘要:
This study explores the controllable synthesis of CuAlO2 using copper hydroxide and pseudo-boehmite powders as raw materials via a simple solid-phase ball milling method, along with its catalytic performance investigation in methanol steam reforming (MSR). Various catalysts were prepared under different conditions, such as calcination temperature, calcination atmosphere, and heating rate. Characterization techniques including BET, XRD, XPS, SEM, and H2-TPR were employed to analyze the samples. The results revealed significant effects of calcination temperature on the phase compositions, specific surface area, reduction performance, and surface properties of the CA-T catalysts. Based on the findings, a synthesis route of CuAlO2 via the solid-phase method was proposed, highlighting the importance of high calcination temperature, nitrogen atmosphere, and low heating rate for CuAlO2 formation. Catalytic evaluation data demonstrated that CuAlO2 could catalyze MSR without pre-reduction, with the catalytic performance of CA-T catalysts being notably influenced by calcination temperature. Among the prepared catalysts, the CA-1100 catalyst exhibited the highest catalytic activity and stability. The findings of this study might be useful for the further study of the catalytic material for sustained release catalysis, including the synthesis of catalytic materials and the regulation of sustained release catalytic performance.
This study explores the controllable synthesis of CuAlO2 using copper hydroxide and pseudo-boehmite powders as raw materials via a simple solid-phase ball milling method, along with its catalytic performance investigation in methanol steam reforming (MSR). Various catalysts were prepared under different conditions, such as calcination temperature, calcination atmosphere, and heating rate. Characterization techniques including BET, XRD, XPS, SEM, and H2-TPR were employed to analyze the samples. The results revealed significant effects of calcination temperature on the phase compositions, specific surface area, reduction performance, and surface properties of the CA-T catalysts. Based on the findings, a synthesis route of CuAlO2 via the solid-phase method was proposed, highlighting the importance of high calcination temperature, nitrogen atmosphere, and low heating rate for CuAlO2 formation. Catalytic evaluation data demonstrated that CuAlO2 could catalyze MSR without pre-reduction, with the catalytic performance of CA-T catalysts being notably influenced by calcination temperature. Among the prepared catalysts, the CA-1100 catalyst exhibited the highest catalytic activity and stability. The findings of this study might be useful for the further study of the catalytic material for sustained release catalysis, including the synthesis of catalytic materials and the regulation of sustained release catalytic performance.
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0009
摘要:
随着全球能源和环境问题的日益严重,单原子催化剂兼具100%原子利用率和独特的电子结构,受到国内外研究机构的广泛关注。单原子催化剂商业化应用的关键在于其规模化制备及在重要工业反应的应用。近年来,火焰喷雾热解法是一种简单、高效、可规模化制备单原子催化剂的合成策略。本综述对单原子催化剂规模化制备方法和火焰喷雾热解制单原子催化剂的研究进展进行综合评述。总结了火焰喷雾热解制Mo、Pt、Pd、La、Zr基单原子催化剂在甲烷无氧转化、CO氧化、CO2加氢制甲醇和光催化反应的研究进展。相比于传统的湿式制备方法,火焰喷雾热解法所制单原子催化剂具有良好的高温结构稳定性、抗积炭性、各组分的物相及其空间分布可控等优势。对火焰喷雾热解制单原子催化剂的发展方向进行了展望。
随着全球能源和环境问题的日益严重,单原子催化剂兼具100%原子利用率和独特的电子结构,受到国内外研究机构的广泛关注。单原子催化剂商业化应用的关键在于其规模化制备及在重要工业反应的应用。近年来,火焰喷雾热解法是一种简单、高效、可规模化制备单原子催化剂的合成策略。本综述对单原子催化剂规模化制备方法和火焰喷雾热解制单原子催化剂的研究进展进行综合评述。总结了火焰喷雾热解制Mo、Pt、Pd、La、Zr基单原子催化剂在甲烷无氧转化、CO氧化、CO2加氢制甲醇和光催化反应的研究进展。相比于传统的湿式制备方法,火焰喷雾热解法所制单原子催化剂具有良好的高温结构稳定性、抗积炭性、各组分的物相及其空间分布可控等优势。对火焰喷雾热解制单原子催化剂的发展方向进行了展望。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60478-0
摘要:
采用溶剂热法合成载体含环磷腈聚合物(POM),再以Ni和Cu作为活性组分通过浸渍还原法负载到载体上,制备出Ni0.4Cu0.6/POM催化剂,并将其用于光催化氨硼烷(AB)水解制氢反应。通过TEM、XRD、XPS、FTIR、UV-vis DRS、PL、EIS等手段对催化剂进行表征,实验结果表明,Ni0.4Cu0.6/POM催化剂具有稳定的球形颗粒结构,在298 K、光照的条件下催化反应的TOF值为1461.0 h−1,是非光条件下的1.6倍,并且在经过20次循环后依然保持着良好的催化活性。
采用溶剂热法合成载体含环磷腈聚合物(POM),再以Ni和Cu作为活性组分通过浸渍还原法负载到载体上,制备出Ni0.4Cu0.6/POM催化剂,并将其用于光催化氨硼烷(AB)水解制氢反应。通过TEM、XRD、XPS、FTIR、UV-vis DRS、PL、EIS等手段对催化剂进行表征,实验结果表明,Ni0.4Cu0.6/POM催化剂具有稳定的球形颗粒结构,在298 K、光照的条件下催化反应的TOF值为1461.0 h−1,是非光条件下的1.6倍,并且在经过20次循环后依然保持着良好的催化活性。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0004
摘要:
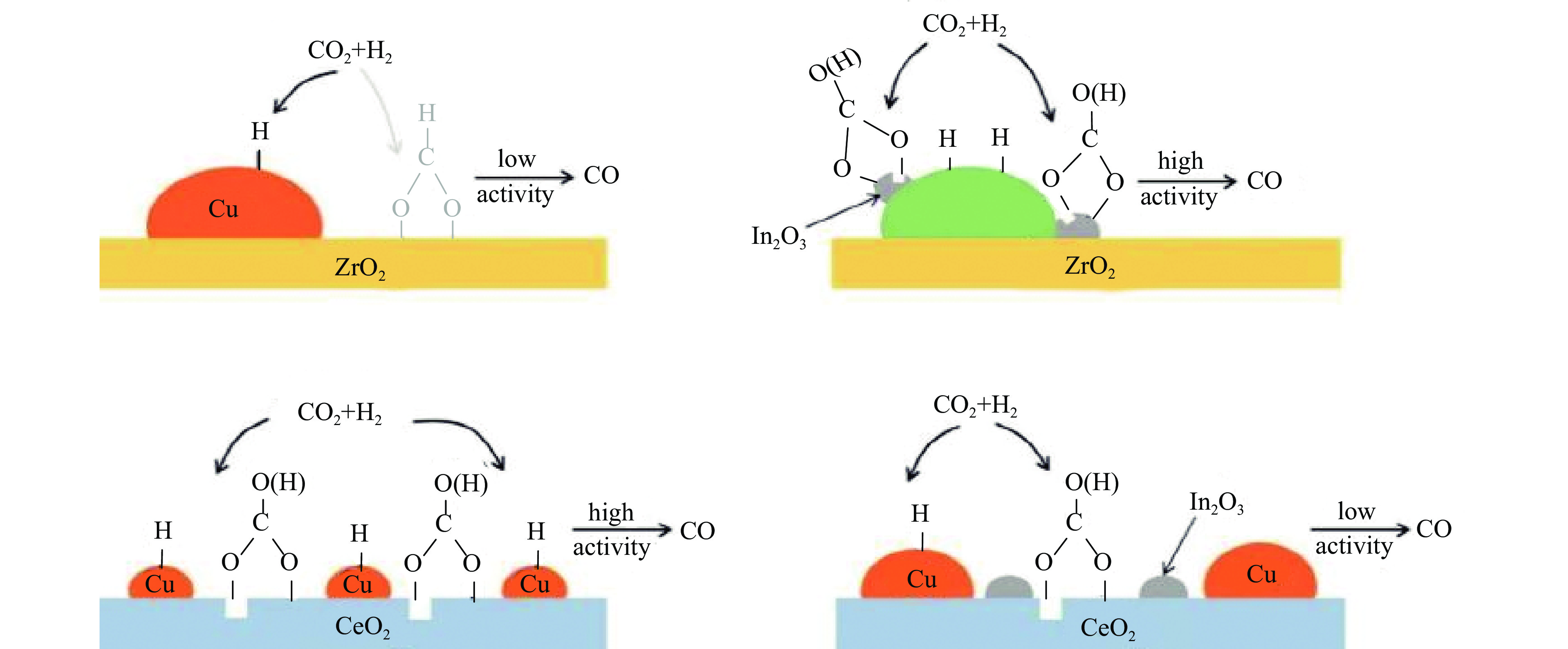
逆水煤气变换(RWGS)反应是降低碳排放和实现碳资源利用的有效途径。CO2经逆水煤气变换反应转化为CO,并通过费托工艺制得烯烃化产品和醇类燃料,对于改善环境以及改变未来能源结构具有重大意义。本工作首先总结了RWGS反应机理,依据CO2转化为CO路径中H2是否直接参还原反应,分为氧化还原机理和缔合机理,其中缔合机理包括甲酸盐、羧酸盐以及碳酸盐三种路径,并介绍了近年来关于RWGS反应机理研究的最新发现;其次综述了RWGS反应中的CO2和H2共进料热催化转化催化剂体系和化学循环催化剂体系的研究进展,分析讨论了Pt、Ni、Cu等金属基催化剂和金属碳化物、磷化物催化材料的RWGS反应性能,以及钙钛矿型储氧材料(OSM)在化学循环反应中的应用。最后,针对目前存在的问题和未来可能的解决方案进行了讨论与展望,以期为后续RWGS催化材料的研究提供一定的借鉴。
逆水煤气变换(RWGS)反应是降低碳排放和实现碳资源利用的有效途径。CO2经逆水煤气变换反应转化为CO,并通过费托工艺制得烯烃化产品和醇类燃料,对于改善环境以及改变未来能源结构具有重大意义。本工作首先总结了RWGS反应机理,依据CO2转化为CO路径中H2是否直接参还原反应,分为氧化还原机理和缔合机理,其中缔合机理包括甲酸盐、羧酸盐以及碳酸盐三种路径,并介绍了近年来关于RWGS反应机理研究的最新发现;其次综述了RWGS反应中的CO2和H2共进料热催化转化催化剂体系和化学循环催化剂体系的研究进展,分析讨论了Pt、Ni、Cu等金属基催化剂和金属碳化物、磷化物催化材料的RWGS反应性能,以及钙钛矿型储氧材料(OSM)在化学循环反应中的应用。最后,针对目前存在的问题和未来可能的解决方案进行了讨论与展望,以期为后续RWGS催化材料的研究提供一定的借鉴。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60466-4
摘要:
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0002
摘要:
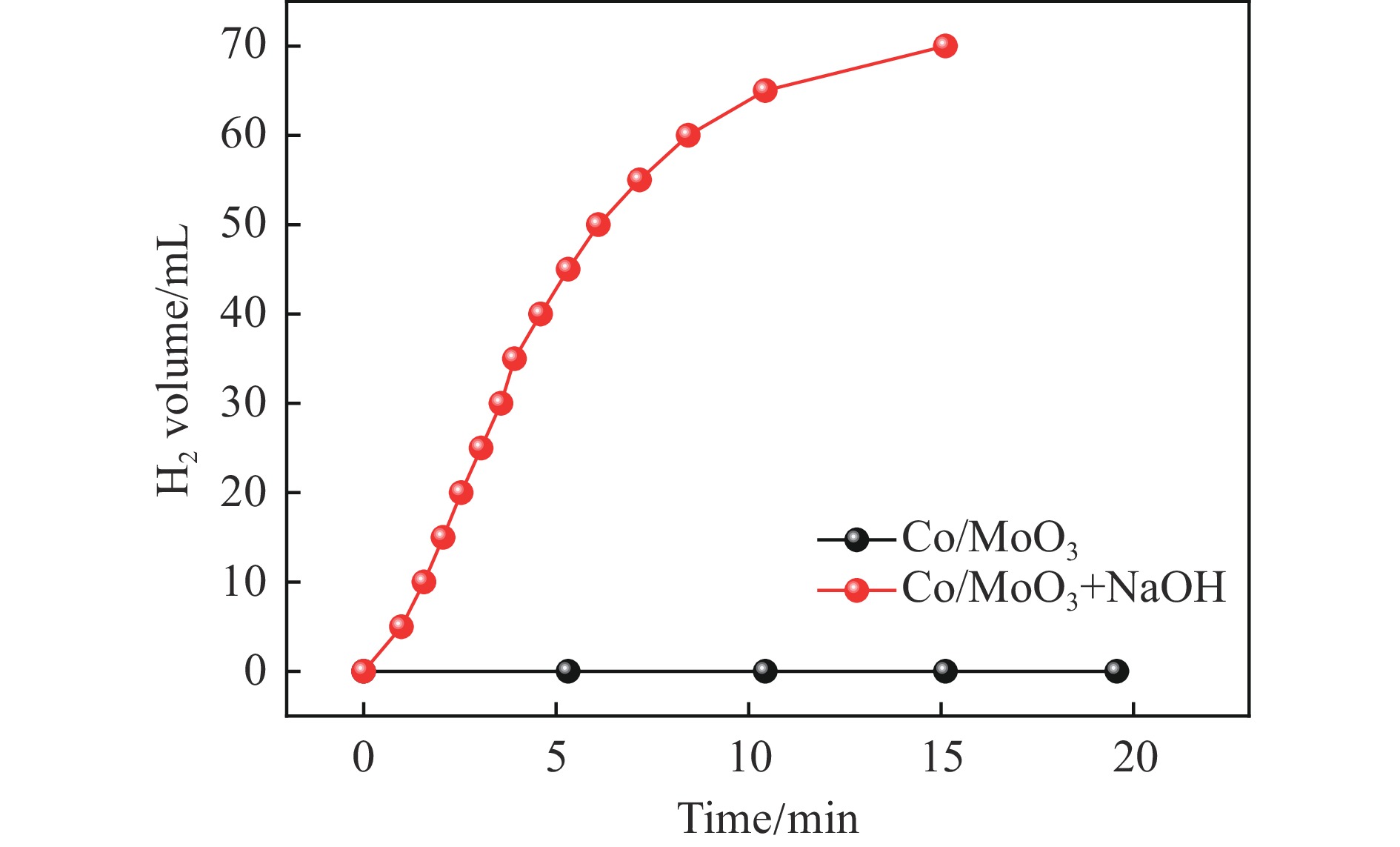
催化剂的构效关系是多相催化中研究催化作用机制的关键。本文以MoO3为载体通过液相还原法制备了Co/MoO3催化剂,用于催化氨硼烷水解释氢反应。Co/MoO3催化剂在无NaOH(碱)加入的体系中没有催化活性,不能催化氨硼烷水解释放氢气,而在有NaOH(碱)加入的体系中表现出优异的催化释氢性能。对Co/MoO3催化剂的组成结构在反应前后变化情况的表征分析表明,Co/MoO3催化剂中的组分是Co和Mo的无定形态存在的物质,非晶态的结构导致了在氨硼烷水解释氢反应中没有催化活性。在NaOH(碱)诱导作用下,Co/MoO3催化剂的组成结构由无定形态转变为针状Co(OH)2分散在片状堆叠的MoO3上的结构。NaOH(碱)的诱导重构作用使Co/MoO3催化剂在氨硼烷水解释氢反应中由没有催化活性转变为可以在15 min内使氨硼烷完全脱氢。
催化剂的构效关系是多相催化中研究催化作用机制的关键。本文以MoO3为载体通过液相还原法制备了Co/MoO3催化剂,用于催化氨硼烷水解释氢反应。Co/MoO3催化剂在无NaOH(碱)加入的体系中没有催化活性,不能催化氨硼烷水解释放氢气,而在有NaOH(碱)加入的体系中表现出优异的催化释氢性能。对Co/MoO3催化剂的组成结构在反应前后变化情况的表征分析表明,Co/MoO3催化剂中的组分是Co和Mo的无定形态存在的物质,非晶态的结构导致了在氨硼烷水解释氢反应中没有催化活性。在NaOH(碱)诱导作用下,Co/MoO3催化剂的组成结构由无定形态转变为针状Co(OH)2分散在片状堆叠的MoO3上的结构。NaOH(碱)的诱导重构作用使Co/MoO3催化剂在氨硼烷水解释氢反应中由没有催化活性转变为可以在15 min内使氨硼烷完全脱氢。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0001
摘要:
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60450-0
摘要:
将CO2捕获并通过加氢转化为醇类等高值化学品是实现CO2减排和碳资源循环利用的重要途径之一。本文对Rh/CeO2催化剂在CO2加氢反应中的性能进行了研究,并结合XRD、Raman、H2-TPR、CO2-TPD、CO-DRIFTS和XPS等表征方法,揭示了Rh负载量(0.1%–2.0%)对其CO2加氢活性和产物选择性的影响。结果表明,在3.0 MPa、250 °C的反应条件下,Rh负载量为0.1%时Rh/CeO2催化剂上CO2加氢产物以乙醇为主。随着Rh含量的增加,CO2转化率增加,但乙醇选择性降低;当Rh负载量为2.0%时,产物以甲醇为主。对于Rh负载量不同的催化剂上的CO2加氢反应,其产物选择性的差异与催化剂中Rh的存在形式和电子性质有关;原子分散的Rh+有利于稳定CO*,CO*与CH3*进行C–C偶联形成乙醇,而金属态的Rh团簇则容易促成CO*加氢生成甲醇。
将CO2捕获并通过加氢转化为醇类等高值化学品是实现CO2减排和碳资源循环利用的重要途径之一。本文对Rh/CeO2催化剂在CO2加氢反应中的性能进行了研究,并结合XRD、Raman、H2-TPR、CO2-TPD、CO-DRIFTS和XPS等表征方法,揭示了Rh负载量(0.1%–2.0%)对其CO2加氢活性和产物选择性的影响。结果表明,在3.0 MPa、250 °C的反应条件下,Rh负载量为0.1%时Rh/CeO2催化剂上CO2加氢产物以乙醇为主。随着Rh含量的增加,CO2转化率增加,但乙醇选择性降低;当Rh负载量为2.0%时,产物以甲醇为主。对于Rh负载量不同的催化剂上的CO2加氢反应,其产物选择性的差异与催化剂中Rh的存在形式和电子性质有关;原子分散的Rh+有利于稳定CO*,CO*与CH3*进行C–C偶联形成乙醇,而金属态的Rh团簇则容易促成CO*加氢生成甲醇。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024021
摘要:
采用胶质晶体模板法制备了不同孔径Cu-ZnO-ZrO2(CZZ)催化剂,并对其CO2加氢制甲醇性能进行了研究。结果表明,通过改变催化剂孔径可以实现ZnO粒径大小的调控,较小的粒径表现出更卓越的催化性能。其中,在孔径为55 nm的(CZZ-55)样品上,ZnO粒径为14.5 nm,CO2转化率为14.83%,甲醇选择性为78.8%,甲醇产率可达345.8 g/(kg·h)。原位漫反射傅里叶变换红外光谱结果表明,在CZZ催化剂上CO2加氢制甲醇遵循甲酸盐路径,ZnO-ZrO2界面是CO2吸附和活化的活性位点,而三维有序大孔结构有助于形成更分散的ZnO-ZrO2活性位,提高了CO2转化率。并且孔径大小对中间体的转化具有一定影响,孔径越小,甲酸盐更容易转化为甲醇。此外,三维有序的大孔结构为产物(水汽和甲醇)快速扩散提供了“高速通道”,有效抑制CO2加氢的副产物水汽对活性位的毒化作用,较大程度提高了催化剂的稳定性,在600 h内无明显失活。
采用胶质晶体模板法制备了不同孔径Cu-ZnO-ZrO2(CZZ)催化剂,并对其CO2加氢制甲醇性能进行了研究。结果表明,通过改变催化剂孔径可以实现ZnO粒径大小的调控,较小的粒径表现出更卓越的催化性能。其中,在孔径为55 nm的(CZZ-55)样品上,ZnO粒径为14.5 nm,CO2转化率为14.83%,甲醇选择性为78.8%,甲醇产率可达345.8 g/(kg·h)。原位漫反射傅里叶变换红外光谱结果表明,在CZZ催化剂上CO2加氢制甲醇遵循甲酸盐路径,ZnO-ZrO2界面是CO2吸附和活化的活性位点,而三维有序大孔结构有助于形成更分散的ZnO-ZrO2活性位,提高了CO2转化率。并且孔径大小对中间体的转化具有一定影响,孔径越小,甲酸盐更容易转化为甲醇。此外,三维有序的大孔结构为产物(水汽和甲醇)快速扩散提供了“高速通道”,有效抑制CO2加氢的副产物水汽对活性位的毒化作用,较大程度提高了催化剂的稳定性,在600 h内无明显失活。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0007
摘要:
Fe基催化剂因价格低廉、结构易于调控、热稳定性优良以及兼具CO2分子C−O键活化和C−C键偶联活性位点等优势,成为热催化CO2加氢制高值化学品C2+醇的理想选择。本工作围绕Fe基催化剂制备、改性和结构-性能关系等,介绍了CO2加氢制C2+醇领域的最新研究进展,讨论了影响Fe基催化剂CO2加氢反应性能的关键因素,即活性位点电子结构特性影响关键反应中间体的吸附行为,进而影响催化反应网络。阐述了金属-载体间电子相互作用和第二元素掺杂改性两大最常用策略调控Fe基催化剂电子结构特性的有效性,讨论了双组分级联催化和CO/CO2共进气两种反应过程强化策略。本工作也展望了该研究领域面临的机遇和挑战,以期推动热催化CO2加氢制C2+醇Fe基催化剂的工程化应用。
Fe基催化剂因价格低廉、结构易于调控、热稳定性优良以及兼具CO2分子C−O键活化和C−C键偶联活性位点等优势,成为热催化CO2加氢制高值化学品C2+醇的理想选择。本工作围绕Fe基催化剂制备、改性和结构-性能关系等,介绍了CO2加氢制C2+醇领域的最新研究进展,讨论了影响Fe基催化剂CO2加氢反应性能的关键因素,即活性位点电子结构特性影响关键反应中间体的吸附行为,进而影响催化反应网络。阐述了金属-载体间电子相互作用和第二元素掺杂改性两大最常用策略调控Fe基催化剂电子结构特性的有效性,讨论了双组分级联催化和CO/CO2共进气两种反应过程强化策略。本工作也展望了该研究领域面临的机遇和挑战,以期推动热催化CO2加氢制C2+醇Fe基催化剂的工程化应用。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60467-6
摘要:
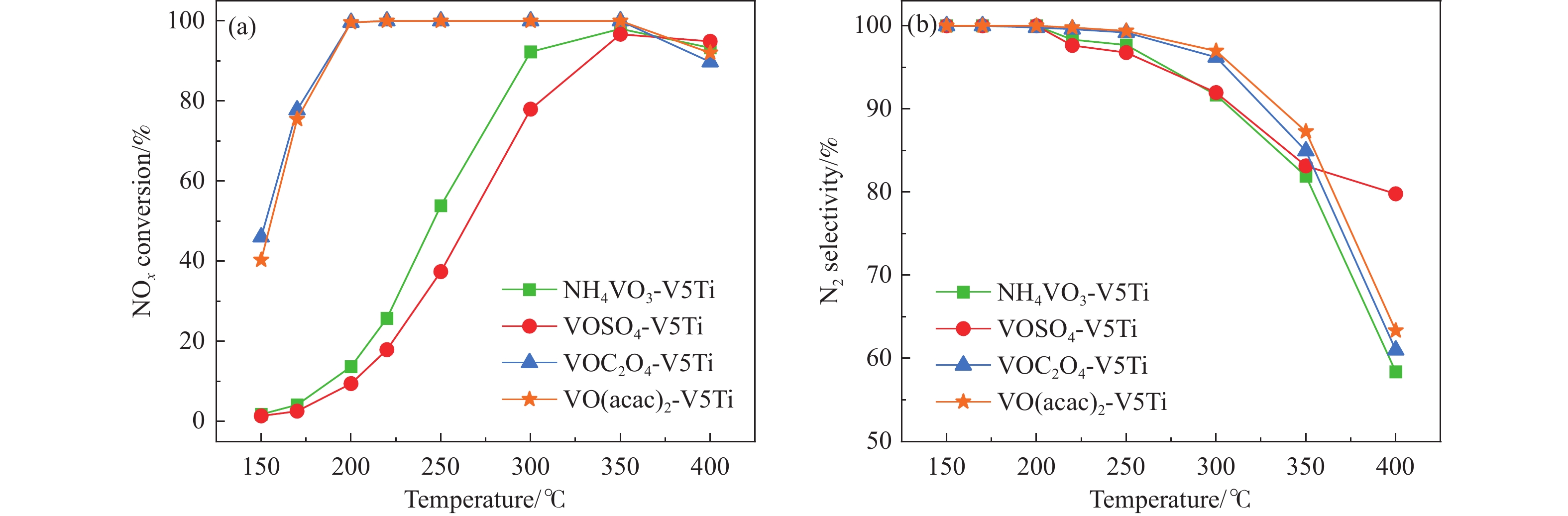
本研究采用固相合成法制备了一系列不同钒前驱体和不同钒负载量的V2O5/TiO2粉体催化剂。通过X射线衍射、X射线光电子能谱、氨气程序升温脱附和氢气程序升温还原等表征对催化剂的物理化学性质进行了分析,并在固定床反应器上对催化剂的脱硝活性进行了评价。以草酸氧钒(VOC2O4·xH2O)和乙酰丙酮氧钒(VO(acac)2)为钒前驱体,钒负载量为5%时,制备的粉体催化剂在200−350 ℃条件下NOx转化率稳定在100%,表现出最高的脱硝活性,相比于以偏钒酸铵(NH4VO3)和硫酸氧钒(VOSO4·xH2O)为钒前驱体制备的催化剂,其最高活性温度往低温区迁移了约150 ℃。而且以VO(acac)2作为钒前驱体制备的低钒含量(1%)催化剂的脱硝活性甚至高于以NH4VO3为钒前驱体制备的的高钒含量(6%)催化剂。结果表明,以VOC2O4和VO(acac)2为钒前驱体能有效调控催化剂上的活性位点和聚合状态,促进不同价态的V原子相互作用,形成更多的还原性V物种(V4+),从而表现出优异的SCR反应活性,同时也为制备低钒高活性的低温脱硝催化剂提供了一种有效方法。
本研究采用固相合成法制备了一系列不同钒前驱体和不同钒负载量的V2O5/TiO2粉体催化剂。通过X射线衍射、X射线光电子能谱、氨气程序升温脱附和氢气程序升温还原等表征对催化剂的物理化学性质进行了分析,并在固定床反应器上对催化剂的脱硝活性进行了评价。以草酸氧钒(VOC2O4·xH2O)和乙酰丙酮氧钒(VO(acac)2)为钒前驱体,钒负载量为5%时,制备的粉体催化剂在200−350 ℃条件下NOx转化率稳定在100%,表现出最高的脱硝活性,相比于以偏钒酸铵(NH4VO3)和硫酸氧钒(VOSO4·xH2O)为钒前驱体制备的催化剂,其最高活性温度往低温区迁移了约150 ℃。而且以VO(acac)2作为钒前驱体制备的低钒含量(1%)催化剂的脱硝活性甚至高于以NH4VO3为钒前驱体制备的的高钒含量(6%)催化剂。结果表明,以VOC2O4和VO(acac)2为钒前驱体能有效调控催化剂上的活性位点和聚合状态,促进不同价态的V原子相互作用,形成更多的还原性V物种(V4+),从而表现出优异的SCR反应活性,同时也为制备低钒高活性的低温脱硝催化剂提供了一种有效方法。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60470-6
摘要:
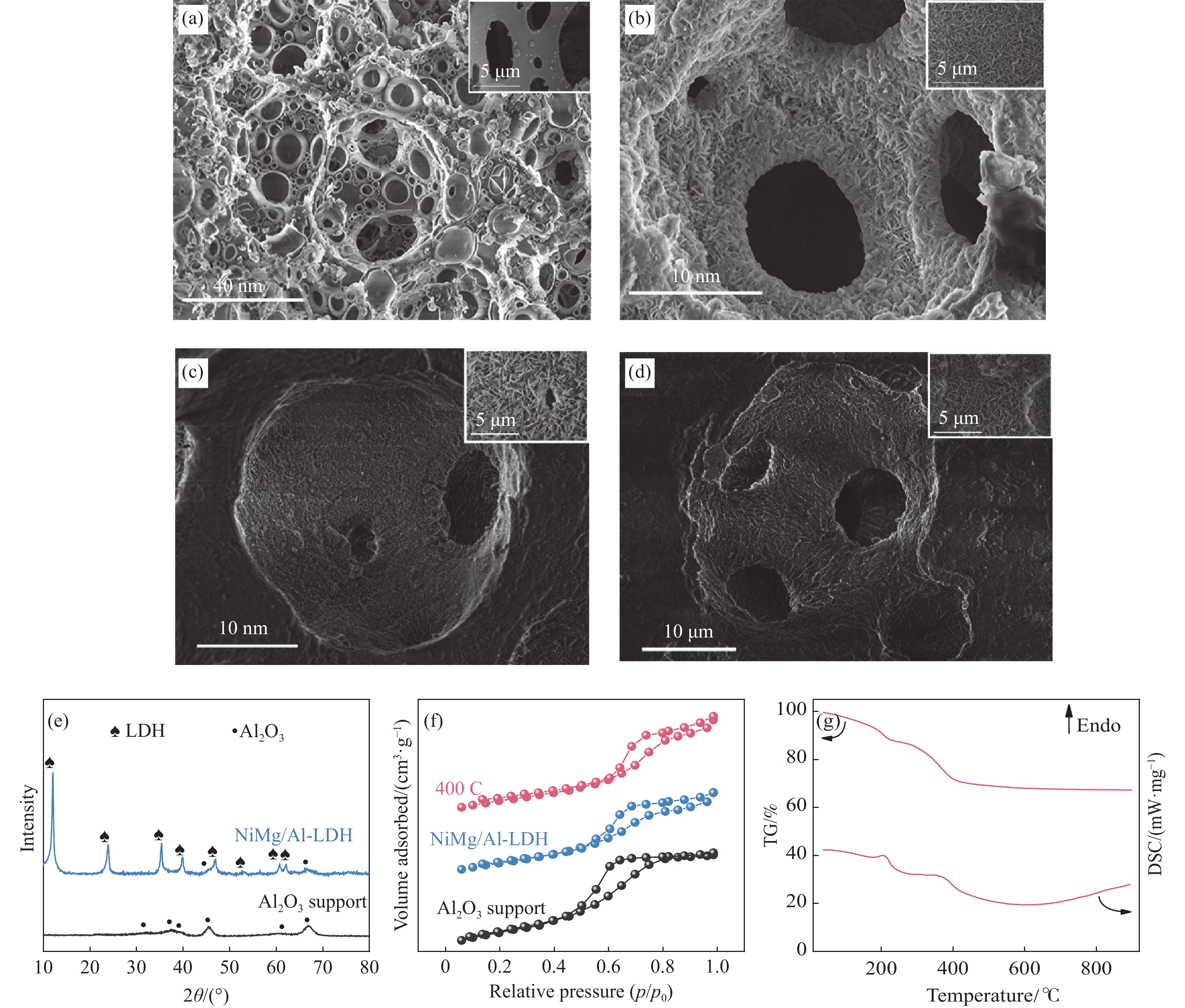
本工作将大孔氧化铝用于CO2甲烷化反应,通过原位生长类水滑石(LDH)的方法在大孔氧化铝表面生成NiMgAl-LDH前驱体,制备出了高比表面积、大孔径和大孔容的Ni-MgO/Al2O3催化剂,并研究了煅烧温度、还原温度和空速对催化剂结构及反应性能的影响。通过调整煅烧温度来控制催化剂的物相组成,通过控制还原温度调节Ni的还原度和避免烧结,提高还原后的催化剂中Ni0的活性位数量,从而提高催化剂活性。结果表明,NiMgAl-LDH前驱体在400 ℃煅烧,650 ℃还原后,制备的Ni-MgO/Al2O3催化剂具有最高的Ni活性比表面积,对应CO2转化率和CH4选择性最优,显示提高Ni表面积是提高性能的一个关键。并且该材料在WHSV = 80000 mL/(g·h)的条件下仍能保持高催化性能,证明其能够适应高空速运行。此外,在550 ℃的测试温度下,该催化剂表现出优良的稳定性,CO2转化率保持在54%, CH4选择性保持在79%。
本工作将大孔氧化铝用于CO2甲烷化反应,通过原位生长类水滑石(LDH)的方法在大孔氧化铝表面生成NiMgAl-LDH前驱体,制备出了高比表面积、大孔径和大孔容的Ni-MgO/Al2O3催化剂,并研究了煅烧温度、还原温度和空速对催化剂结构及反应性能的影响。通过调整煅烧温度来控制催化剂的物相组成,通过控制还原温度调节Ni的还原度和避免烧结,提高还原后的催化剂中Ni0的活性位数量,从而提高催化剂活性。结果表明,NiMgAl-LDH前驱体在400 ℃煅烧,650 ℃还原后,制备的Ni-MgO/Al2O3催化剂具有最高的Ni活性比表面积,对应CO2转化率和CH4选择性最优,显示提高Ni表面积是提高性能的一个关键。并且该材料在WHSV = 80000 mL/(g·h)的条件下仍能保持高催化性能,证明其能够适应高空速运行。此外,在550 ℃的测试温度下,该催化剂表现出优良的稳定性,CO2转化率保持在54%, CH4选择性保持在79%。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024025
摘要:
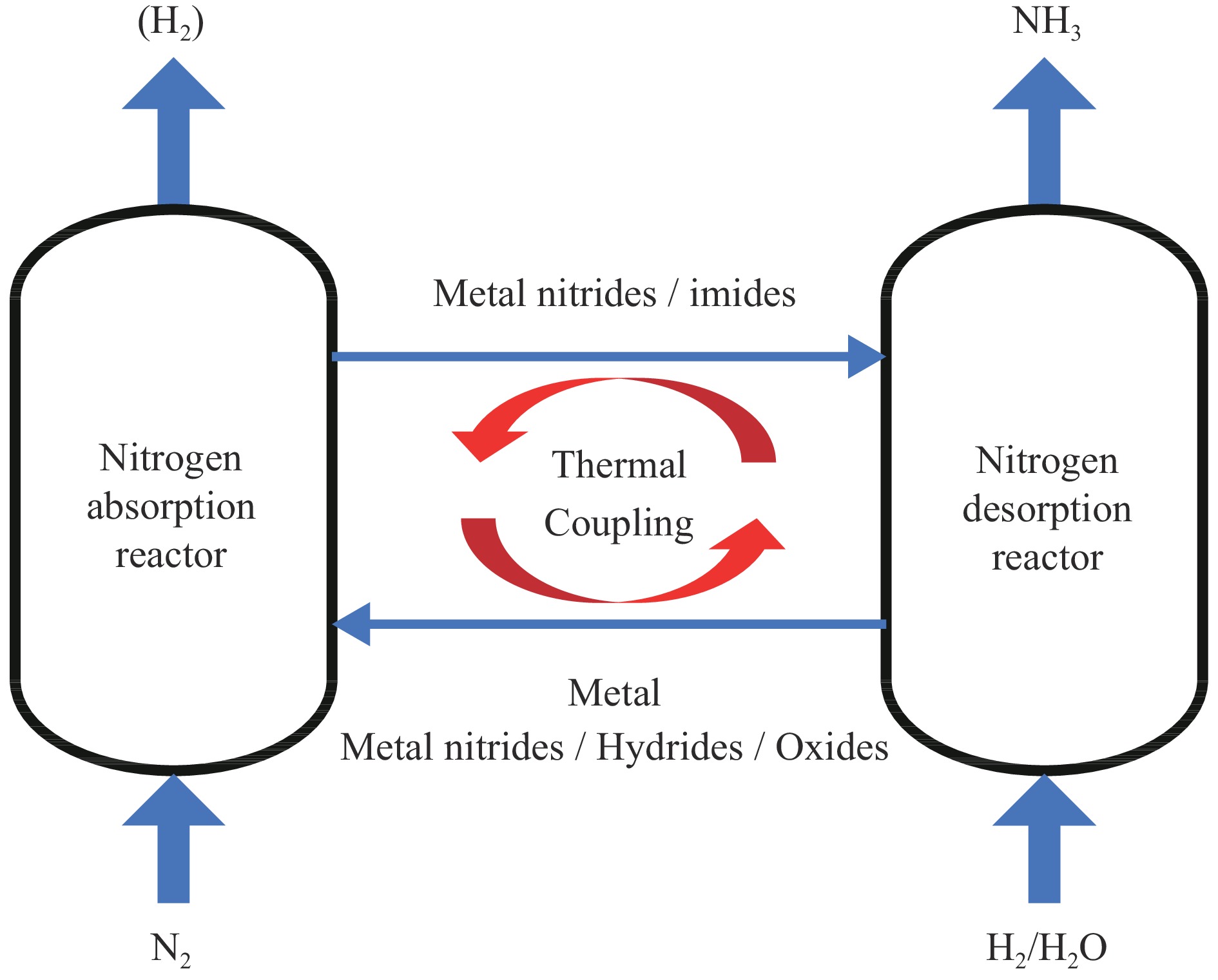
“双碳”背景下,氮或将成为取代碳的重要元素,可完成无碳燃料氨以及其他化合物的生产。其中,氨不仅是重要的化工原料,更是良好的能源载体。化学链技术通过重新设计反应路径,将过程分解为不同空间或时间内进行的两个或多个子反应,通过载体介质的反应和再生在耦合系统中实现物质和能量传递。作为一种新兴的清洁、有效能源转化手段,化学链技术近年来得到了广泛的关注。基于此,本工作对化学链技术在含氮化合物合成与转化领域中的研究进行综述,概述了以Haber-Bosch工艺为基础的多相催化和光、电等外场力驱动的化学链合成氨工艺的新发展,并对其进行总结和讨论。在含氮化合物合成方面,介绍了化学链技术用于氨氧化制一氧化氮以及烷基硝酸酯为关键中间体的烷烃化学链制醇类物质的过程。最后对基于化学链的氮转化与利用面临的挑战进行了分析和讨论,以期为今后化学链制含氮化学品提供参考。
“双碳”背景下,氮或将成为取代碳的重要元素,可完成无碳燃料氨以及其他化合物的生产。其中,氨不仅是重要的化工原料,更是良好的能源载体。化学链技术通过重新设计反应路径,将过程分解为不同空间或时间内进行的两个或多个子反应,通过载体介质的反应和再生在耦合系统中实现物质和能量传递。作为一种新兴的清洁、有效能源转化手段,化学链技术近年来得到了广泛的关注。基于此,本工作对化学链技术在含氮化合物合成与转化领域中的研究进行综述,概述了以Haber-Bosch工艺为基础的多相催化和光、电等外场力驱动的化学链合成氨工艺的新发展,并对其进行总结和讨论。在含氮化合物合成方面,介绍了化学链技术用于氨氧化制一氧化氮以及烷基硝酸酯为关键中间体的烷烃化学链制醇类物质的过程。最后对基于化学链的氮转化与利用面临的挑战进行了分析和讨论,以期为今后化学链制含氮化学品提供参考。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60471-8
摘要:
利用固定床反应器开展了淖毛湖煤和脱碱木质素的共热解实验,并研究了共热解产物的组成和产率变化规律。研究结果表明,共热解会降低半焦产率,促进热解气产率的提升,热解气产率最大提升了33.1%,共热解对于CH4、CO的生成有明显的促进作用;在煤和木质素混合比例为1∶1时,煤和木质素的热解挥发分间交互作用表现最明显,热解焦油产率表现出正协同作用。在共热解过程中,愈创木酚向单酚、双酚转化,相较于理论计算值,单酚、双酚化合物的含量分别增加了2.9%和9.8%,而愈创木酚的含量降低了5.1%;其原因可能是羰基和羧基的断裂与挥发分间的交互作用增强,抑制了醚类、醛类、酸类化合物的生成,促进了酚类的生成、含氧气体的释放与热解焦油的稳定。木质素的引入,显著促进了煤热解焦油轻质化,焦油中轻质组分比例接近90 %。
利用固定床反应器开展了淖毛湖煤和脱碱木质素的共热解实验,并研究了共热解产物的组成和产率变化规律。研究结果表明,共热解会降低半焦产率,促进热解气产率的提升,热解气产率最大提升了33.1%,共热解对于CH4、CO的生成有明显的促进作用;在煤和木质素混合比例为1∶1时,煤和木质素的热解挥发分间交互作用表现最明显,热解焦油产率表现出正协同作用。在共热解过程中,愈创木酚向单酚、双酚转化,相较于理论计算值,单酚、双酚化合物的含量分别增加了2.9%和9.8%,而愈创木酚的含量降低了5.1%;其原因可能是羰基和羧基的断裂与挥发分间的交互作用增强,抑制了醚类、醛类、酸类化合物的生成,促进了酚类的生成、含氧气体的释放与热解焦油的稳定。木质素的引入,显著促进了煤热解焦油轻质化,焦油中轻质组分比例接近90 %。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0003
摘要:
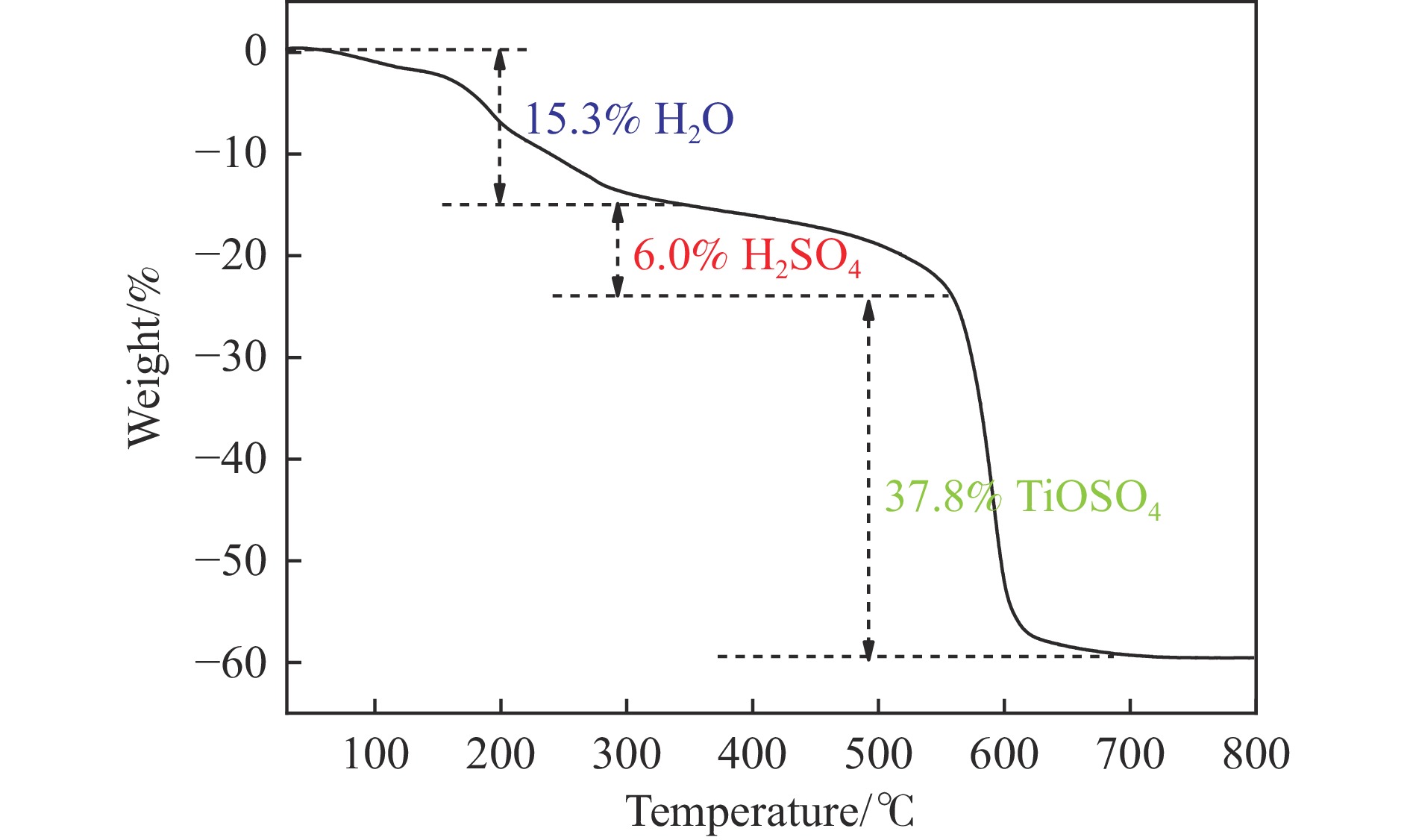
通过焙烧硫酸氧钛水合物前驱体制备了一系列硫掺杂TiO2负载Ni纳米颗粒催化剂。考察了不同焙烧温度对TiO2表面硫物种含量及光催化苯乙炔选择性加氢反应性能的影响。采用XRD、UV-vis DRS、TEM等技术对催化剂的结构和形貌进行了表征。XPS和原位DRIFTS结果表明,TiO2表面的硫物种向Ni发生电子转移,富电子态的Ni可以促进苯乙烯的脱附;原位DRIFTS结果表明,光照可以促进催化剂表面H2的解离和活化,提高苯乙炔的转化频率。本工作有助于促进对高效高选择性加氢光催化剂的设计和理解。
通过焙烧硫酸氧钛水合物前驱体制备了一系列硫掺杂TiO2负载Ni纳米颗粒催化剂。考察了不同焙烧温度对TiO2表面硫物种含量及光催化苯乙炔选择性加氢反应性能的影响。采用XRD、UV-vis DRS、TEM等技术对催化剂的结构和形貌进行了表征。XPS和原位DRIFTS结果表明,TiO2表面的硫物种向Ni发生电子转移,富电子态的Ni可以促进苯乙烯的脱附;原位DRIFTS结果表明,光照可以促进催化剂表面H2的解离和活化,提高苯乙炔的转化频率。本工作有助于促进对高效高选择性加氢光催化剂的设计和理解。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60469-X
摘要:
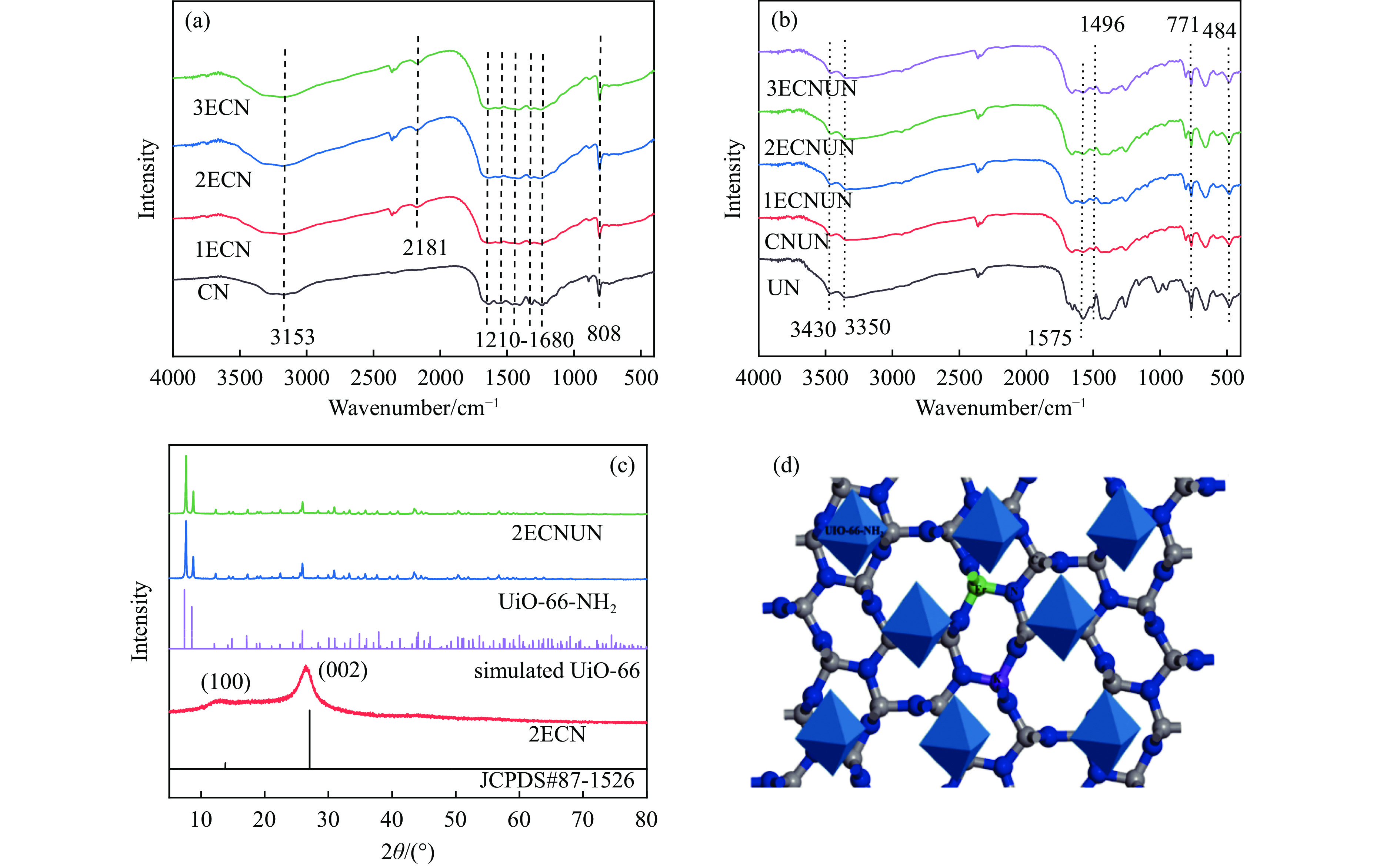
Biomass-derived platform molecules, such as furfural, are abundant and renewable feedstock for valuable chemical production. It is critical to synthesize highly efficient photocatalysts for selective oxidation under visible light. The Er@K-C3N4/UiO-66-NH2 catalyst was synthesized using a straight-forward hydrothermal technique, and exhibited exceptional efficiency in the photocatalytic oxidation of furfural to furoic acid. The catalyst was thoroughly characterized, confirming the effective adjustment of the band gap energy of Er@K-C3N4/UiO-66-NH2. Upon the optimized reaction conditions, the conversion rate of furfural reached 89.3%, with a corresponding yield of furoic acid at 79.8%. The primary reactive oxygen species was identified as ·O2 − from ESR spectra and scavenger tests. The incorporation of Er and K into the catalyst enhanced the photogenerated carriers transfer rate, hence increasing the separating efficiency of photogenerated electron-hole pairs. This study expands the potential applications of rare earth element doped g-C3N4 in the photocatalytic selective oxidation of furfurans.
Biomass-derived platform molecules, such as furfural, are abundant and renewable feedstock for valuable chemical production. It is critical to synthesize highly efficient photocatalysts for selective oxidation under visible light. The Er@K-C3N4/UiO-66-NH2 catalyst was synthesized using a straight-forward hydrothermal technique, and exhibited exceptional efficiency in the photocatalytic oxidation of furfural to furoic acid. The catalyst was thoroughly characterized, confirming the effective adjustment of the band gap energy of Er@K-C3N4/UiO-66-NH2. Upon the optimized reaction conditions, the conversion rate of furfural reached 89.3%, with a corresponding yield of furoic acid at 79.8%. The primary reactive oxygen species was identified as ·O2 − from ESR spectra and scavenger tests. The incorporation of Er and K into the catalyst enhanced the photogenerated carriers transfer rate, hence increasing the separating efficiency of photogenerated electron-hole pairs. This study expands the potential applications of rare earth element doped g-C3N4 in the photocatalytic selective oxidation of furfurans.
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024027
摘要:
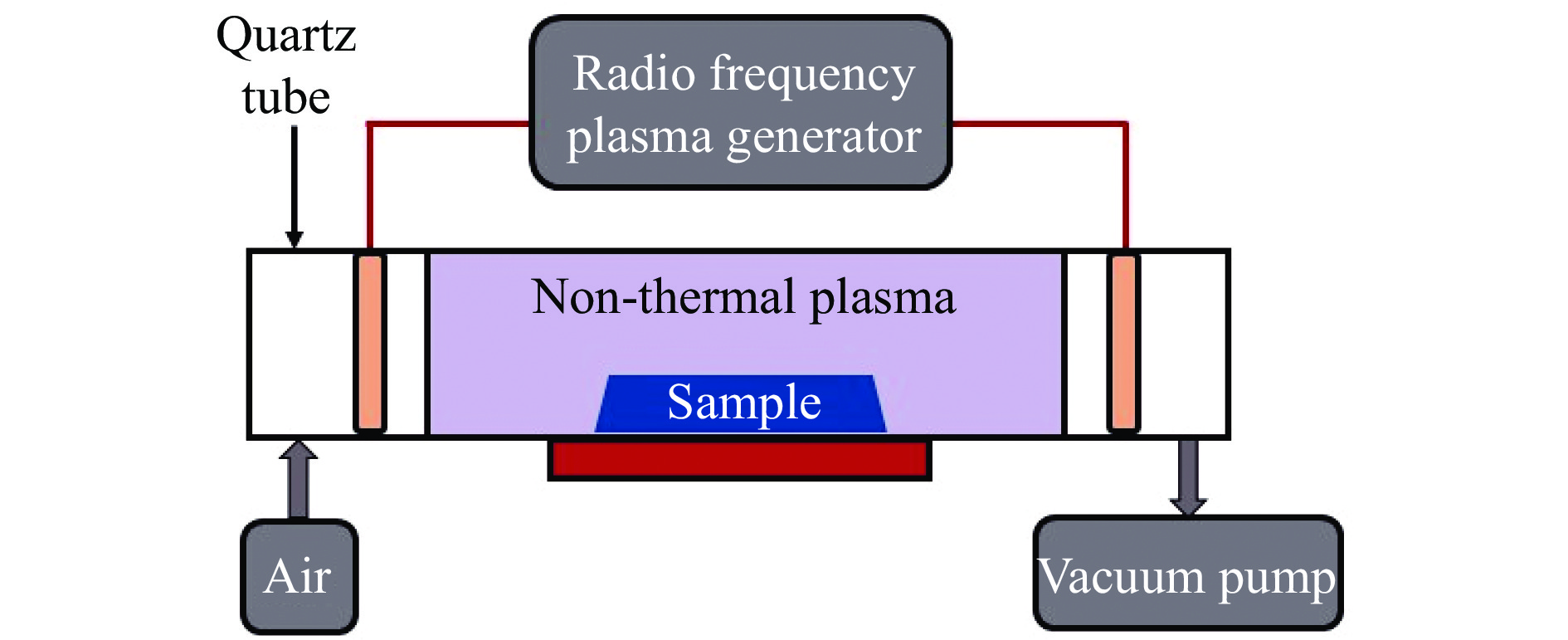
合成气一步制乙醇是利用非石油资源生产乙醇的重要方法,如何提高乙醇选择性、创制高效催化剂是改善过程经济性的重点。本研究从硫化钼前体出发,分别采用传统热法和射频低温等离子体法制备钼基氧硫复合物催化剂并考察其催化合成气制乙醇反应性能。利用XRD、UV-visible、HR-TEM、SEM、HAADF-STEM、XPS、CO-TPD、H2-TPD、CO2-TPD和In-situ DRIFTS等表征手段研究不同制备方法下合成的钼基氧硫复合物催化剂的不同物化性质,进而探究特征差异引发的催化反应性能变化。其中,MOS-P催化剂表现出最佳性能,在6 MPa、320 ℃、空速4500 h−1的反应条件下,CO转化率达到22.5%,总醇选择性可达71.4%,其中,总醇中乙醇占比为29.1%。有关研究将为合成气定向转化提供理论指导并为新型钼基材料的设计与制备提供借鉴。
合成气一步制乙醇是利用非石油资源生产乙醇的重要方法,如何提高乙醇选择性、创制高效催化剂是改善过程经济性的重点。本研究从硫化钼前体出发,分别采用传统热法和射频低温等离子体法制备钼基氧硫复合物催化剂并考察其催化合成气制乙醇反应性能。利用XRD、UV-visible、HR-TEM、SEM、HAADF-STEM、XPS、CO-TPD、H2-TPD、CO2-TPD和In-situ DRIFTS等表征手段研究不同制备方法下合成的钼基氧硫复合物催化剂的不同物化性质,进而探究特征差异引发的催化反应性能变化。其中,MOS-P催化剂表现出最佳性能,在6 MPa、320 ℃、空速4500 h−1的反应条件下,CO转化率达到22.5%,总醇选择性可达71.4%,其中,总醇中乙醇占比为29.1%。有关研究将为合成气定向转化提供理论指导并为新型钼基材料的设计与制备提供借鉴。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60475-5
摘要:
生物质气化制合成气(H2/CO)过程中伴随副产物焦油的产生,会引发环境污染问题和阻碍气化技术发展。低温等离子体与镍基催化剂结合能够利用等离子体的低温反应及催化剂的高选择性优势,将焦油定向重整转化为合成气。但镍颗粒在重整过程中会形成积炭沉积易导致催化剂失活,所以设计和改性催化剂提高其抗积炭性能成为解决催化剂失活问题的关键。本工作综述了近年来低温等离子体催化重整焦油体系内镍基催化剂失活原理以及催化剂的设计和改性以提高其抗积炭性能、低温等离子体重整焦油反应器类型及其机理、低温等离子体与催化剂的协同作用等方面的研究进展,并对协同体系重整焦油未来发展方向进行展望。本论文为低温等离子体催化重整焦油体系内镍基催化剂的设计与开发提供了参考依据。
生物质气化制合成气(H2/CO)过程中伴随副产物焦油的产生,会引发环境污染问题和阻碍气化技术发展。低温等离子体与镍基催化剂结合能够利用等离子体的低温反应及催化剂的高选择性优势,将焦油定向重整转化为合成气。但镍颗粒在重整过程中会形成积炭沉积易导致催化剂失活,所以设计和改性催化剂提高其抗积炭性能成为解决催化剂失活问题的关键。本工作综述了近年来低温等离子体催化重整焦油体系内镍基催化剂失活原理以及催化剂的设计和改性以提高其抗积炭性能、低温等离子体重整焦油反应器类型及其机理、低温等离子体与催化剂的协同作用等方面的研究进展,并对协同体系重整焦油未来发展方向进行展望。本论文为低温等离子体催化重整焦油体系内镍基催化剂的设计与开发提供了参考依据。
当前状态:
, 最新更新时间: ,
doi: 10.3724/2097-213X.2024.JFCT.0005
摘要:
采用两步水热法制备了一系列Mo-Sn催化剂,通过改变锡氧化物焙烧温度,考察钼氧化物与不同性质锡氧化物之间的作用方式对二甲醚(DME)低温氧化制甲酸甲酯(MF)性能的影响。结果表明,催化剂的性能与处理条件密切相关,当80 ℃焙烧Sn氧化物,与Mo进行水热作用,再通过500 ℃焙烧得到的Mo1Sn2-80Sn-500催化剂表现出较好的活性,110 ℃反应温度下,MF选择性达97.7%,二甲醚转化率为14.7%。通过XRD、Raman、FT-IR、低温ESR、NH3-TPD、CO2-TPD及H2-TPR对催化剂的表面物理化学性质以及钼物种的配位结构进行了表征。结果表明,Sn氧化物焙烧温度的改变影响了钼氧化物在SnO2表面的存在形式,较低温度处理Sn氧化物后,催化剂中MoO3与MoOx共存,且Mo-Sn界面处Mo5+配位结构增多,催化剂表面较多的酸量有利于DME氧化反应进行,强碱不利于MF生成。
采用两步水热法制备了一系列Mo-Sn催化剂,通过改变锡氧化物焙烧温度,考察钼氧化物与不同性质锡氧化物之间的作用方式对二甲醚(DME)低温氧化制甲酸甲酯(MF)性能的影响。结果表明,催化剂的性能与处理条件密切相关,当80 ℃焙烧Sn氧化物,与Mo进行水热作用,再通过500 ℃焙烧得到的Mo1Sn2-80Sn-500催化剂表现出较好的活性,110 ℃反应温度下,MF选择性达97.7%,二甲醚转化率为14.7%。通过XRD、Raman、FT-IR、低温ESR、NH3-TPD、CO2-TPD及H2-TPR对催化剂的表面物理化学性质以及钼物种的配位结构进行了表征。结果表明,Sn氧化物焙烧温度的改变影响了钼氧化物在SnO2表面的存在形式,较低温度处理Sn氧化物后,催化剂中MoO3与MoOx共存,且Mo-Sn界面处Mo5+配位结构增多,催化剂表面较多的酸量有利于DME氧化反应进行,强碱不利于MF生成。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60464-0
摘要:
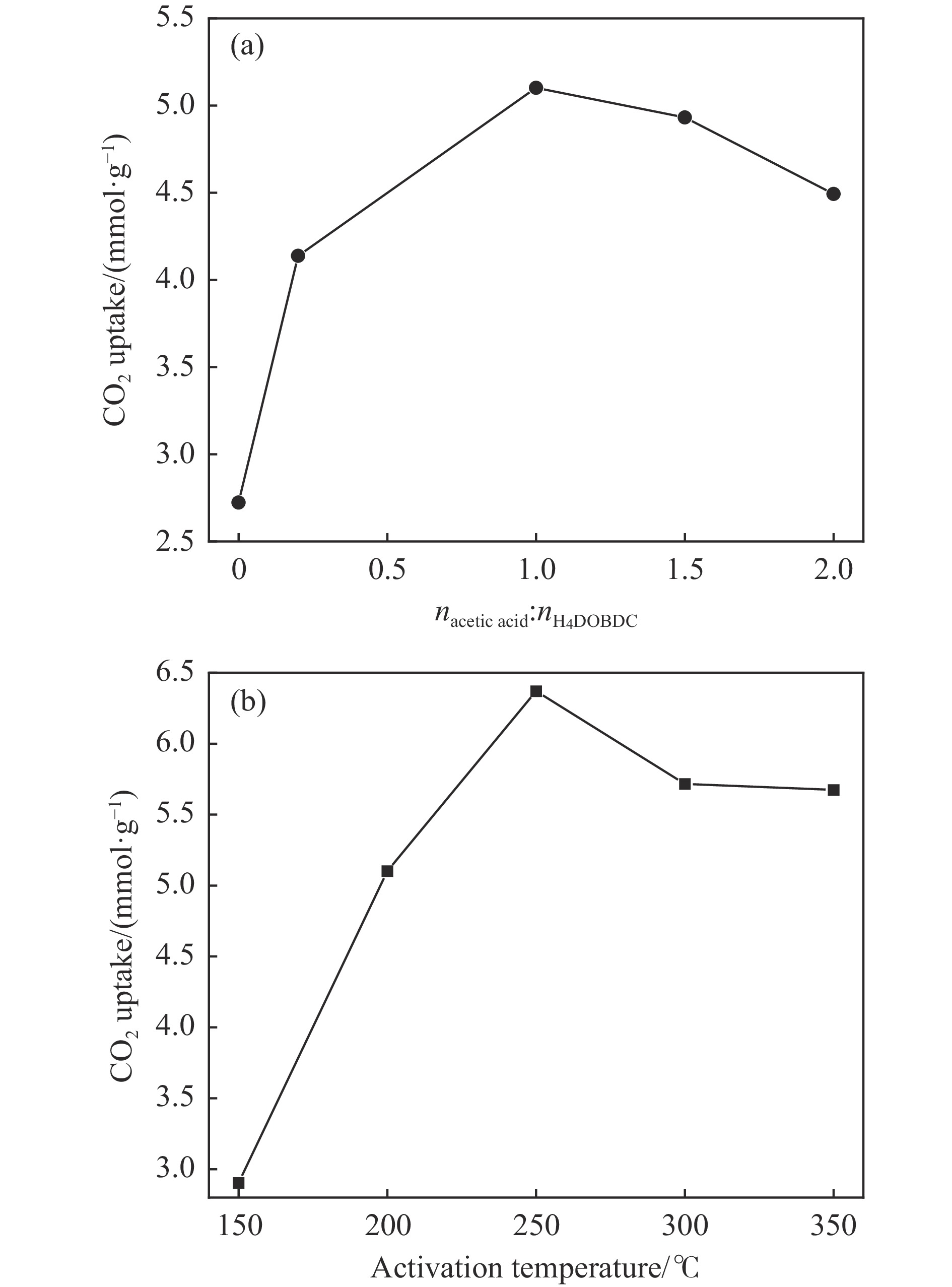
To enhance the separation selectivity of Mg-MOF-74 towards CO2 in a CO2/N2 mixture, a series of Mg-MOF-74 and Nix/Mg1−x-MOF-74 adsorbents were prepared by solvothermal synthesis in this paper. It was found that the adsorption capacity of Mg-MOF-74 for CO2 could be effectively increased by optimizing the amount of acetic acid. On this basis, the bimetal MOF-74 adsorbent was prepared by metal modification. The multi-component dynamic adsorption penetration analysis was utilized to examine the CO2 adsorption capacity and CO2/N2 selectivity of the diverse adsorbent materials. The results showed that Ni0.11/Mg0.89-MOF-74 showed a CO2 adsorption capacity of 7.02 mmol/g under pure CO2 atmosphere and had a selectivity of 20.50 for CO2/N2 under 15% CO2/85% N2 conditions, which was 10.2% and 18.02% higher than that of Mg-MOF-74 respectively. Combining XPS, SEM and FT-IR characterization analysis, it was attributed to the effect of the more stable unsaturated metal sites Ni into the Mg-MOF-74 on the pore structure and the synergistic interaction between the two metals. Density Functional Theory (DFT) simulations revealed that the synergistic interaction between modulated the electrostatic potential strength and gradient of the material, which was more favorable for the adsorption of CO2 molecules with small diameters and large quadrupole moment. In addition, the Ni0.11/Mg0.89-MOF-74 showed commendable cyclic stability, underscoring its promising potential for practical applications.
To enhance the separation selectivity of Mg-MOF-74 towards CO2 in a CO2/N2 mixture, a series of Mg-MOF-74 and Nix/Mg1−x-MOF-74 adsorbents were prepared by solvothermal synthesis in this paper. It was found that the adsorption capacity of Mg-MOF-74 for CO2 could be effectively increased by optimizing the amount of acetic acid. On this basis, the bimetal MOF-74 adsorbent was prepared by metal modification. The multi-component dynamic adsorption penetration analysis was utilized to examine the CO2 adsorption capacity and CO2/N2 selectivity of the diverse adsorbent materials. The results showed that Ni0.11/Mg0.89-MOF-74 showed a CO2 adsorption capacity of 7.02 mmol/g under pure CO2 atmosphere and had a selectivity of 20.50 for CO2/N2 under 15% CO2/85% N2 conditions, which was 10.2% and 18.02% higher than that of Mg-MOF-74 respectively. Combining XPS, SEM and FT-IR characterization analysis, it was attributed to the effect of the more stable unsaturated metal sites Ni into the Mg-MOF-74 on the pore structure and the synergistic interaction between the two metals. Density Functional Theory (DFT) simulations revealed that the synergistic interaction between modulated the electrostatic potential strength and gradient of the material, which was more favorable for the adsorption of CO2 molecules with small diameters and large quadrupole moment. In addition, the Ni0.11/Mg0.89-MOF-74 showed commendable cyclic stability, underscoring its promising potential for practical applications.
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60465-2
摘要:
本研究制备了核壳型和负载型的Ce-OMS-2复合物(Ce-OMS-2@CeO2和CeO2/Ce-OMS-2),并对其结构和性能进行了表征和测试。结果表明,核壳型Ce-OMS-2@CeO2材料由于其介孔结构的保持能够明显提升反应气体NO的传质和吸附,提升脱硝效率。同时,核壳型Ce-OMS-2@CeO2催化剂显著降低了硫酸氢铵(ABS)的分解温度,使得催化剂表面活性组分不易被ABS沉积覆盖,从而维持Ce-OMS-2@CeO2高效的抗硫抗水脱硝性能。因此,核壳型Ce-OMS-2@CeO2催化剂表现出优异的SCR脱硝性能和抗硫抗水性能(在无SO2下,100−200 ℃ NO转化率为~100%;在有SO2下,NO转化率≥~80%可维持在4 h以上)。本工作为开发高效稳定的Mn基低温SCR脱硝催化剂提供了一种有效策略。
本研究制备了核壳型和负载型的Ce-OMS-2复合物(Ce-OMS-2@CeO2和CeO2/Ce-OMS-2),并对其结构和性能进行了表征和测试。结果表明,核壳型Ce-OMS-2@CeO2材料由于其介孔结构的保持能够明显提升反应气体NO的传质和吸附,提升脱硝效率。同时,核壳型Ce-OMS-2@CeO2催化剂显著降低了硫酸氢铵(ABS)的分解温度,使得催化剂表面活性组分不易被ABS沉积覆盖,从而维持Ce-OMS-2@CeO2高效的抗硫抗水脱硝性能。因此,核壳型Ce-OMS-2@CeO2催化剂表现出优异的SCR脱硝性能和抗硫抗水性能(在无SO2下,100−200 ℃ NO转化率为~100%;在有SO2下,NO转化率≥~80%可维持在4 h以上)。本工作为开发高效稳定的Mn基低温SCR脱硝催化剂提供了一种有效策略。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60442-1
摘要:
以硝酸钴和硝酸铜制备溶液A,苯二甲酸(PTA)和N,N-二甲基甲酰胺(DMF)制备溶液B,两种溶液通过溶剂热法制备Co/Cu拉瓦希尔骨架系列材料(Co/Cu-MIL前驱体),进一步直接碳化前驱体制备出MOFs衍生物,即双金属碳纳米棒(CoxCu1−x/CNR)催化剂。通过SEM、TEM、XRD、XPS等表征手段探究其形貌和组成。结果表明,Co/Cu-MIL经过高温焙烧后成功得到CoxCu1−x/CNR,当x=0.5、溶剂热温度为120 ℃、焙烧温度为650 ℃时得到的催化剂催化活性最优,Co0.5Cu0.5/CNR催化剂催化氨硼烷(AB)水解制氢的TOF值为2718.21 h−1,反应的活化能为51.64 kJ/mol,且催化剂的循环稳定性较好,在循环10次后催化活性虽然有所下降,但对AB仍然保持100%的转化率。
以硝酸钴和硝酸铜制备溶液A,苯二甲酸(PTA)和N,N-二甲基甲酰胺(DMF)制备溶液B,两种溶液通过溶剂热法制备Co/Cu拉瓦希尔骨架系列材料(Co/Cu-MIL前驱体),进一步直接碳化前驱体制备出MOFs衍生物,即双金属碳纳米棒(CoxCu1−x/CNR)催化剂。通过SEM、TEM、XRD、XPS等表征手段探究其形貌和组成。结果表明,Co/Cu-MIL经过高温焙烧后成功得到CoxCu1−x/CNR,当x=0.5、溶剂热温度为120 ℃、焙烧温度为650 ℃时得到的催化剂催化活性最优,Co0.5Cu0.5/CNR催化剂催化氨硼烷(AB)水解制氢的TOF值为2718.21 h−1,反应的活化能为51.64 kJ/mol,且催化剂的循环稳定性较好,在循环10次后催化活性虽然有所下降,但对AB仍然保持100%的转化率。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60447-0
摘要:
生物油组成复杂,含氧量较高,制约其高值化利用。本研究以商业活性炭(AC)和HY分子筛为复合催化剂,通过改变催化区的装填方式研究其对稻草和杨木屑两种典型生物质热解挥发物提质的影响规律。结果表明,AC和HY分子筛装填方式影响生物质热解产物分布和生物油组成。在HY与AC按1∶1比例均匀混合(YACM)作用后,生物油产率最低。但YACM方式有利于生物油的脱氧和芳香烃的生成,稻草和杨木屑热解生物油中的芳烃含量在YACM作用下可分别由提质前的13.8%和8.0%提高至56.4%和53.1%。上层HY分子筛和下层AC(YTACL)的催化方式有利于酚类物质生成。对单环芳烃的选择性遵循YTACL>ACTYL>YACM,而对双环芳烃的选择性为YACM>ACTYL>YTACL。分析认为,AC孔径较HY分子筛小,酸性低于分子筛,其活性中心有助于呋喃化合物重排生成环戊酮、2-环戊烯酮、甲基环戊烯酮,后重排形成苯酚,因此,YTACL的装填方式对苯酚、甲酚、甲苯、乙苯、对二甲苯的生成有较好的促进作用。HY分子筛的酸性强,有利于芳构化反应发生,因此,ACTYL装填方式表现出对萘、甲基萘、蒽、芘的生成较高的选择性。该工作为生物油的组成调控以及芳烃和酚类物质的富集等提供重要指导。
生物油组成复杂,含氧量较高,制约其高值化利用。本研究以商业活性炭(AC)和HY分子筛为复合催化剂,通过改变催化区的装填方式研究其对稻草和杨木屑两种典型生物质热解挥发物提质的影响规律。结果表明,AC和HY分子筛装填方式影响生物质热解产物分布和生物油组成。在HY与AC按1∶1比例均匀混合(YACM)作用后,生物油产率最低。但YACM方式有利于生物油的脱氧和芳香烃的生成,稻草和杨木屑热解生物油中的芳烃含量在YACM作用下可分别由提质前的13.8%和8.0%提高至56.4%和53.1%。上层HY分子筛和下层AC(YTACL)的催化方式有利于酚类物质生成。对单环芳烃的选择性遵循YTACL>ACTYL>YACM,而对双环芳烃的选择性为YACM>ACTYL>YTACL。分析认为,AC孔径较HY分子筛小,酸性低于分子筛,其活性中心有助于呋喃化合物重排生成环戊酮、2-环戊烯酮、甲基环戊烯酮,后重排形成苯酚,因此,YTACL的装填方式对苯酚、甲酚、甲苯、乙苯、对二甲苯的生成有较好的促进作用。HY分子筛的酸性强,有利于芳构化反应发生,因此,ACTYL装填方式表现出对萘、甲基萘、蒽、芘的生成较高的选择性。该工作为生物油的组成调控以及芳烃和酚类物质的富集等提供重要指导。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024019
摘要:
加氢脱硫技术对实现劣质油品清洁化、低碳化与多元化高效利用至关重要,其关键是高性能催化剂的开发,核心之一是适宜催化剂载体材料的创新。本工作分别总结了向Al2O3及TiO2中引入第二组元后作为加氢脱硫催化剂载体的研究进展。第二组元氧化物的引入克服了Al2O3载体酸类型单一及商用催化剂金属与载体间相互作用过强等缺点,同时保持了较大的比表面积;第二组元氧化物能够有效提升TiO2载体材料的热稳定性及比表面积的同时调节了载体材料表面酸性等。究其原因在于第二组元的引入可显著改变Al2O3或TiO2表面羟基环境,进而促进了活性金属前驱体在载体表面的锚定和分散,有利于更多NiMo(W)S活性相的形成,提升了催化剂的加氢脱硫性能。
加氢脱硫技术对实现劣质油品清洁化、低碳化与多元化高效利用至关重要,其关键是高性能催化剂的开发,核心之一是适宜催化剂载体材料的创新。本工作分别总结了向Al2O3及TiO2中引入第二组元后作为加氢脱硫催化剂载体的研究进展。第二组元氧化物的引入克服了Al2O3载体酸类型单一及商用催化剂金属与载体间相互作用过强等缺点,同时保持了较大的比表面积;第二组元氧化物能够有效提升TiO2载体材料的热稳定性及比表面积的同时调节了载体材料表面酸性等。究其原因在于第二组元的引入可显著改变Al2O3或TiO2表面羟基环境,进而促进了活性金属前驱体在载体表面的锚定和分散,有利于更多NiMo(W)S活性相的形成,提升了催化剂的加氢脱硫性能。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024016
摘要:
气候变化的加剧要求更加绿色高效的碳减排技术和产品的开发。固废和生物质衍生的常规CO2碳质吸附剂材料吸附效果较差,需要进行额外的活化改性以提升多孔碳的吸附性能。本工作以废纸为原料,经过简单预处理和溶胶凝胶炭化工艺,得到微孔高度发达的超微孔废纸碳气凝胶,探究了不同种类废纸和制备温度的影响。对材料的理化特性和CO2吸附性能进行了表征和测试,结果表明,废纸碳气凝胶的孔隙结构发达,呈类蜂窝状。打印纸为原料,800 ℃下制备的废纸碳气凝胶具有大比表面积1369.94 m2/g、高孔容0.59 cm3/g和孔径为0.4−0.8 nm的超微孔。无需改性,0 ℃时的CO2吸附量为247 mg/g,对应CO2/N2吸附选择性为11,25 ℃时的动力学吸附量为151 mg/g,七次吸脱附循环的平均波动幅度小于5%,对烟气浓度CO2(10%)的捕获量为42 mg/g。废纸碳气凝胶展现出优异的CO2吸附性能和再生稳定性,优于固废和生物质衍生的常规炭材料。本工作也为固废处置和资源化利用提供了新思路。
气候变化的加剧要求更加绿色高效的碳减排技术和产品的开发。固废和生物质衍生的常规CO2碳质吸附剂材料吸附效果较差,需要进行额外的活化改性以提升多孔碳的吸附性能。本工作以废纸为原料,经过简单预处理和溶胶凝胶炭化工艺,得到微孔高度发达的超微孔废纸碳气凝胶,探究了不同种类废纸和制备温度的影响。对材料的理化特性和CO2吸附性能进行了表征和测试,结果表明,废纸碳气凝胶的孔隙结构发达,呈类蜂窝状。打印纸为原料,800 ℃下制备的废纸碳气凝胶具有大比表面积1369.94 m2/g、高孔容0.59 cm3/g和孔径为0.4−0.8 nm的超微孔。无需改性,0 ℃时的CO2吸附量为247 mg/g,对应CO2/N2吸附选择性为11,25 ℃时的动力学吸附量为151 mg/g,七次吸脱附循环的平均波动幅度小于5%,对烟气浓度CO2(10%)的捕获量为42 mg/g。废纸碳气凝胶展现出优异的CO2吸附性能和再生稳定性,优于固废和生物质衍生的常规炭材料。本工作也为固废处置和资源化利用提供了新思路。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60435-4
摘要:
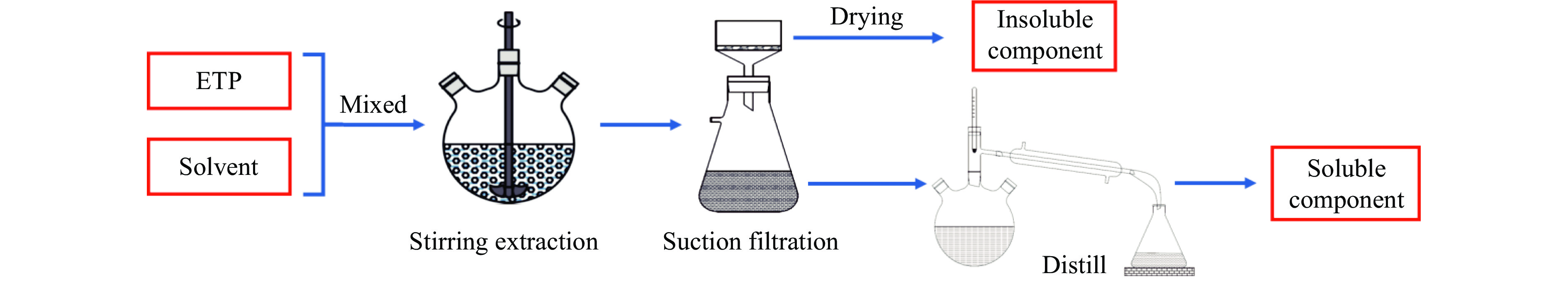
乙烯渣油沥青作为乙烯渣油中的重质组分具有含碳量高、芳香度较高及杂原子(S、N)含量低的特点,被广泛用作制备石油基人造炭材料的优选原料。为充分研究乙烯渣油沥青的成焦性,本研究选取四种溶剂(甲醇、正丁醇、正庚烷、二甲基亚砜)对乙烯渣油沥青进行萃取分离,获得八种乙烯渣油沥青组分(四种可溶组分和四种不溶组分),并对获得的乙烯渣油沥青组分进行热转化和炭化处理(热转化温度和炭化温度分别为500和1400 ℃)获得系列乙烯渣油沥青焦。采用红外光谱、热重分析仪、1H-NMR对乙烯渣油沥青组分的基础物性进行研究;利用偏光显微镜、X射线单晶衍射仪、拉曼光谱仪、扫描电子显微镜等对系列石油系沥青焦结构进行研究。结果表明,乙烯渣油沥青不溶组分芳香性略高于可溶组分,且不溶组分支链略少于可溶组分;不溶组分热转化和炭化后得到的乙烯渣油沥青焦显微强度高于可溶组分获得的乙烯渣油沥青焦,且乙烯渣油沥青沥青焦HS-C的真密度高达2.0554 g/cm3。
乙烯渣油沥青作为乙烯渣油中的重质组分具有含碳量高、芳香度较高及杂原子(S、N)含量低的特点,被广泛用作制备石油基人造炭材料的优选原料。为充分研究乙烯渣油沥青的成焦性,本研究选取四种溶剂(甲醇、正丁醇、正庚烷、二甲基亚砜)对乙烯渣油沥青进行萃取分离,获得八种乙烯渣油沥青组分(四种可溶组分和四种不溶组分),并对获得的乙烯渣油沥青组分进行热转化和炭化处理(热转化温度和炭化温度分别为500和1400 ℃)获得系列乙烯渣油沥青焦。采用红外光谱、热重分析仪、1H-NMR对乙烯渣油沥青组分的基础物性进行研究;利用偏光显微镜、X射线单晶衍射仪、拉曼光谱仪、扫描电子显微镜等对系列石油系沥青焦结构进行研究。结果表明,乙烯渣油沥青不溶组分芳香性略高于可溶组分,且不溶组分支链略少于可溶组分;不溶组分热转化和炭化后得到的乙烯渣油沥青焦显微强度高于可溶组分获得的乙烯渣油沥青焦,且乙烯渣油沥青沥青焦HS-C的真密度高达2.0554 g/cm3。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60456-1
摘要:
开发低成本、高性能的析氧电催化剂提升电化学水分解的效率,对于氢能的大规模利用具有重要的意义。钴基氢氧化物是一类极具潜力的析氧(OER)电催化剂,但是较差的导电性与催化活性严重制约了其应用推广。本研究采用一步低温熔盐法合成了铈掺杂的硝酸氢氧化钴电催化剂(Ce-CoNH/CF)。该催化剂在1 mol/L KOH电解液中具有最低的过电位(448 mV @ 1000 mA/cm2)。塔菲尔(Tafel)斜率、循环伏安(CV)和电化学阻抗谱(EIS)测试表明,快速的反应动力学、高效的电化学活性比表面积(ECSA)和极低的电荷转移电阻(Rct)共同作用使得催化剂具有优异的性能。并且在实验室条件下的模拟工业化测试也表明,Ce-CoNH/CF在工业级高温、高浓度的电解液(6 mol/L KOH,70 ℃)中同样展现出了出色的析氧性能。
开发低成本、高性能的析氧电催化剂提升电化学水分解的效率,对于氢能的大规模利用具有重要的意义。钴基氢氧化物是一类极具潜力的析氧(OER)电催化剂,但是较差的导电性与催化活性严重制约了其应用推广。本研究采用一步低温熔盐法合成了铈掺杂的硝酸氢氧化钴电催化剂(Ce-CoNH/CF)。该催化剂在1 mol/L KOH电解液中具有最低的过电位(448 mV @ 1000 mA/cm2)。塔菲尔(Tafel)斜率、循环伏安(CV)和电化学阻抗谱(EIS)测试表明,快速的反应动力学、高效的电化学活性比表面积(ECSA)和极低的电荷转移电阻(Rct)共同作用使得催化剂具有优异的性能。并且在实验室条件下的模拟工业化测试也表明,Ce-CoNH/CF在工业级高温、高浓度的电解液(6 mol/L KOH,70 ℃)中同样展现出了出色的析氧性能。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60457-3
摘要:
联环己烷是一种高储氢密度、低沸点储氢试剂。与联苯加氢相比,苯和环己烯烷基化制环己基苯再加氢是一种有望实现大规模制备联环己烷的途径。在成熟的烷基化技术基础上,需进一步开展高效环己基苯加氢催化剂的研发。本研究首先使用酸化的USY分子筛催化苯和环己烯烷基化至环己基苯,获得100%转化率和产物选择性。进一步通过原子层沉积(ALD)在γ-Al2O3表面预先沉积不同厚度的TiO2膜后再负载铂颗粒制得Pt/TiO2/γ-Al2O3催化剂,研究TiO2膜提升催化剂环己基苯加氢性能机制。TEM、CO脉冲吸附、CO-DRIFTs、准原位XPS、H-D交换和H2-TPR表征显示,与Pt/γ-Al2O3相比,Pt/TiO2/γ-Al2O3催化剂不改变Pt颗粒的分散度,但能够形成新的Pt-TiO2相互作用,提高铂表面电子密度、平面活性位点比例和降低氢溢流能垒,提升环己烷基苯加氢性能。研究为进一步发展联环己烷有机液态储氢试剂提供理论支持。相关金属-载体相互作用调控策略可应用于其他芳香性分子高效加氢催化剂的研制。
联环己烷是一种高储氢密度、低沸点储氢试剂。与联苯加氢相比,苯和环己烯烷基化制环己基苯再加氢是一种有望实现大规模制备联环己烷的途径。在成熟的烷基化技术基础上,需进一步开展高效环己基苯加氢催化剂的研发。本研究首先使用酸化的USY分子筛催化苯和环己烯烷基化至环己基苯,获得100%转化率和产物选择性。进一步通过原子层沉积(ALD)在γ-Al2O3表面预先沉积不同厚度的TiO2膜后再负载铂颗粒制得Pt/TiO2/γ-Al2O3催化剂,研究TiO2膜提升催化剂环己基苯加氢性能机制。TEM、CO脉冲吸附、CO-DRIFTs、准原位XPS、H-D交换和H2-TPR表征显示,与Pt/γ-Al2O3相比,Pt/TiO2/γ-Al2O3催化剂不改变Pt颗粒的分散度,但能够形成新的Pt-TiO2相互作用,提高铂表面电子密度、平面活性位点比例和降低氢溢流能垒,提升环己烷基苯加氢性能。研究为进一步发展联环己烷有机液态储氢试剂提供理论支持。相关金属-载体相互作用调控策略可应用于其他芳香性分子高效加氢催化剂的研制。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60476-7
摘要:
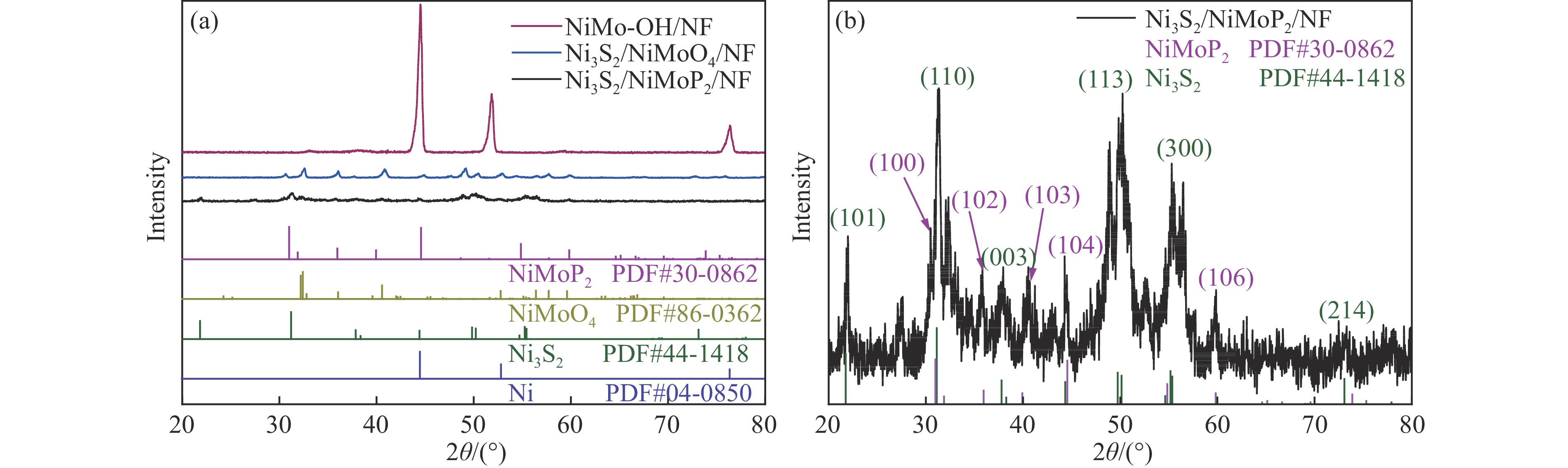
水-尿素电解是实现氨氮废水脱氮及高效绿色产氢的有效途径之一,但该技术面临的关键难题是缺乏高催化活性和大电流耐久性的双功能电催化剂。本文采用水热耦合气相磷化法,在泡沫镍(NF)上合成了一种贝壳状Ni3S2/NiMoP2异质结构,得益于其片状层叠的非均相纳米结构、丰富的氧空位和高效的电子传质,在析氢反应(HER)和尿素氧化反应(UOR)中表现出优异的催化活性,产生1000 mA/cm2的大电流密度分别仅需要−0.205和1.423 Vvs. RHE的超低电位。双功能Ni3S2/NiMoP2组装的水-尿素系统显著提升了电解活性,仅需要1.580 V的槽压来驱动500 mA/cm2电流密度实现HER和UOR,比全水解系统的电压低159 mV。此外,它在高电流条件下表现出优异的耐久性,可以稳定连续运行长达100小时。
水-尿素电解是实现氨氮废水脱氮及高效绿色产氢的有效途径之一,但该技术面临的关键难题是缺乏高催化活性和大电流耐久性的双功能电催化剂。本文采用水热耦合气相磷化法,在泡沫镍(NF)上合成了一种贝壳状Ni3S2/NiMoP2异质结构,得益于其片状层叠的非均相纳米结构、丰富的氧空位和高效的电子传质,在析氢反应(HER)和尿素氧化反应(UOR)中表现出优异的催化活性,产生1000 mA/cm2的大电流密度分别仅需要−0.205和1.423 Vvs. RHE的超低电位。双功能Ni3S2/NiMoP2组装的水-尿素系统显著提升了电解活性,仅需要1.580 V的槽压来驱动500 mA/cm2电流密度实现HER和UOR,比全水解系统的电压低159 mV。此外,它在高电流条件下表现出优异的耐久性,可以稳定连续运行长达100小时。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60443-3
摘要:
吡啶及其衍生物统称为吡啶碱,其广泛应用于农药、医药等领域。Chichibabin醛氨缩合反应是目前工业制取吡啶碱最广泛的路线。目前,使用最广泛的ZSM-5分子筛受制于硅铝骨架结构的不稳定性,高活性反应周期较短(5 h),针对这一问题,本研究选用热稳定性、水热稳定性优异的Silicalite-1分子筛,使用聚乙烯吡咯烷酮(PVP)作为胶体分散剂,在水热合成分子筛的过程中向骨架中引入Fe,结合XRD、SEM、TG、BET、NH3-TPD、Py-FTIR等表征方法探究了晶化条件对Silicalite-1分子筛结晶度、孔结构和酸性质的影响。实验结果表明,在晶种投入量15%、PVP添加量3.75%时产品相对结晶度达到最高(103%),粒径约为200 nm。改性后的Silicalite-1具有更丰富的酸位点,醛氨缩合反应的初始活性由66%增加至85%,在反应进行15 h后,原料转化率和吡啶碱收率分别保持在66%和40%以上。研究提出的原位改性Silicalite-1分子筛策略极大扩宽了纯硅沸石在酸催化领域的应用,具有显著的科研价值和工业化潜力。
吡啶及其衍生物统称为吡啶碱,其广泛应用于农药、医药等领域。Chichibabin醛氨缩合反应是目前工业制取吡啶碱最广泛的路线。目前,使用最广泛的ZSM-5分子筛受制于硅铝骨架结构的不稳定性,高活性反应周期较短(5 h),针对这一问题,本研究选用热稳定性、水热稳定性优异的Silicalite-1分子筛,使用聚乙烯吡咯烷酮(PVP)作为胶体分散剂,在水热合成分子筛的过程中向骨架中引入Fe,结合XRD、SEM、TG、BET、NH3-TPD、Py-FTIR等表征方法探究了晶化条件对Silicalite-1分子筛结晶度、孔结构和酸性质的影响。实验结果表明,在晶种投入量15%、PVP添加量3.75%时产品相对结晶度达到最高(103%),粒径约为200 nm。改性后的Silicalite-1具有更丰富的酸位点,醛氨缩合反应的初始活性由66%增加至85%,在反应进行15 h后,原料转化率和吡啶碱收率分别保持在66%和40%以上。研究提出的原位改性Silicalite-1分子筛策略极大扩宽了纯硅沸石在酸催化领域的应用,具有显著的科研价值和工业化潜力。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60453-6
摘要:
生物质衍生物乙酰丙酸甲酯(ML)加氢制备高值化学品γ-戊内酯(GVL)通常需要在高压氢气中进行,存在成本高和危险性大等问题。本研究采用Pt/CoxAl催化剂,将ML加氢反应与甲醇水液相重整(APRM)相耦合制备GVL,甲醇重整所获得的氢气原位用于ML的加氢反应,避免了外部氢源的使用,并考察了催化剂组成、反应溶液浓度和反应温度等条件对催化反应性能的影响。结果表明,Pt/CoxAl催化剂在该耦合反应体系中具有优异的催化性能,GVL收率高达98.2%,六次循环后性能仍保持稳定。多种表征手段证明,Pt0是ARPM和ML加氢反应的活性中心,Brønsted酸位点则促进ML水解和中间体的内酯化反应,两者之间的协同作用推动了GVL的生成。Pt与CoxAl载体之间存在强相互作用,Co含量适宜时,Pt/CoxAl催化剂具有较高的Pt分散度和丰富的Brønsted酸位点,因而表现出优异的催化性能。这些结果对探索新型高效的生物质衍生物制备燃料和化学品反应过程具有重要的参考价值。
生物质衍生物乙酰丙酸甲酯(ML)加氢制备高值化学品γ-戊内酯(GVL)通常需要在高压氢气中进行,存在成本高和危险性大等问题。本研究采用Pt/CoxAl催化剂,将ML加氢反应与甲醇水液相重整(APRM)相耦合制备GVL,甲醇重整所获得的氢气原位用于ML的加氢反应,避免了外部氢源的使用,并考察了催化剂组成、反应溶液浓度和反应温度等条件对催化反应性能的影响。结果表明,Pt/CoxAl催化剂在该耦合反应体系中具有优异的催化性能,GVL收率高达98.2%,六次循环后性能仍保持稳定。多种表征手段证明,Pt0是ARPM和ML加氢反应的活性中心,Brønsted酸位点则促进ML水解和中间体的内酯化反应,两者之间的协同作用推动了GVL的生成。Pt与CoxAl载体之间存在强相互作用,Co含量适宜时,Pt/CoxAl催化剂具有较高的Pt分散度和丰富的Brønsted酸位点,因而表现出优异的催化性能。这些结果对探索新型高效的生物质衍生物制备燃料和化学品反应过程具有重要的参考价值。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024015
摘要:
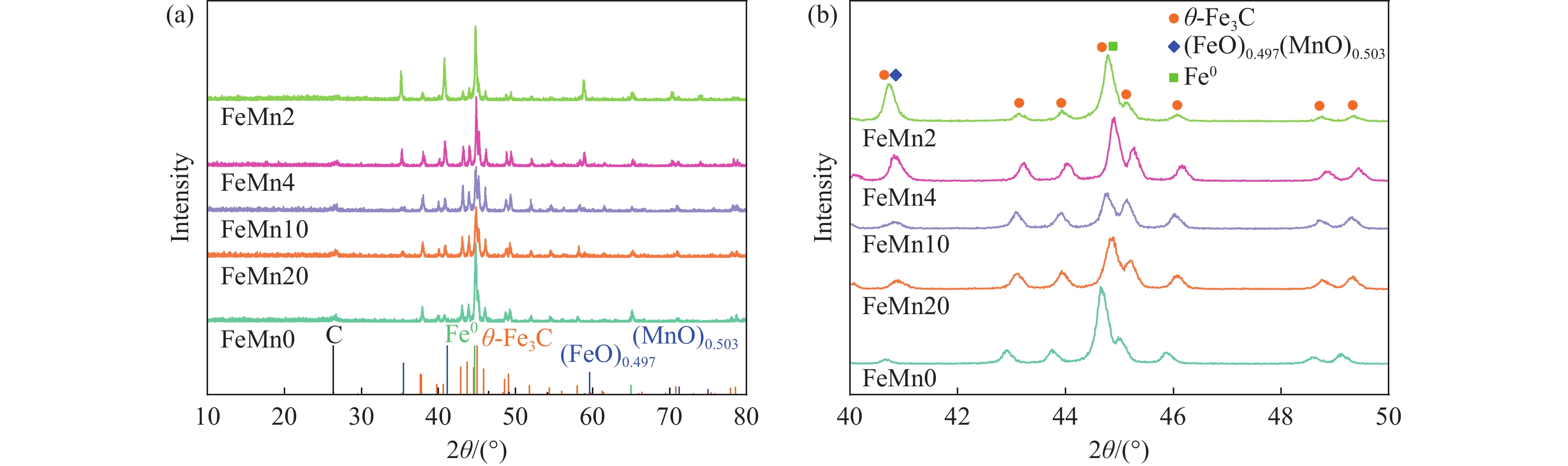
本研究制备了系列含Fe、Mn的有机凝胶前驱体,在氩气氛围下通过高温热处理,凝胶中铁物种被有机物原位分解进行还原和碳化,制备出了θ-Fe3C含量不同的费托合成催化剂。采用XRD、N2吸附-脱附、Raman、CO-TPD、CO2-TPD、XPS和TEM等手段对催化剂的结构组成、表面性质以及活性物种的电子价态进行了系统的表征和分析。实验结果表明,热处理后获得的催化剂含石墨碳、θ-Fe3C、Fe0和(FeO)0.497(MnO)0.503物相,费托反应后催化剂的结构保持稳定,物相种类不发生变化。考察了反应条件对催化性能的影响,FeMn10催化剂具有较优的催化性能,CO转化率为57.3%,低碳烯烃(C2−C4)选择性为37.1%,其中,θ-Fe3C物相作为催化活性位点,催化剂的活性和低碳烯烃的选择性与θ-Fe3C的含量具有正相关性。
本研究制备了系列含Fe、Mn的有机凝胶前驱体,在氩气氛围下通过高温热处理,凝胶中铁物种被有机物原位分解进行还原和碳化,制备出了θ-Fe3C含量不同的费托合成催化剂。采用XRD、N2吸附-脱附、Raman、CO-TPD、CO2-TPD、XPS和TEM等手段对催化剂的结构组成、表面性质以及活性物种的电子价态进行了系统的表征和分析。实验结果表明,热处理后获得的催化剂含石墨碳、θ-Fe3C、Fe0和(FeO)0.497(MnO)0.503物相,费托反应后催化剂的结构保持稳定,物相种类不发生变化。考察了反应条件对催化性能的影响,FeMn10催化剂具有较优的催化性能,CO转化率为57.3%,低碳烯烃(C2−C4)选择性为37.1%,其中,θ-Fe3C物相作为催化活性位点,催化剂的活性和低碳烯烃的选择性与θ-Fe3C的含量具有正相关性。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60455-X
摘要:
焦炉煤气制甲醇的复杂工况给铜基甲醇合成催化剂的稳定性和寿命带来挑战。本工作制备了一系列不同镁含量的锌孔雀石样品,采用原位X射线衍射、热重-质谱、N2物理吸附、H2程序升温还原、CO2程序升温脱附等方法对锌孔雀石及焙烧后的样品进行表征,考察镁元素的添加对锌孔雀石结构和甲醇合成催化性能的影响。结果发现,镁元素的添加提高了锌孔雀石结构内部的铜取代程度,促进了焙烧后催化剂内部高温碳酸盐的形成。随着镁含量的增加,焙烧后催化剂的比表面积逐渐增大,还原后的单质铜晶粒尺寸逐渐减小。原位XRD结果表明,少量镁元素在催化剂热处理过程中能有效抑制单质铜晶粒尺寸生长。评价显示催化剂的初始活性随着镁元素的添加呈现先升高后降低的趋势,热处理后含镁催化剂活性仍保持在相对较高的水平。镁元素的适量添加有助于提高铜基甲醇合成催化剂初始活性和热稳定性。
焦炉煤气制甲醇的复杂工况给铜基甲醇合成催化剂的稳定性和寿命带来挑战。本工作制备了一系列不同镁含量的锌孔雀石样品,采用原位X射线衍射、热重-质谱、N2物理吸附、H2程序升温还原、CO2程序升温脱附等方法对锌孔雀石及焙烧后的样品进行表征,考察镁元素的添加对锌孔雀石结构和甲醇合成催化性能的影响。结果发现,镁元素的添加提高了锌孔雀石结构内部的铜取代程度,促进了焙烧后催化剂内部高温碳酸盐的形成。随着镁含量的增加,焙烧后催化剂的比表面积逐渐增大,还原后的单质铜晶粒尺寸逐渐减小。原位XRD结果表明,少量镁元素在催化剂热处理过程中能有效抑制单质铜晶粒尺寸生长。评价显示催化剂的初始活性随着镁元素的添加呈现先升高后降低的趋势,热处理后含镁催化剂活性仍保持在相对较高的水平。镁元素的适量添加有助于提高铜基甲醇合成催化剂初始活性和热稳定性。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60444-5
摘要:
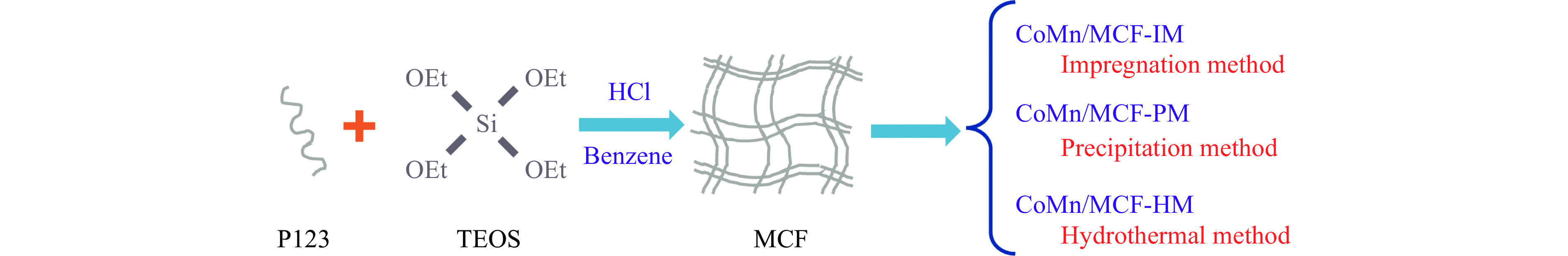
本研究采用浸渍法、沉淀法和水热合成法制备了一系列泡沫硅负载CoMn基催化剂,并结合XRD、H2-TPR、N2物理吸附-脱附、TEM和XPS等表征技术考察了制备方法对催化剂在合成气制低碳醇反应中的性能影响。研究表明,催化剂表面存在Co2+(Co2C)、Co0物种,水热合成法制备的催化剂表面Co2C-Co0活性位点存在良好的协同作用有利于醇的生成,较高比例的Co2C也促进了CO的非解离吸附和插入,从而呈现最高的醇选择性。在t=260 ℃,p=5.0 MPa,GHSV=4500 h−1,H2/CO(体积比)=2∶1的反应条件下,该泡沫催化剂可实现CO转化率11.1%,总醇选择性34.7%,C2+OH选择性34.5%的反应性能。
本研究采用浸渍法、沉淀法和水热合成法制备了一系列泡沫硅负载CoMn基催化剂,并结合XRD、H2-TPR、N2物理吸附-脱附、TEM和XPS等表征技术考察了制备方法对催化剂在合成气制低碳醇反应中的性能影响。研究表明,催化剂表面存在Co2+(Co2C)、Co0物种,水热合成法制备的催化剂表面Co2C-Co0活性位点存在良好的协同作用有利于醇的生成,较高比例的Co2C也促进了CO的非解离吸附和插入,从而呈现最高的醇选择性。在t=260 ℃,p=5.0 MPa,GHSV=4500 h−1,H2/CO(体积比)=2∶1的反应条件下,该泡沫催化剂可实现CO转化率11.1%,总醇选择性34.7%,C2+OH选择性34.5%的反应性能。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60472-X
摘要:
铂是光催化二氧化碳(CO2)还原制甲烷(CH4)最有效的助催化剂之一,但仍面临CO2还原速率和CH4选择性低的难题。本研究利用原子层沉积(ALD)制备了Pt颗粒尺寸可调(0.55−1.80 nm)的Pt/TiO2催化剂并将其用于光催化还原CO2制CH4反应。Pt/TiO2催化CO2还原速率和CH4选择性随Pt颗粒尺寸增加呈现出先增加后降低的火山型趋势,其中,Pt颗粒尺寸为1.35 nm时,催化剂的甲烷收率最高(71.9 μmol/(g·h))且没有检测到H2的生成,烷烃产物(CH4,C2H6,C3H8)选择性为100%,其中CH4的电子选择性和碳基产物中选择性分别高达81.69 %和90.20 %。CO-DRIFTS,XPS,CO2-TPD,H2O-TPD,H2-TPD等分析结果表明,Pt尺寸为1.35 nm时,具有最优的活化CO2能力、合适的活化H2O能力和较高的氢脱附温度,使H2O活化生成活性氢速率与CO2还原消耗活性氢速率相匹配,展示出最佳性能。本研究对开发高活性和高选择性的光催化CO2还原催化剂具有重要参考价值。
铂是光催化二氧化碳(CO2)还原制甲烷(CH4)最有效的助催化剂之一,但仍面临CO2还原速率和CH4选择性低的难题。本研究利用原子层沉积(ALD)制备了Pt颗粒尺寸可调(0.55−1.80 nm)的Pt/TiO2催化剂并将其用于光催化还原CO2制CH4反应。Pt/TiO2催化CO2还原速率和CH4选择性随Pt颗粒尺寸增加呈现出先增加后降低的火山型趋势,其中,Pt颗粒尺寸为1.35 nm时,催化剂的甲烷收率最高(71.9 μmol/(g·h))且没有检测到H2的生成,烷烃产物(CH4,C2H6,C3H8)选择性为100%,其中CH4的电子选择性和碳基产物中选择性分别高达81.69 %和90.20 %。CO-DRIFTS,XPS,CO2-TPD,H2O-TPD,H2-TPD等分析结果表明,Pt尺寸为1.35 nm时,具有最优的活化CO2能力、合适的活化H2O能力和较高的氢脱附温度,使H2O活化生成活性氢速率与CO2还原消耗活性氢速率相匹配,展示出最佳性能。本研究对开发高活性和高选择性的光催化CO2还原催化剂具有重要参考价值。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60468-8
摘要:
In recent years, photocatalytic N2 reduction for ammonia synthesis at room temperature and atmospheric pressure has gradually become a research hotspot, exhibiting extremely high development potential. However, the low photogenerated charge separation efficiency and the lack of effective active sites seriously constrain the reaction efficiencies of semiconductor photocatalysts for N2 reduction of ammonia synthesis. Therefore, the rational design of catalytic materials is the key to enhance the photocatalytic N2 reduction reaction of ammonia synthesis. Transition metal Ru as the active center not only accelerates the adsorption and activation of N2 molecules, but also has good selectivity for N2 reduction. Moreover, the interaction between the metal and the support can effectively regulate the electronic structure of the active site, accelerate the photogenerated electron transfer, and significantly enhance the photocatalytic activity. Based on this, this review systematically investigates the Ru co-semiconductors to realize efficient photocatalytic N2 reduction for ammonia synthesis, and introduces its basic principles. Specifically, the Ru co-semiconductor photocatalytic material systems are introduced, such as TiO2-based, g-C3N4-based, and metal oxide materials, including the design of catalysts, crystal structures, and other characteristics. In addition, the modification strategies of photocatalytic N2 reduction ammonia synthesis materials are also presented, including loading/doping, defect engineering, construction of heterojunctions, and crystal surface modulation. Furthermore, the progress and shortcomings of the application of Ru co-semiconductors in these processes are summarized and comprehensively discussed, and the future outlook of Ru co-semiconductors in photocatalytic N2 reduction ammonia synthesis applications is proposed.
In recent years, photocatalytic N2 reduction for ammonia synthesis at room temperature and atmospheric pressure has gradually become a research hotspot, exhibiting extremely high development potential. However, the low photogenerated charge separation efficiency and the lack of effective active sites seriously constrain the reaction efficiencies of semiconductor photocatalysts for N2 reduction of ammonia synthesis. Therefore, the rational design of catalytic materials is the key to enhance the photocatalytic N2 reduction reaction of ammonia synthesis. Transition metal Ru as the active center not only accelerates the adsorption and activation of N2 molecules, but also has good selectivity for N2 reduction. Moreover, the interaction between the metal and the support can effectively regulate the electronic structure of the active site, accelerate the photogenerated electron transfer, and significantly enhance the photocatalytic activity. Based on this, this review systematically investigates the Ru co-semiconductors to realize efficient photocatalytic N2 reduction for ammonia synthesis, and introduces its basic principles. Specifically, the Ru co-semiconductor photocatalytic material systems are introduced, such as TiO2-based, g-C3N4-based, and metal oxide materials, including the design of catalysts, crystal structures, and other characteristics. In addition, the modification strategies of photocatalytic N2 reduction ammonia synthesis materials are also presented, including loading/doping, defect engineering, construction of heterojunctions, and crystal surface modulation. Furthermore, the progress and shortcomings of the application of Ru co-semiconductors in these processes are summarized and comprehensively discussed, and the future outlook of Ru co-semiconductors in photocatalytic N2 reduction ammonia synthesis applications is proposed.
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60454-8
摘要:
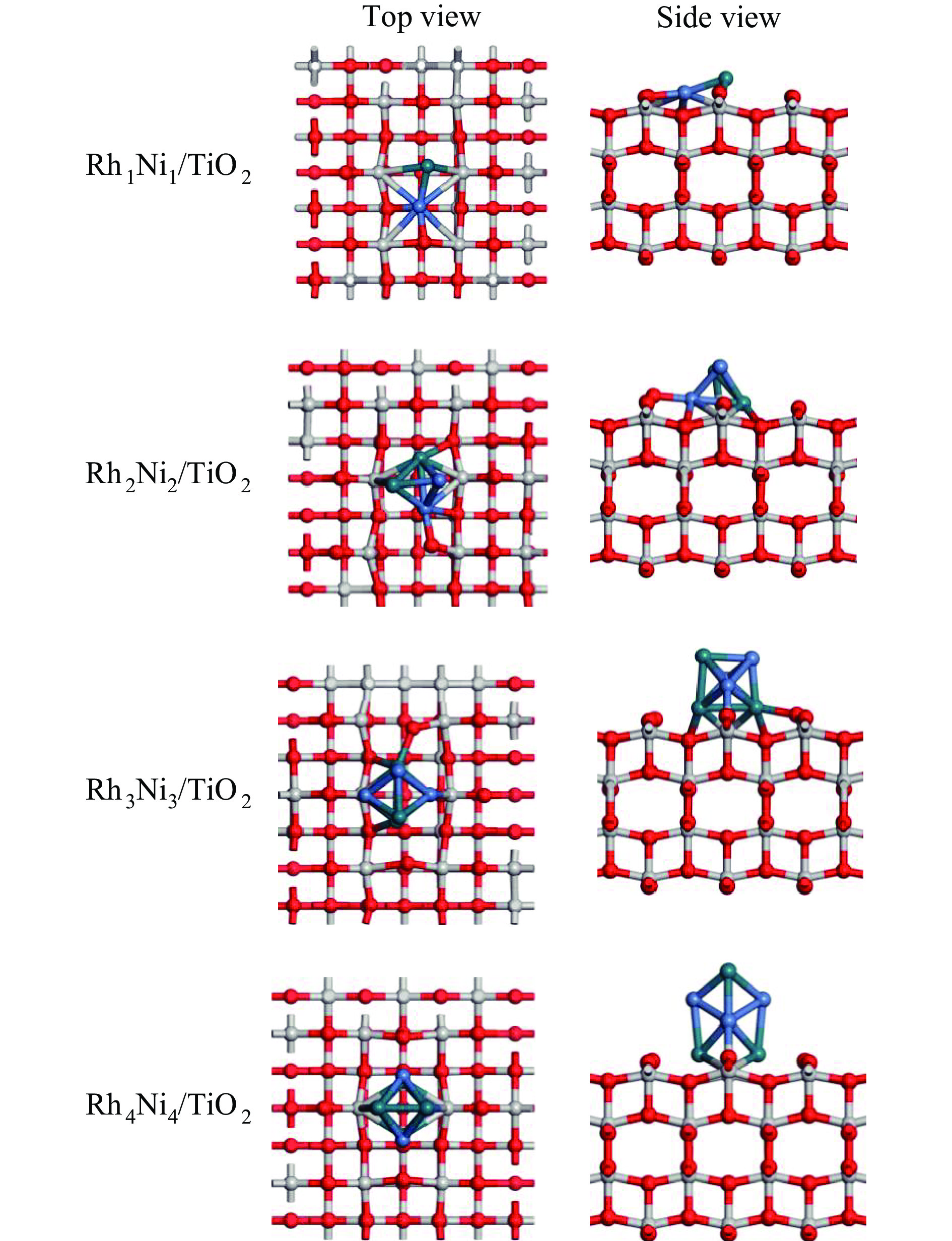
为明确RhnNin合金团簇尺寸诱导的金属-载体相互作用对合成气制乙醇反应性能的调控机制,本工作采用密度泛函理论(DFT)和微观动力学方法研究了不同Rh-Ni合金团簇尺寸RhnNin/TiO2(n = 1、2、3、4)上合成气制乙醇反应。结果表明,Rh1Ni1/TiO2和Rh3Ni3/TiO2能够显著促进CO活化转化及C−C链的形成,并抑制甲烷的生成。其中,Rh1Ni1/TiO2表现出最高的乙醇生成活性和相对选择性。电子性质分析表明,在Rh1Ni1/TiO2催化剂上,合金团簇上Ni原子及载体上Ti和O原子向Rh原子转移的电荷最多,合金团簇上Rh-Ni间相互作用最强,且合金团簇与TiO2载体间的相互作用最强,催化剂的催化活性最高。在525 K下,从头算分子动力学模拟(AIMD)模拟显示Rh1Ni1/TiO2催化剂具有较高的热稳定性。
为明确RhnNin合金团簇尺寸诱导的金属-载体相互作用对合成气制乙醇反应性能的调控机制,本工作采用密度泛函理论(DFT)和微观动力学方法研究了不同Rh-Ni合金团簇尺寸RhnNin/TiO2(n = 1、2、3、4)上合成气制乙醇反应。结果表明,Rh1Ni1/TiO2和Rh3Ni3/TiO2能够显著促进CO活化转化及C−C链的形成,并抑制甲烷的生成。其中,Rh1Ni1/TiO2表现出最高的乙醇生成活性和相对选择性。电子性质分析表明,在Rh1Ni1/TiO2催化剂上,合金团簇上Ni原子及载体上Ti和O原子向Rh原子转移的电荷最多,合金团簇上Rh-Ni间相互作用最强,且合金团簇与TiO2载体间的相互作用最强,催化剂的催化活性最高。在525 K下,从头算分子动力学模拟(AIMD)模拟显示Rh1Ni1/TiO2催化剂具有较高的热稳定性。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024026
摘要:
以低温等离子体和催化剂耦合法将H2S和CO2混合酸气一步转化为合成气,既完成了两者清洁化处理,又实现了资源化利用,是一条制备合成气的新路线。本研究采用铜、锌为助剂对硫化钼催化剂改性,显著提升了其催化H2S-CO2制合成气反应性能。结合多种分析表征手段对比两种助剂引入后对硫化钼催化剂结构、组成、形貌、化合价态等物化特征的影响。通过控制低温等离子体放电条件,深入探究了两种助剂对低温等离子体下催化转化H2S和CO2酸气制合成气的反应性能影响规律和关键因素。研究发现,引入铜、锌助剂后,硫化钼活性相粒径减小且分散度高,提供了更多活性位点。同时也增强了对H2S和CO2分子吸附强度,从而更利于H2S和CO2分子的吸附活化,揭示出低温等离子体与改性硫化钼催化剂协同反应的构效关联。有关理论研究丰富拓展了低温等离子体-催化协同理论,并为改性硫化钼材料的合成提供借鉴。
以低温等离子体和催化剂耦合法将H2S和CO2混合酸气一步转化为合成气,既完成了两者清洁化处理,又实现了资源化利用,是一条制备合成气的新路线。本研究采用铜、锌为助剂对硫化钼催化剂改性,显著提升了其催化H2S-CO2制合成气反应性能。结合多种分析表征手段对比两种助剂引入后对硫化钼催化剂结构、组成、形貌、化合价态等物化特征的影响。通过控制低温等离子体放电条件,深入探究了两种助剂对低温等离子体下催化转化H2S和CO2酸气制合成气的反应性能影响规律和关键因素。研究发现,引入铜、锌助剂后,硫化钼活性相粒径减小且分散度高,提供了更多活性位点。同时也增强了对H2S和CO2分子吸附强度,从而更利于H2S和CO2分子的吸附活化,揭示出低温等离子体与改性硫化钼催化剂协同反应的构效关联。有关理论研究丰富拓展了低温等离子体-催化协同理论,并为改性硫化钼材料的合成提供借鉴。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60460-3
摘要:
本研究采用密度泛函理论 (DFT) 和微动力学模型分析了 Rh16/In2O3 催化剂上二氧化碳 (CO2) 氢化成甲醇 (CH3OH) 的情况;研究了 Rh16/In2O3 界面上 H2 的自发解离和 CO2 的有效吸附,其中, In2O3 中的氧空位提供了有利的效果。此外,Bader 电荷分析显示 Rh16 上带有轻微的正电荷,这对于理解催化剂的电子特性和活性非常重要。证实了RWGS+CO-Hydro 途径是甲醇合成的主要途径,其特点是经过一系列中间转化:CO2*→COOH*→CO*+OH*→HCO*→CH2O*→CH2OH*→ CH3OH*。在不同温度 (373−873K) 和压力 (10−2−103 bar) 下进行的反应速率控制程度分析 (DRC) 揭示了两个关键的动力学现象:在较低温度和较高压力下,转化步骤 CO* + H* → HCO * 显着影响总体反应速率;而在较高温度下,CH2O* + H* → CH3O* 的步骤占主导地位。
本研究采用密度泛函理论 (DFT) 和微动力学模型分析了 Rh16/In2O3 催化剂上二氧化碳 (CO2) 氢化成甲醇 (CH3OH) 的情况;研究了 Rh16/In2O3 界面上 H2 的自发解离和 CO2 的有效吸附,其中, In2O3 中的氧空位提供了有利的效果。此外,Bader 电荷分析显示 Rh16 上带有轻微的正电荷,这对于理解催化剂的电子特性和活性非常重要。证实了RWGS+CO-Hydro 途径是甲醇合成的主要途径,其特点是经过一系列中间转化:CO2*→COOH*→CO*+OH*→HCO*→CH2O*→CH2OH*→ CH3OH*。在不同温度 (373−873K) 和压力 (10−2−103 bar) 下进行的反应速率控制程度分析 (DRC) 揭示了两个关键的动力学现象:在较低温度和较高压力下,转化步骤 CO* + H* → HCO * 显着影响总体反应速率;而在较高温度下,CH2O* + H* → CH3O* 的步骤占主导地位。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60458-5
摘要:
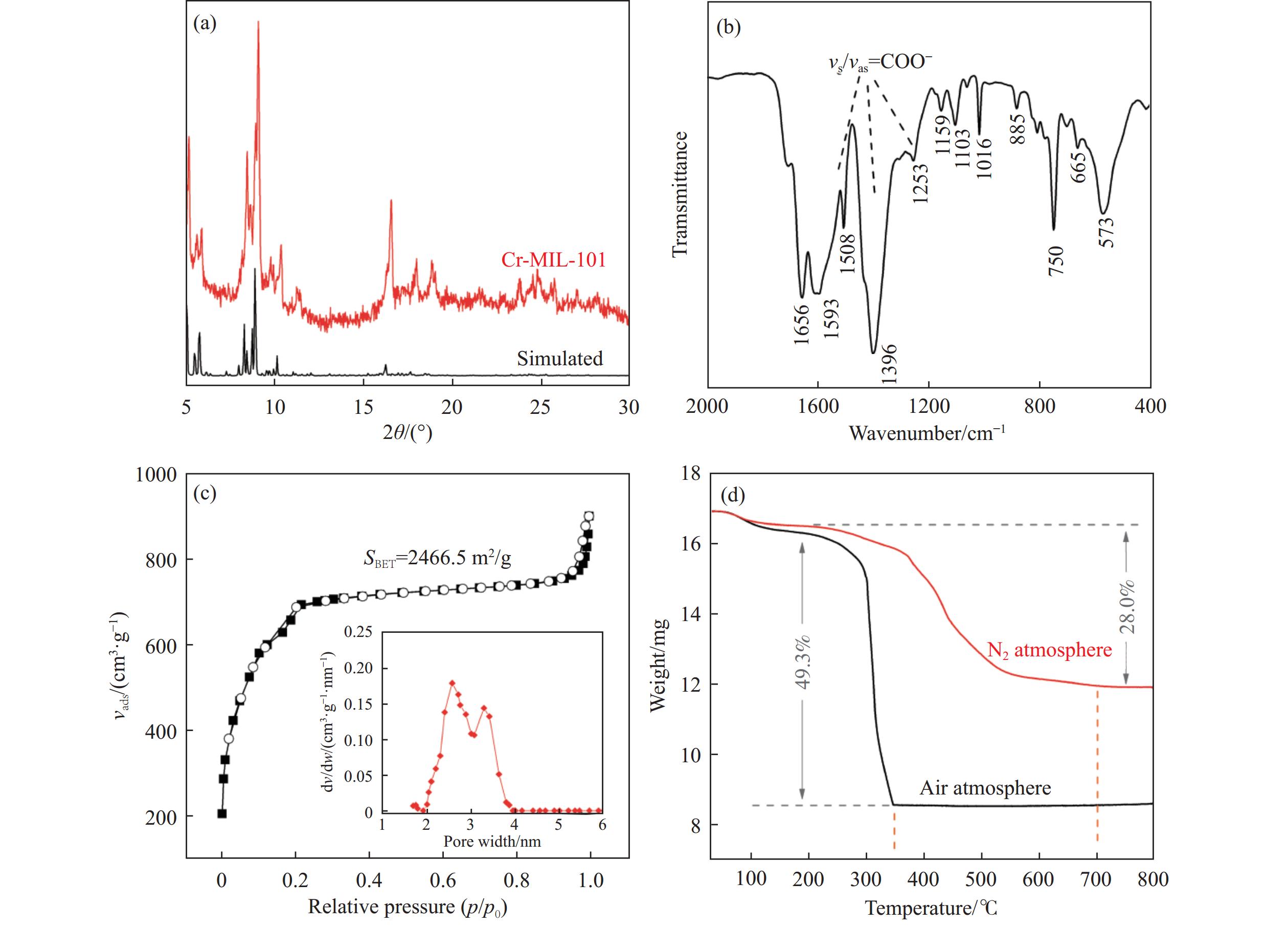
通过热解大比表面Cr-MIL-101制备纳米Cr2O3(n-Cr2O3),考察其催化正己烷脱氢反应性能,并比较与沉淀法p-Cr2O3-1、焙烧铬盐得到的p-Cr2O3-2以及工业Cr2O3/Al2O3催化正己烷脱氢活性差异。n-Cr2O3能够催化正己烷高效脱氢为己烯和苯,并且其催化脱氢活性与焙烧温度有关。600 ℃焙烧的n-Cr2O3催化正己烷脱氢转化率最高40.6%,对产物己烯和苯的选择性分别为20.1%和69.3%。提高焙烧温度,n-Cr2O3催化正己烷脱氢活性下降但稳定性增强,催化剂积炭量减少。p-Cr2O3-1和p-Cr2O3-2催化正己烷脱氢转化率很低(<7.5%),比活性分别为1.5和1.7 g/(m2·h),低于n-Cr2O3-600的(2.0 g/(m2·h))。通过BET、XRD、TEM和FT-IR等表征发现,n-Cr2O3为具有较大比表面的纳米颗粒(10−20 nm),多暴露晶面和脱氢活性位,而p-Cr2O3是比表面非常小的大颗粒,所暴露脱氢活性位少。相比之下,Cr2O3/Al2O3催化剂由于大比表面Al2O3的分散作用,催化正己烷脱氢效率更高(2.4 g/(m2·h))。因此,由Cr-MIL-101焙烧得到的n-Cr2O3催化正己烷脱氢的高活性源于这种纳米Cr2O3所具有的独特性质:小颗粒,大比表面,多暴露活性位。
通过热解大比表面Cr-MIL-101制备纳米Cr2O3(n-Cr2O3),考察其催化正己烷脱氢反应性能,并比较与沉淀法p-Cr2O3-1、焙烧铬盐得到的p-Cr2O3-2以及工业Cr2O3/Al2O3催化正己烷脱氢活性差异。n-Cr2O3能够催化正己烷高效脱氢为己烯和苯,并且其催化脱氢活性与焙烧温度有关。600 ℃焙烧的n-Cr2O3催化正己烷脱氢转化率最高40.6%,对产物己烯和苯的选择性分别为20.1%和69.3%。提高焙烧温度,n-Cr2O3催化正己烷脱氢活性下降但稳定性增强,催化剂积炭量减少。p-Cr2O3-1和p-Cr2O3-2催化正己烷脱氢转化率很低(<7.5%),比活性分别为1.5和1.7 g/(m2·h),低于n-Cr2O3-600的(2.0 g/(m2·h))。通过BET、XRD、TEM和FT-IR等表征发现,n-Cr2O3为具有较大比表面的纳米颗粒(10−20 nm),多暴露晶面和脱氢活性位,而p-Cr2O3是比表面非常小的大颗粒,所暴露脱氢活性位少。相比之下,Cr2O3/Al2O3催化剂由于大比表面Al2O3的分散作用,催化正己烷脱氢效率更高(2.4 g/(m2·h))。因此,由Cr-MIL-101焙烧得到的n-Cr2O3催化正己烷脱氢的高活性源于这种纳米Cr2O3所具有的独特性质:小颗粒,大比表面,多暴露活性位。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60459-7
摘要:
采用共沉淀法制备了一系列Cu/ZnO/Al2O3催化剂,通过XRD、BET、H2-TPR、N2O化学吸附、XPS表征技术,研究了Al2O3前驱体对催化剂结构的影响,同时对其在甲醇重整制氢中的性能进行了考察。结果表明,当Al3+与Cu2+、Zn2+同时共沉淀时,Al3+对碱式碳酸盐中Cu2+-Zn2+部分取代生成类水滑石结构,增强了Zn-Al之间的相互作用。相反,在Cu2+、Zn2+完成共沉淀后,引入Al2O3前驱体对消除Al3+对碱式碳酸盐中Cu-Zn取代的不良影响具有积极作用,有利于促进Cu-ZnO间的相互作用、CuO物种的分散和催化剂的还原,进一步促进表面Cu的分散,有利于其活性的提升。其中,以拟薄水铝石为铝源制备的催化剂呈现出优异的活性。在水醇物质的量比为1.2,反应温度为493 K的条件下,甲醇转化率可达94.8%,H2时空收率可达97.5 mol/(kg·h),并且连续运行25 h其活性仍保持相对稳定。在反应条件下,经过723 K的10 h热处理后,该催化剂的活性损失率仅为5.37%。
采用共沉淀法制备了一系列Cu/ZnO/Al2O3催化剂,通过XRD、BET、H2-TPR、N2O化学吸附、XPS表征技术,研究了Al2O3前驱体对催化剂结构的影响,同时对其在甲醇重整制氢中的性能进行了考察。结果表明,当Al3+与Cu2+、Zn2+同时共沉淀时,Al3+对碱式碳酸盐中Cu2+-Zn2+部分取代生成类水滑石结构,增强了Zn-Al之间的相互作用。相反,在Cu2+、Zn2+完成共沉淀后,引入Al2O3前驱体对消除Al3+对碱式碳酸盐中Cu-Zn取代的不良影响具有积极作用,有利于促进Cu-ZnO间的相互作用、CuO物种的分散和催化剂的还原,进一步促进表面Cu的分散,有利于其活性的提升。其中,以拟薄水铝石为铝源制备的催化剂呈现出优异的活性。在水醇物质的量比为1.2,反应温度为493 K的条件下,甲醇转化率可达94.8%,H2时空收率可达97.5 mol/(kg·h),并且连续运行25 h其活性仍保持相对稳定。在反应条件下,经过723 K的10 h热处理后,该催化剂的活性损失率仅为5.37%。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024030
摘要:
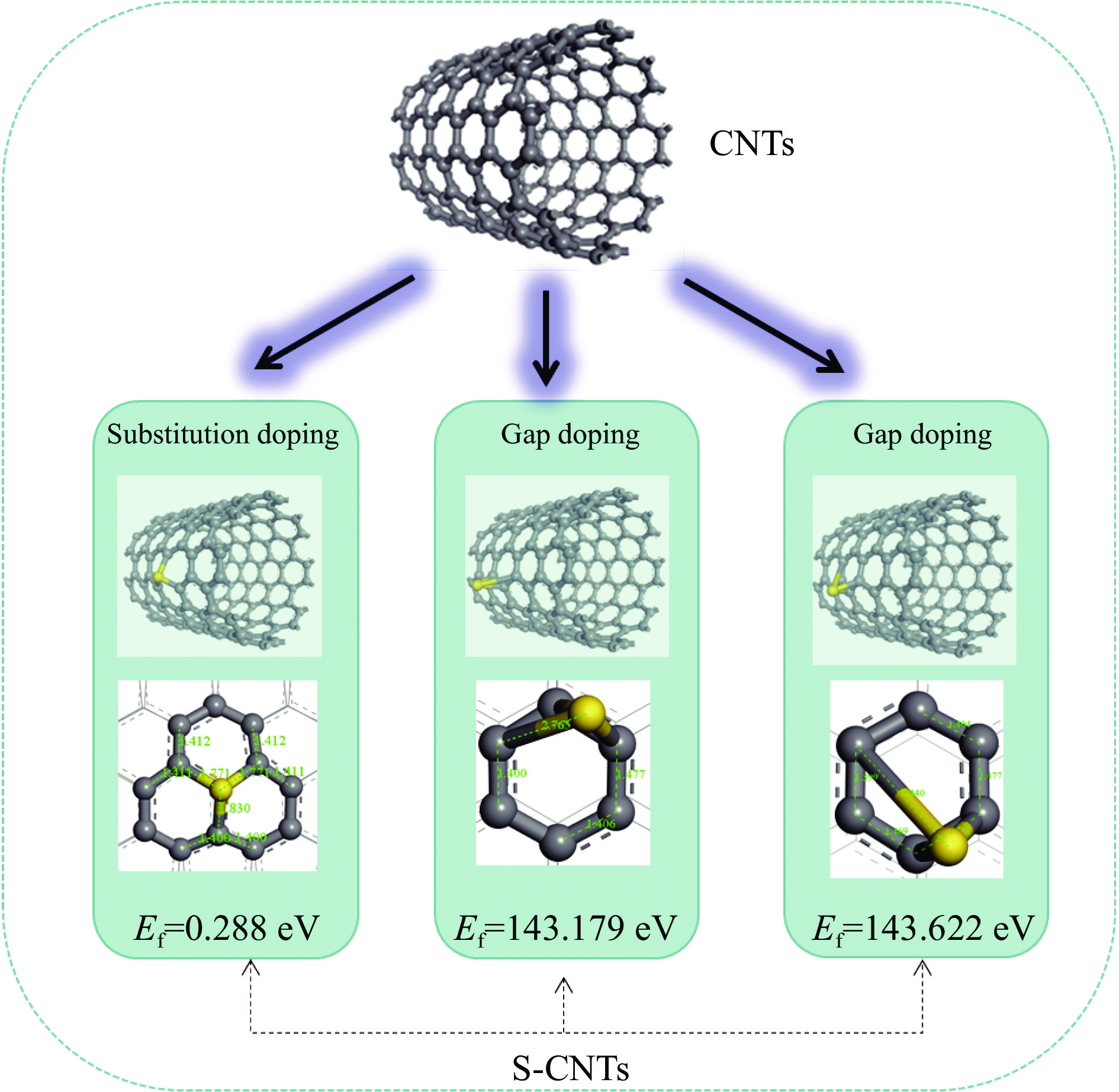
醇胺法吸收CO2是目前最成熟的碳捕集技术,虽然吸收效率高、稳定性好,但过高的解吸能耗限制其大规模工业推广应用。催化解吸提供了降低CO2解吸能耗的可能性。本工作利用基于密度泛函理论(DFT)的量子化学模拟方法,探索了硫掺杂碳纳米管(S-CNTs)催化单乙醇胺(MEA)溶液吸收-解吸CO2反应机理。通过过渡态搜索发现,以S-CNTs为催化剂的解吸过程,决速步骤的反应能垒降低了1.15 kcal/mol。局部态密度分析表明(PDOS),产物氨基甲酸酯吸附质子化胺MEACOO-_MEAH+和吸收中间产物MEA+COO−中的C、N、O原子在CNTS和S-CNTs表面吸附时PDOS差距较大。此外,与未改性CNTs相比,S-CNTS上电荷密度增加,掺杂的硫原子附近碳原子具有明显的电负性。相比于CNTs,吸收中间产物MEA+COO−和吸收产物MEACOO−-MEAH+均向S-CNTs转移了更多的电荷,表明更多的电荷转移有利于CO2的释放。本工作旨在通过CO2催化解吸机理的研究为催化剂的设计提供一定的理论依据。
醇胺法吸收CO2是目前最成熟的碳捕集技术,虽然吸收效率高、稳定性好,但过高的解吸能耗限制其大规模工业推广应用。催化解吸提供了降低CO2解吸能耗的可能性。本工作利用基于密度泛函理论(DFT)的量子化学模拟方法,探索了硫掺杂碳纳米管(S-CNTs)催化单乙醇胺(MEA)溶液吸收-解吸CO2反应机理。通过过渡态搜索发现,以S-CNTs为催化剂的解吸过程,决速步骤的反应能垒降低了1.15 kcal/mol。局部态密度分析表明(PDOS),产物氨基甲酸酯吸附质子化胺MEACOO-_MEAH+和吸收中间产物MEA+COO−中的C、N、O原子在CNTS和S-CNTs表面吸附时PDOS差距较大。此外,与未改性CNTs相比,S-CNTS上电荷密度增加,掺杂的硫原子附近碳原子具有明显的电负性。相比于CNTs,吸收中间产物MEA+COO−和吸收产物MEACOO−-MEAH+均向S-CNTs转移了更多的电荷,表明更多的电荷转移有利于CO2的释放。本工作旨在通过CO2催化解吸机理的研究为催化剂的设计提供一定的理论依据。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024023
摘要:
苯与合成气的烷基化反应旨在利用非石油资源生产甲苯、二甲苯等烷基苯,本工作采用甲醇蒸汽预积炭制备一系列的ZSM-5分子筛,并与ZnCrOx复合形成双功能催化剂,探索对苯和合成气烷基化制烷基苯的影响。通过对预积炭时间的考察,发现适当的预积炭能显著提高ZSM-5分子筛的催化性能。在450 ℃、4.0 MPa的反应条件下,预积炭改性24 h的催化剂苯转化率达到20.18%。对预积炭改性的ZSM-5分子筛和反应后的复合催化剂进行分析,表明预积炭改变了分子筛的酸性。预积炭覆盖了ZSM-5分子筛上的一些B酸位点,降低了B酸/L酸比,从而提高苯的转化率。最后,通过烃池机理对苯与合成气烷基化反应的失活机理进行合理推测。
苯与合成气的烷基化反应旨在利用非石油资源生产甲苯、二甲苯等烷基苯,本工作采用甲醇蒸汽预积炭制备一系列的ZSM-5分子筛,并与ZnCrOx复合形成双功能催化剂,探索对苯和合成气烷基化制烷基苯的影响。通过对预积炭时间的考察,发现适当的预积炭能显著提高ZSM-5分子筛的催化性能。在450 ℃、4.0 MPa的反应条件下,预积炭改性24 h的催化剂苯转化率达到20.18%。对预积炭改性的ZSM-5分子筛和反应后的复合催化剂进行分析,表明预积炭改变了分子筛的酸性。预积炭覆盖了ZSM-5分子筛上的一些B酸位点,降低了B酸/L酸比,从而提高苯的转化率。最后,通过烃池机理对苯与合成气烷基化反应的失活机理进行合理推测。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024022
摘要:
分别采用沉积沉淀法(DP)和浸渍法(IMP)制备Ni/SiO2催化剂前体,前体经H2还原得到Ni/SiO2-DP和Ni/SiO2-IMP。对所制备的催化剂进行X射线衍射、X射线光电子能谱、N2吸附-脱附、化学吸附、傅里叶变换红外、透射电镜和拉曼光谱表征,并考察其与介质阻挡放电等离子体(DBD)协同催化甲烷干重整(DRM)制合成气反应性能。研究结果表明,相较于Ni/SiO2-IMP,Ni/SiO2-DP因其较小的Ni颗粒尺寸、Ni与载体的强相互作用以及对反应物分子较强的吸附活化能力,具有更高的催化活性和稳定性。对Ni/SiO2-DP制备条件考察结果表明,H2等离子体还原(PR)的Ni/SiO2-DP-PR比程序升温还原(TPR)的Ni/SiO2-DP-TPR具有更高的催化活性。沉积沉淀时间为10 h,H2等离子体还原时间为30 min时,CH4和CO2转化率分别为72.5%和78.2%,H2和CO选择性分别为86.7%和94.2%,能量利用率为4.36 mmol/kJ。
分别采用沉积沉淀法(DP)和浸渍法(IMP)制备Ni/SiO2催化剂前体,前体经H2还原得到Ni/SiO2-DP和Ni/SiO2-IMP。对所制备的催化剂进行X射线衍射、X射线光电子能谱、N2吸附-脱附、化学吸附、傅里叶变换红外、透射电镜和拉曼光谱表征,并考察其与介质阻挡放电等离子体(DBD)协同催化甲烷干重整(DRM)制合成气反应性能。研究结果表明,相较于Ni/SiO2-IMP,Ni/SiO2-DP因其较小的Ni颗粒尺寸、Ni与载体的强相互作用以及对反应物分子较强的吸附活化能力,具有更高的催化活性和稳定性。对Ni/SiO2-DP制备条件考察结果表明,H2等离子体还原(PR)的Ni/SiO2-DP-PR比程序升温还原(TPR)的Ni/SiO2-DP-TPR具有更高的催化活性。沉积沉淀时间为10 h,H2等离子体还原时间为30 min时,CH4和CO2转化率分别为72.5%和78.2%,H2和CO选择性分别为86.7%和94.2%,能量利用率为4.36 mmol/kJ。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60463-9
摘要:
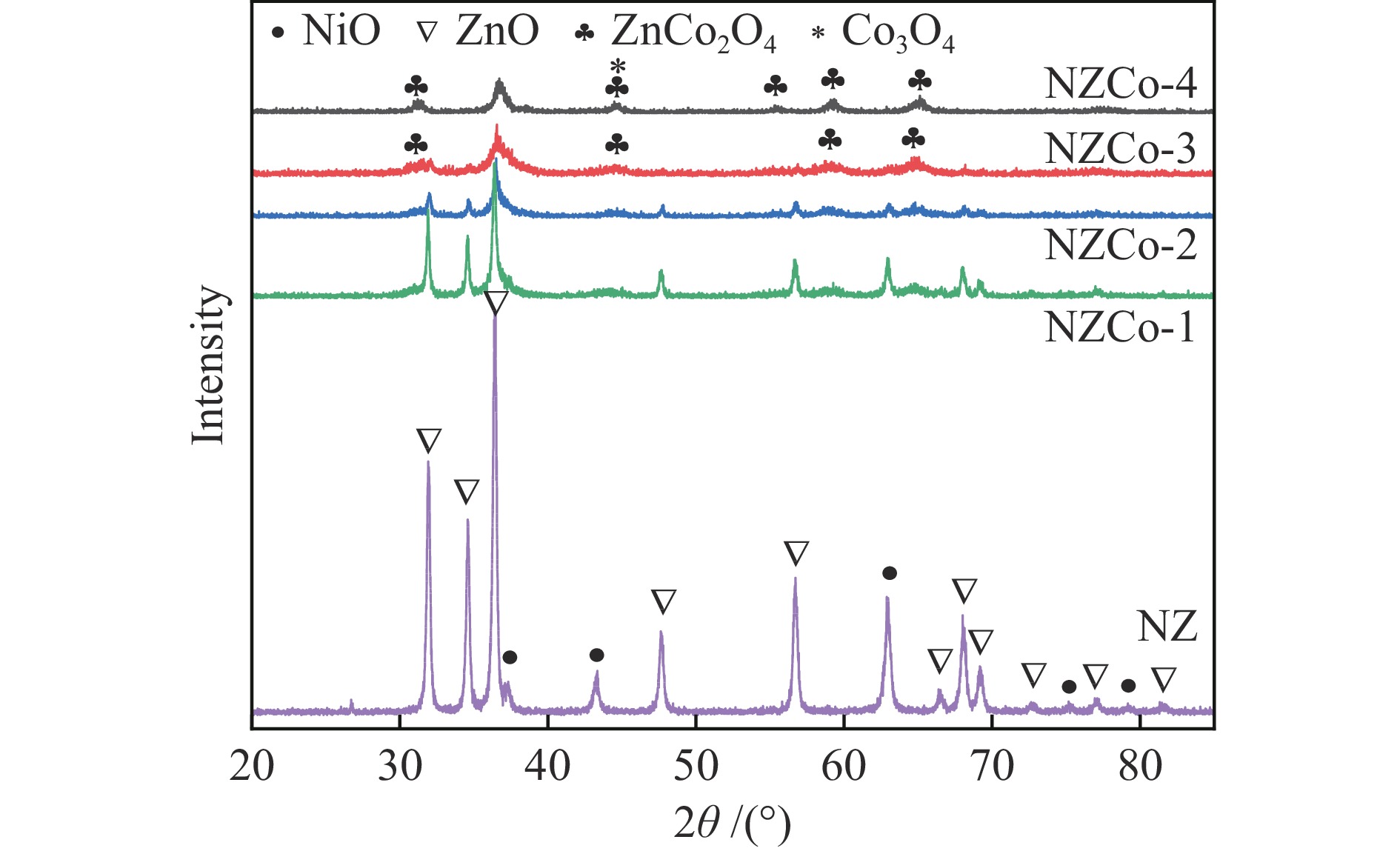
采用共沉淀法在ZnO中引入金属Co并使其形成复合金属氧化物,通过共沉淀-浸渍法构筑了不同Co含量的复合金属氧化物脱硫剂,考察其脱硫活性和再生性能。采用XRD、TEM、N2低温吸附-脱附、XPS和H2-TPR等对脱硫剂的结构和性质进行系统表征,证实得到了Ni/ZnCo2O4@ZnO结构的复合金属氧化物脱硫剂。复合金属氧化物脱硫剂中ZnCo2O4的形成有利于脱硫剂的颗粒尺寸减小、分散度提升、比表面积增加。反应后XRD显示,ZnCo2O4也可作为H2S的吸附剂,从而提高了脱硫剂的硫吸附容量。所有的复合金属氧化物脱硫剂的脱硫性能显著优于Ni/ZnO,其中,Zn∶Co物质的量比为1∶1的脱硫剂NZCo-3具有最优的脱硫性能,该脱硫剂在反应温度300 ℃,氢压3 MPa,质量空速2.2 h−1,氢油体积比300的条件下脱硫率为100%,且经过六次循环后仍能够保持优异的脱硫性能。该研究结果为合理设计Ni/ZnO脱硫剂以提高其脱硫性能和再生性能提供新的思路。
采用共沉淀法在ZnO中引入金属Co并使其形成复合金属氧化物,通过共沉淀-浸渍法构筑了不同Co含量的复合金属氧化物脱硫剂,考察其脱硫活性和再生性能。采用XRD、TEM、N2低温吸附-脱附、XPS和H2-TPR等对脱硫剂的结构和性质进行系统表征,证实得到了Ni/ZnCo2O4@ZnO结构的复合金属氧化物脱硫剂。复合金属氧化物脱硫剂中ZnCo2O4的形成有利于脱硫剂的颗粒尺寸减小、分散度提升、比表面积增加。反应后XRD显示,ZnCo2O4也可作为H2S的吸附剂,从而提高了脱硫剂的硫吸附容量。所有的复合金属氧化物脱硫剂的脱硫性能显著优于Ni/ZnO,其中,Zn∶Co物质的量比为1∶1的脱硫剂NZCo-3具有最优的脱硫性能,该脱硫剂在反应温度300 ℃,氢压3 MPa,质量空速2.2 h−1,氢油体积比300的条件下脱硫率为100%,且经过六次循环后仍能够保持优异的脱硫性能。该研究结果为合理设计Ni/ZnO脱硫剂以提高其脱硫性能和再生性能提供新的思路。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60462-7
摘要:
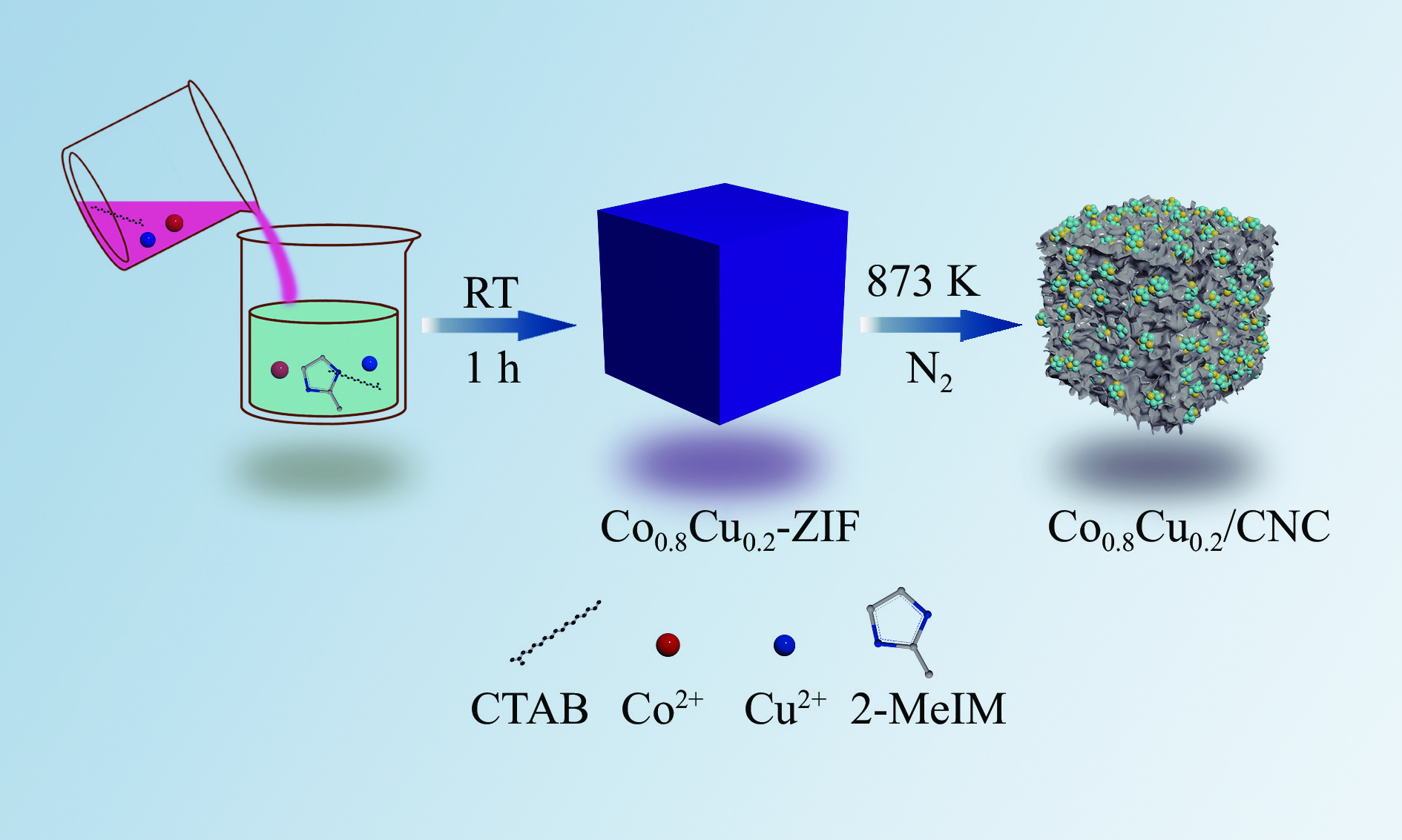
氨硼烷(NH3BH3,AB)被认为是一种便携式制氢的理想储氢材料。本工作在常温下搅拌反应物得到Co0.8Cu0.2-ZIF前驱体,并高温焙烧此前驱体制备了一种双金属碳立方体(Co0.8Cu0.2/CNC)催化剂。此外,采用多种表征方法对催化剂的微观结构以及组成成分进行了研究。通过单一变量法探究了催化剂的变化规律。结果表明,少量Cu的加入会对Co0.8Cu0.2/CNC催化剂的立方体形貌有一定的稳固作用。当分散剂为CTAB且焙烧温度为873 K时,其催化AB水解制氢的活化能(Ea)为50.79 kJ/mol,转化频率(TOF)值高达23.37 min−1。此外,该催化剂经过25次循环后仍然可以催化AB完全水解制氢,表明该催化剂的稳定性能良好。
氨硼烷(NH3BH3,AB)被认为是一种便携式制氢的理想储氢材料。本工作在常温下搅拌反应物得到Co0.8Cu0.2-ZIF前驱体,并高温焙烧此前驱体制备了一种双金属碳立方体(Co0.8Cu0.2/CNC)催化剂。此外,采用多种表征方法对催化剂的微观结构以及组成成分进行了研究。通过单一变量法探究了催化剂的变化规律。结果表明,少量Cu的加入会对Co0.8Cu0.2/CNC催化剂的立方体形貌有一定的稳固作用。当分散剂为CTAB且焙烧温度为873 K时,其催化AB水解制氢的活化能(Ea)为50.79 kJ/mol,转化频率(TOF)值高达23.37 min−1。此外,该催化剂经过25次循环后仍然可以催化AB完全水解制氢,表明该催化剂的稳定性能良好。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024024
摘要:
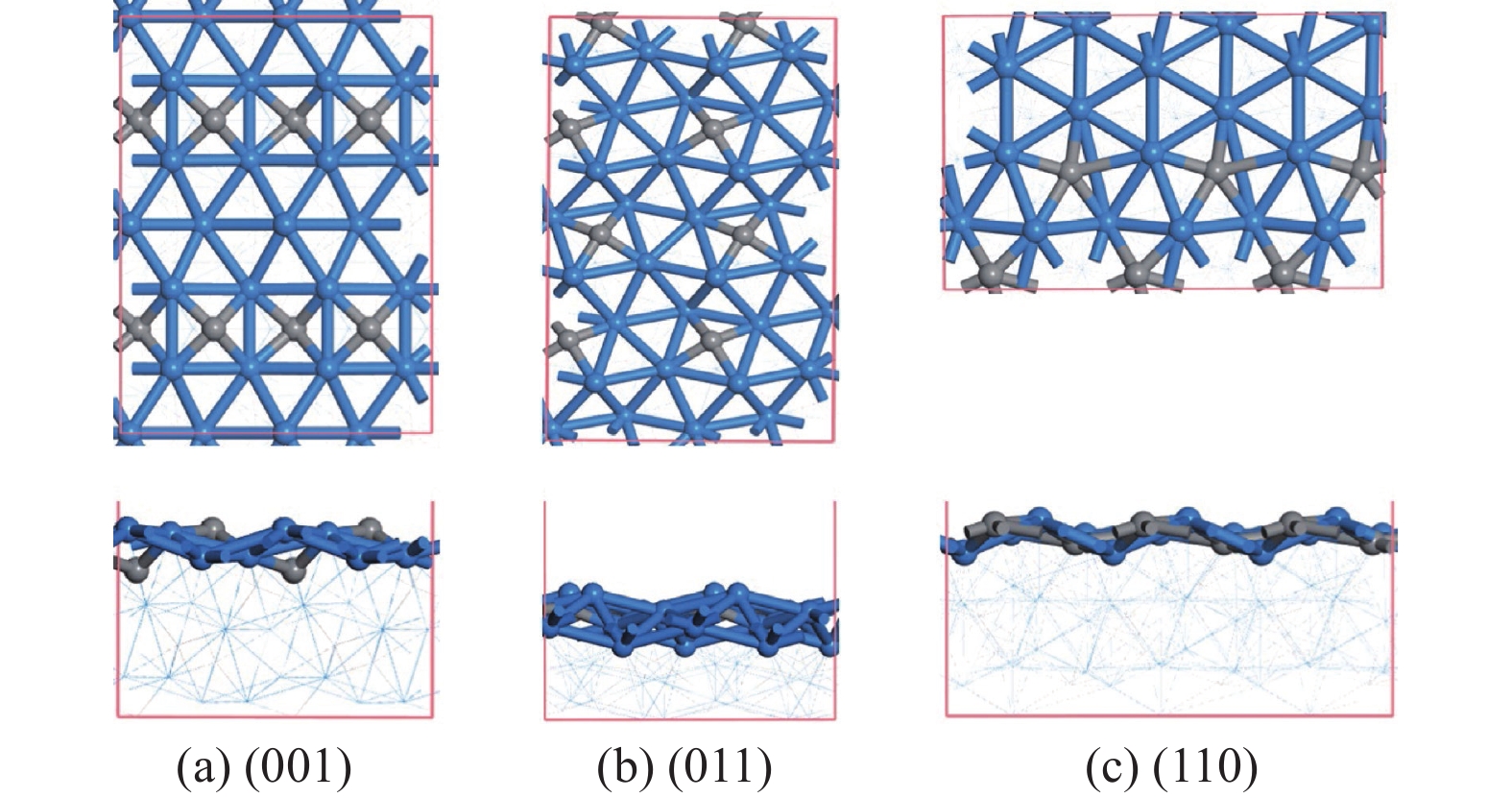
碳化铁θ-Fe3C作为费托反应活性相之一,其氧化造成了催化剂的严重失活。探究氧原子在碳化铁晶面的吸附及移除机理有助于理解氧化过程,为提高催化剂的稳定性提供参考。本工作通过理论计算研究了低覆盖度下氧原子在θ-Fe3C不同晶面的吸附,其在(110)晶面吸附最强,(001)晶面次之,(011)晶面吸附最弱,即(110)容易氧化。原子热力学研究表明,增大H2O分压或降低温度有利于氧原子吸附,容易造成表面氧化。此外,在典型费托反应条件下(110)晶面氧原子的覆盖度最高,进一步证明该晶面易氧化,与低覆盖度吸附结果一致。对移除路径进行计算得出,(011)晶面吸附氧原子直接与CO反应以CO2方式移除能垒较低(0.84 eV);(001)与(110)晶面吸附氧原子主要通过OH歧化以H2O方式移除,但后者形成O-H键需要克服的能垒更高(1.72 vs 1.47 eV)。
碳化铁θ-Fe3C作为费托反应活性相之一,其氧化造成了催化剂的严重失活。探究氧原子在碳化铁晶面的吸附及移除机理有助于理解氧化过程,为提高催化剂的稳定性提供参考。本工作通过理论计算研究了低覆盖度下氧原子在θ-Fe3C不同晶面的吸附,其在(110)晶面吸附最强,(001)晶面次之,(011)晶面吸附最弱,即(110)容易氧化。原子热力学研究表明,增大H2O分压或降低温度有利于氧原子吸附,容易造成表面氧化。此外,在典型费托反应条件下(110)晶面氧原子的覆盖度最高,进一步证明该晶面易氧化,与低覆盖度吸附结果一致。对移除路径进行计算得出,(011)晶面吸附氧原子直接与CO反应以CO2方式移除能垒较低(0.84 eV);(001)与(110)晶面吸附氧原子主要通过OH歧化以H2O方式移除,但后者形成O-H键需要克服的能垒更高(1.72 vs 1.47 eV)。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024028
摘要:
餐厨垃圾是城市垃圾的主要组成部分,同时具有危害性与资源性双重属性。工业化餐厨垃圾处理方法主要以填埋、焚烧发电和厌氧发酵为主,高值化利用方法和技术仍然欠缺,严重制约了餐厨垃圾的资源化发展。本工作首先介绍了淀粉类餐厨垃圾的特性、现有处理技术及预处理方法,并对当前技术手段的优劣进行阐述。最后,介绍了淀粉类餐厨垃圾制备葡萄糖、5-羟甲基糠醛、乙酰丙酸(酯)、γ-戊内酯和乳酸(酯)等高附加值化学品的方法和反应机理,归纳并总结了化学法催化淀粉类餐厨垃圾转化制备高附加值化学品依旧存在的问题与不足。尽管如此,化学转化法是实现淀粉类餐厨垃圾绿色、资源化利用的有效手段之一。
餐厨垃圾是城市垃圾的主要组成部分,同时具有危害性与资源性双重属性。工业化餐厨垃圾处理方法主要以填埋、焚烧发电和厌氧发酵为主,高值化利用方法和技术仍然欠缺,严重制约了餐厨垃圾的资源化发展。本工作首先介绍了淀粉类餐厨垃圾的特性、现有处理技术及预处理方法,并对当前技术手段的优劣进行阐述。最后,介绍了淀粉类餐厨垃圾制备葡萄糖、5-羟甲基糠醛、乙酰丙酸(酯)、γ-戊内酯和乳酸(酯)等高附加值化学品的方法和反应机理,归纳并总结了化学法催化淀粉类餐厨垃圾转化制备高附加值化学品依旧存在的问题与不足。尽管如此,化学转化法是实现淀粉类餐厨垃圾绿色、资源化利用的有效手段之一。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60461-5
摘要:
Gasifition of furfural residue with coal can realize its efficient and clean utilization. But the high alkali metal content in furfural slag is easy to cause the corrosion of gasifier refractory. Two gasification coals with different silica alumina ratio and a furfural residue were selected in the study. The effects of furfural residue additions on corrosion of silica brick, corundum brick, high alumina brick and mullite brick were investigated by using XRD, SEM-EDS and Factsage Software, and the corrosion mechanism was analyzed. With increasing furfural residue addition, the permeability of the slags to high-aluminium-bearing refractories first decreases and then increases, while the permeability on silica brick shows a slight decrease trend. Leucite (KAlSi2O6) with high-melting temperature is generated from the reaction of K2O and SiO2 in slag with Al2O3 in refractories after furfural residue is added, which hinders the infiltration of slag in refractories. Kaliophilite(KAlSiO4) of low-melting point is formed when K2O content increases, and this contributes to the infiltration of slag in refractories. The acid-base reaction between slag and silica brick is distinctly occurred, more slag reacts with SiO2 in the silicon brick, resulting in a decrease in the amount of slag infiltrating into the silicon brick as furfural residue is added. The corrosion of silica brick is mainly caused by the acid-base reaction, while the corrosion of three alumina based refractory bricks of corundum, mullite and high alumina brick is determined by slag infiltration. A linear correlation between the percolation rate and slag viscosity is established, the slag permeability increase with decreasing viscosity, resutling in stronger permeability for the high Si/Al slag with lower viscosity.
Gasifition of furfural residue with coal can realize its efficient and clean utilization. But the high alkali metal content in furfural slag is easy to cause the corrosion of gasifier refractory. Two gasification coals with different silica alumina ratio and a furfural residue were selected in the study. The effects of furfural residue additions on corrosion of silica brick, corundum brick, high alumina brick and mullite brick were investigated by using XRD, SEM-EDS and Factsage Software, and the corrosion mechanism was analyzed. With increasing furfural residue addition, the permeability of the slags to high-aluminium-bearing refractories first decreases and then increases, while the permeability on silica brick shows a slight decrease trend. Leucite (KAlSi2O6) with high-melting temperature is generated from the reaction of K2O and SiO2 in slag with Al2O3 in refractories after furfural residue is added, which hinders the infiltration of slag in refractories. Kaliophilite(KAlSiO4) of low-melting point is formed when K2O content increases, and this contributes to the infiltration of slag in refractories. The acid-base reaction between slag and silica brick is distinctly occurred, more slag reacts with SiO2 in the silicon brick, resulting in a decrease in the amount of slag infiltrating into the silicon brick as furfural residue is added. The corrosion of silica brick is mainly caused by the acid-base reaction, while the corrosion of three alumina based refractory bricks of corundum, mullite and high alumina brick is determined by slag infiltration. A linear correlation between the percolation rate and slag viscosity is established, the slag permeability increase with decreasing viscosity, resutling in stronger permeability for the high Si/Al slag with lower viscosity.
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60449-4
摘要:
由CO和CO2加氢制甲烷是目前氢能储存及燃料和化学品可持续生产的有效途径之一,但目前对该反应过程(尤其是针对CO和CO2混合物加氢)的一些细节尚不明晰。为此,作为前期有关CO和CO2加氢制烯烃和醇研究工作的补充,本工作对CO和CO2,尤其是两者混合物的加氢制甲烷反应过程进行了热力学分析。结果证实,与单独CO或CO2相比,二者混合物加氢制甲烷更为合适,总碳基甲烷收率可作为评估甲烷合成反应过程效率的重要指标。CO加氢的甲烷平衡收率比CO2加氢的高,而CO和CO2混合物加氢的总碳基甲烷平衡收率位于两者之间;对于CO和CO2混合物加氢,尽管CO和CO2的平衡转化率随进料组成不同会有很大的变化,但其总碳基甲烷平衡收率随着原料中CO2/(CO+CO2)物质的量比的增大而线形降低。整体上看,在温度低于400 ℃和压力高于0.1 MPa时,无论是CO、CO2、还是两者混合物的化学计量比加氢,其总碳基甲烷平衡收率均高于85%。这些结果无疑对高效CO和CO2加氢制甲烷催化剂研制及反应过程的设计和操作优化有重要的参考价值。
由CO和CO2加氢制甲烷是目前氢能储存及燃料和化学品可持续生产的有效途径之一,但目前对该反应过程(尤其是针对CO和CO2混合物加氢)的一些细节尚不明晰。为此,作为前期有关CO和CO2加氢制烯烃和醇研究工作的补充,本工作对CO和CO2,尤其是两者混合物的加氢制甲烷反应过程进行了热力学分析。结果证实,与单独CO或CO2相比,二者混合物加氢制甲烷更为合适,总碳基甲烷收率可作为评估甲烷合成反应过程效率的重要指标。CO加氢的甲烷平衡收率比CO2加氢的高,而CO和CO2混合物加氢的总碳基甲烷平衡收率位于两者之间;对于CO和CO2混合物加氢,尽管CO和CO2的平衡转化率随进料组成不同会有很大的变化,但其总碳基甲烷平衡收率随着原料中CO2/(CO+CO2)物质的量比的增大而线形降低。整体上看,在温度低于400 ℃和压力高于0.1 MPa时,无论是CO、CO2、还是两者混合物的化学计量比加氢,其总碳基甲烷平衡收率均高于85%。这些结果无疑对高效CO和CO2加氢制甲烷催化剂研制及反应过程的设计和操作优化有重要的参考价值。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024017
摘要:
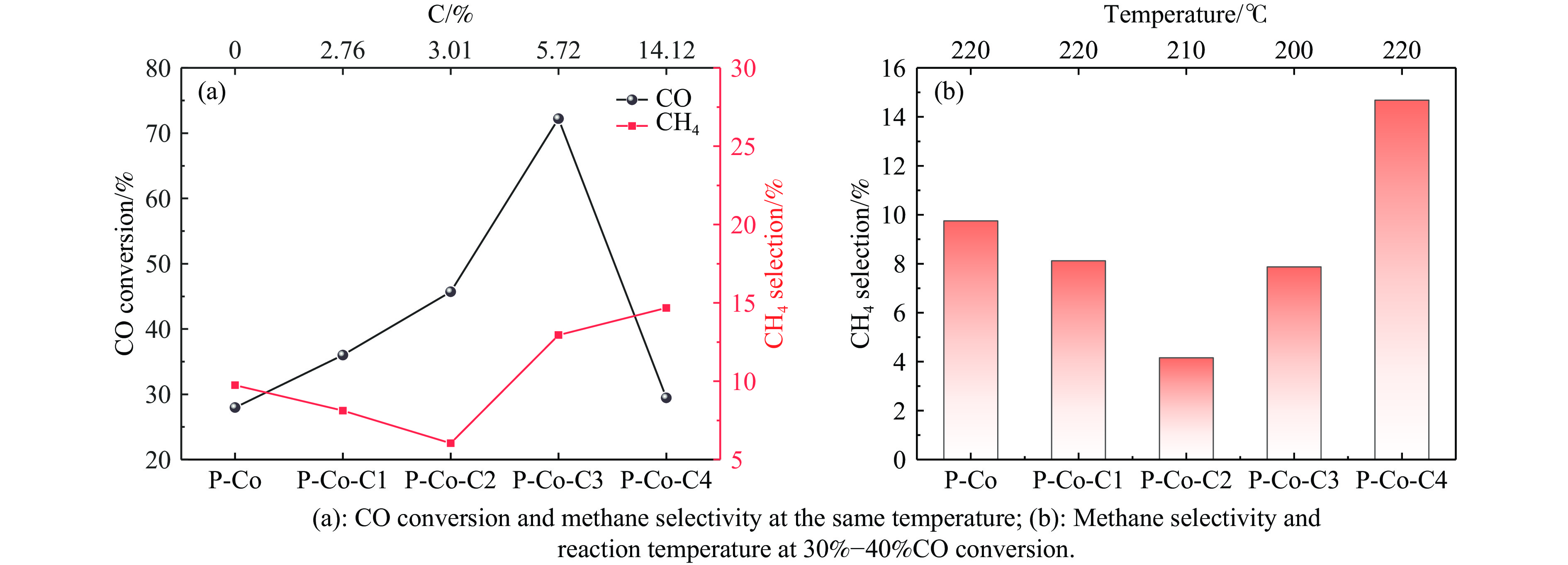
费托合成是将合成气催化转化为长链重质烃的工艺过程。在此过程中,CO活化、歧化反应以及烃类脱氢反应都可以在催化剂表面形成碳物种,而碳物种对费托反应的作用一直存在争议。本工作通过对主要暴露面为HCP-Co(10-11)的单晶钴进行不同条件的预处理,成功构建了具有不同含量原子碳物种的模型催化剂,并采用程序升温加氢、拉曼光谱和红外光谱等表征手段对催化剂中的原子碳物种含量和存在形式进行分析。结果表明,在引入原子碳物种后,钴基催化剂的活性和CH4选择性与原子碳物种数量和存在形式密切相关。含碳量为5.72%的P-Co-C3催化剂具有较高的CO转化率,可达72.2%;而含碳量为3.01%的P-Co-C2催化剂具有较低的CH4选择性,仅为4.2%。此外,表征结果进一步证明该原子碳物种是以C(无定形碳)和CxHy两种形式共存,其在反应过程中可能参与了FT反应,进而提升其反应性能。
费托合成是将合成气催化转化为长链重质烃的工艺过程。在此过程中,CO活化、歧化反应以及烃类脱氢反应都可以在催化剂表面形成碳物种,而碳物种对费托反应的作用一直存在争议。本工作通过对主要暴露面为HCP-Co(10-11)的单晶钴进行不同条件的预处理,成功构建了具有不同含量原子碳物种的模型催化剂,并采用程序升温加氢、拉曼光谱和红外光谱等表征手段对催化剂中的原子碳物种含量和存在形式进行分析。结果表明,在引入原子碳物种后,钴基催化剂的活性和CH4选择性与原子碳物种数量和存在形式密切相关。含碳量为5.72%的P-Co-C3催化剂具有较高的CO转化率,可达72.2%;而含碳量为3.01%的P-Co-C2催化剂具有较低的CH4选择性,仅为4.2%。此外,表征结果进一步证明该原子碳物种是以C(无定形碳)和CxHy两种形式共存,其在反应过程中可能参与了FT反应,进而提升其反应性能。
当前状态:
, 最新更新时间: ,
doi: 10.19906/j.cnki.JFCT.2024020
摘要:
本研究采用均匀沉淀-浸渍和浸渍-浸渍两种分步法分别制备了Ni-Ag/SiO2-p和Ni-Ag/SiO2-i两种Ni-Ag双金属催化剂,用于催化草酸二甲酯(DMO)选择性加氢制乙醇酸甲酯(MG)。利用X射线衍射、红外光谱、透射电镜、N2物理吸附、程序升温脱附实验和X射线光电子能谱表征技术对两种催化剂进行了系统的表征和结构解析,发现催化剂的制备方法对双金属催化剂的结构和性能有着重要影响,Ni-Ag/SiO2-p催化剂中由于层状硅酸镍结构的存在,Ag、Ni活性物种的金属分散度相比于Ni-Ag/SiO2-i催化剂更高,这促进了反应物H2和DMO的吸附与活化;催化剂性能评价结果表明,均匀沉淀-浸渍法制备的Ag负载量仅为0.48%的Ni-Ag/SiO2-p催化剂显著提升了DMO加氢活性,在220 ℃、2.0 MPa、液时空速0.5 h-1、氢酯比50的条件下,DMO转化率和MG选择性达到了99.1%和87.6%。研究结果可为设计和优化DMO加氢制MG催化剂提供一定借鉴。
本研究采用均匀沉淀-浸渍和浸渍-浸渍两种分步法分别制备了Ni-Ag/SiO2-p和Ni-Ag/SiO2-i两种Ni-Ag双金属催化剂,用于催化草酸二甲酯(DMO)选择性加氢制乙醇酸甲酯(MG)。利用X射线衍射、红外光谱、透射电镜、N2物理吸附、程序升温脱附实验和X射线光电子能谱表征技术对两种催化剂进行了系统的表征和结构解析,发现催化剂的制备方法对双金属催化剂的结构和性能有着重要影响,Ni-Ag/SiO2-p催化剂中由于层状硅酸镍结构的存在,Ag、Ni活性物种的金属分散度相比于Ni-Ag/SiO2-i催化剂更高,这促进了反应物H2和DMO的吸附与活化;催化剂性能评价结果表明,均匀沉淀-浸渍法制备的Ag负载量仅为0.48%的Ni-Ag/SiO2-p催化剂显著提升了DMO加氢活性,在220 ℃、2.0 MPa、液时空速0.5 h-1、氢酯比50的条件下,DMO转化率和MG选择性达到了99.1%和87.6%。研究结果可为设计和优化DMO加氢制MG催化剂提供一定借鉴。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60451-2
摘要:
本研究通过对比CO、H2、N2三种气氛下的液化行为,探究了CO对神华上湾煤液化过程的影响,并进一步研究不同CO/H2比以及催化剂对合成气条件下液化过程的影响。结果显示,在CO气氛下煤直接液化的油产率达到43.1%,比H2气氛中低4.2%,但比N2气氛下高10.2%,添加神华863催化剂后液化效果得到进一步的提升,表明,CO在液化过程中可通过水煤气变换反应和CO与煤有机结构间的反应促进煤液化。对液化产物进行GC-MS、FI-TR等分析发现,CO使液化油中苯系物、脂肪烃与含氧化合物同时增多,对液化残渣中官能团与自由基浓度的影响不明显。在CO+H2合成气下的实验结果表明,在20%CO的合成气中煤液化具有最高的油产率,达到57.4%;适当提高煤的含水量能够提升液化效果;神华863催化剂对液化过程与水煤气变换反应均具有良好的催化作用。研究工作为煤在合成气下的直接液化提供理论基础。
本研究通过对比CO、H2、N2三种气氛下的液化行为,探究了CO对神华上湾煤液化过程的影响,并进一步研究不同CO/H2比以及催化剂对合成气条件下液化过程的影响。结果显示,在CO气氛下煤直接液化的油产率达到43.1%,比H2气氛中低4.2%,但比N2气氛下高10.2%,添加神华863催化剂后液化效果得到进一步的提升,表明,CO在液化过程中可通过水煤气变换反应和CO与煤有机结构间的反应促进煤液化。对液化产物进行GC-MS、FI-TR等分析发现,CO使液化油中苯系物、脂肪烃与含氧化合物同时增多,对液化残渣中官能团与自由基浓度的影响不明显。在CO+H2合成气下的实验结果表明,在20%CO的合成气中煤液化具有最高的油产率,达到57.4%;适当提高煤的含水量能够提升液化效果;神华863催化剂对液化过程与水煤气变换反应均具有良好的催化作用。研究工作为煤在合成气下的直接液化提供理论基础。
当前状态:
, 最新更新时间: ,
doi: 10.1016/S1872-5813(24)60452-4
摘要:
Ni/TiO2 catalyst is widely employed for photo-driven DRM reaction while the influence of crystal structure of TiO2 remains unclear. In this work, the rutile/anatase ratio in supports was successfully controlled by varying the calcination temperature of anatase-TiO2. Structural characterizations revealed that a distinct TiOx coating on the Ni nanoparticles (NPs) was evident for Ni/TiO2-700 catalyst due to strong metal-support interaction. It is observed that the TiOx overlayer gradually disappeared as the ratio of rutile/anatase increased, thereby enhancing the exposure of Ni active sites. The exposed Ni sites enhanced visible light absorption and boosted the dissociation capability of CH4, which led to the much elevated catalytic activity for Ni/ TiO2-950 in which rutile dominated. Therefore, the catalytic activity of solar-driven DRM reaction was significantly influenced by the rutile/anatase ratio. Ni/TiO2-950, characterized by a predominant rutile phase, exhibited the highest DRM reactivity, with remarkable H2 and CO production rates reaching as high as 87.4 and 220.2 mmol/(g·h), respectively. These rates were approximately 257 and 130 times higher, respectively, compared to those obtained on Ni/TiO2-700 with anatase. This study suggests that the optimization of crystal structure of TiO2 support can effectively enhance the performance of photothermal DRM reaction.
Ni/TiO2 catalyst is widely employed for photo-driven DRM reaction while the influence of crystal structure of TiO2 remains unclear. In this work, the rutile/anatase ratio in supports was successfully controlled by varying the calcination temperature of anatase-TiO2. Structural characterizations revealed that a distinct TiOx coating on the Ni nanoparticles (NPs) was evident for Ni/TiO2-700 catalyst due to strong metal-support interaction. It is observed that the TiOx overlayer gradually disappeared as the ratio of rutile/anatase increased, thereby enhancing the exposure of Ni active sites. The exposed Ni sites enhanced visible light absorption and boosted the dissociation capability of CH4, which led to the much elevated catalytic activity for Ni/ TiO2-950 in which rutile dominated. Therefore, the catalytic activity of solar-driven DRM reaction was significantly influenced by the rutile/anatase ratio. Ni/TiO2-950, characterized by a predominant rutile phase, exhibited the highest DRM reactivity, with remarkable H2 and CO production rates reaching as high as 87.4 and 220.2 mmol/(g·h), respectively. These rates were approximately 257 and 130 times higher, respectively, compared to those obtained on Ni/TiO2-700 with anatase. This study suggests that the optimization of crystal structure of TiO2 support can effectively enhance the performance of photothermal DRM reaction.
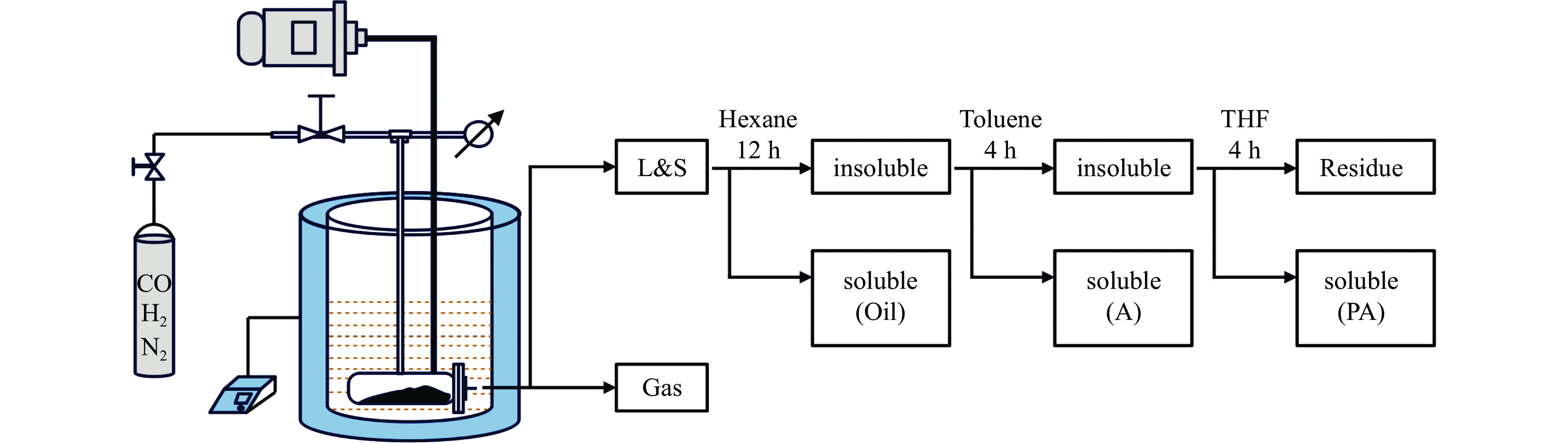
2024, 52(8): 1035-1044.
doi: 10.19906/j.cnki.JFCT.2024007
摘要:
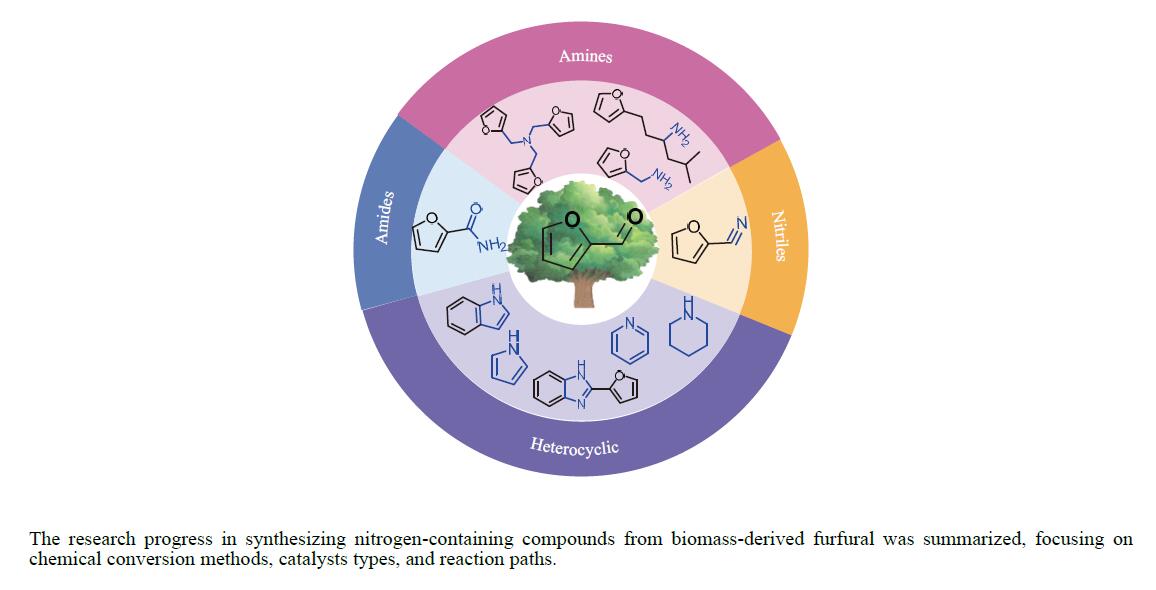
糠醛作为最有潜力的生物质基平台化合物之一,可通过化学催化转化为一系列高附加值的化学品和燃料。其中,含氮化合物具有广泛的生物活性,常用于合成药物分子和生物塑料等功能性材料。糠醛通过还原胺化、氨氧化、氧化偶联等过程,可以合成不同类型的含氮化合物,具有巨大的研究前景和应用潜力。本论文综述了近年来以糠醛为原料合成各种高值含氮化合物的研究进展,包括胺类化合物(伯胺、仲胺和叔胺)、腈类化合物、酰胺类化合物和杂环类化合物(苯并杂环类、噻唑类、吡咯、吲哚、哌啶和吡啶等)。重点关注合成方法、催化剂类型、反应路径和反应机理,同时分析了催化剂和氮源对产物分布的影响。该综述为今后生物质基糠醛转化为含氮化合物的研究提供了一些基础信息,为发展更多高效的糠醛催化转化体系提供依据和系统性知识。
糠醛作为最有潜力的生物质基平台化合物之一,可通过化学催化转化为一系列高附加值的化学品和燃料。其中,含氮化合物具有广泛的生物活性,常用于合成药物分子和生物塑料等功能性材料。糠醛通过还原胺化、氨氧化、氧化偶联等过程,可以合成不同类型的含氮化合物,具有巨大的研究前景和应用潜力。本论文综述了近年来以糠醛为原料合成各种高值含氮化合物的研究进展,包括胺类化合物(伯胺、仲胺和叔胺)、腈类化合物、酰胺类化合物和杂环类化合物(苯并杂环类、噻唑类、吡咯、吲哚、哌啶和吡啶等)。重点关注合成方法、催化剂类型、反应路径和反应机理,同时分析了催化剂和氮源对产物分布的影响。该综述为今后生物质基糠醛转化为含氮化合物的研究提供了一些基础信息,为发展更多高效的糠醛催化转化体系提供依据和系统性知识。
2024, 52(8): 1045-1056.
doi: 10.1016/S1872-5813(24)60445-7
摘要:
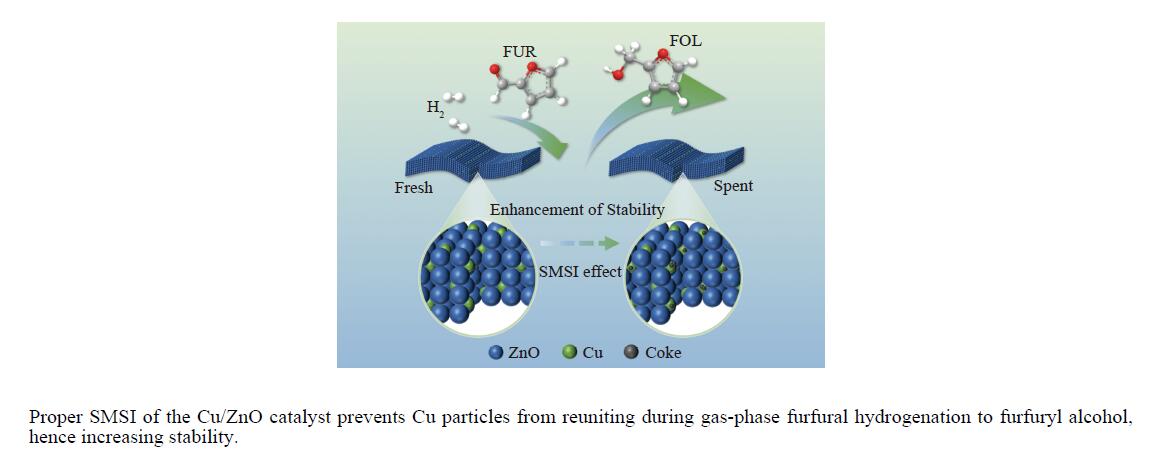
采用共沉淀法制备了系列Cu/Zn比不同的Cu/ZnO催化剂,研究了Cu/Zn比与金属载体强相互作用(SMSI)的关系及其对糠醛气相加氢制糠醇催化反应性能的影响。XRD、H2-TPR、SEM、HRTEM和XPS等表征结果显示,Cu/ZnO催化剂中的金属载体强相互作用改变了催化剂的微观结构。ZnO载体对活性金属Cu颗粒具有不同程度的几何修饰,影响了Cu表面的电子状态。不同Cu/Zn比的Cu/ZnO催化剂的SMSI作用顺序为:20Cu/ZnO > 40Cu/ZnO > 60Cu/ZnO > 80Cu/ZnO。在同一反应条件下,20Cu/ZnO催化剂的糠醛转化率高于80%的时间仅为5 h,而60Cu/ZnO催化剂的糠醛转化率高于80%的时间可以达到28 h,表明适当的SMSI作用有利于提升Cu/ZnO催化剂在糠醛加氢反应中的稳定性,而过强的SMSI作用会抑制其催化活性。
采用共沉淀法制备了系列Cu/Zn比不同的Cu/ZnO催化剂,研究了Cu/Zn比与金属载体强相互作用(SMSI)的关系及其对糠醛气相加氢制糠醇催化反应性能的影响。XRD、H2-TPR、SEM、HRTEM和XPS等表征结果显示,Cu/ZnO催化剂中的金属载体强相互作用改变了催化剂的微观结构。ZnO载体对活性金属Cu颗粒具有不同程度的几何修饰,影响了Cu表面的电子状态。不同Cu/Zn比的Cu/ZnO催化剂的SMSI作用顺序为:20Cu/ZnO > 40Cu/ZnO > 60Cu/ZnO > 80Cu/ZnO。在同一反应条件下,20Cu/ZnO催化剂的糠醛转化率高于80%的时间仅为5 h,而60Cu/ZnO催化剂的糠醛转化率高于80%的时间可以达到28 h,表明适当的SMSI作用有利于提升Cu/ZnO催化剂在糠醛加氢反应中的稳定性,而过强的SMSI作用会抑制其催化活性。
2024, 52(8): 1057-1069.
doi: 10.19906/j.cnki.JFCT.2024014
摘要:
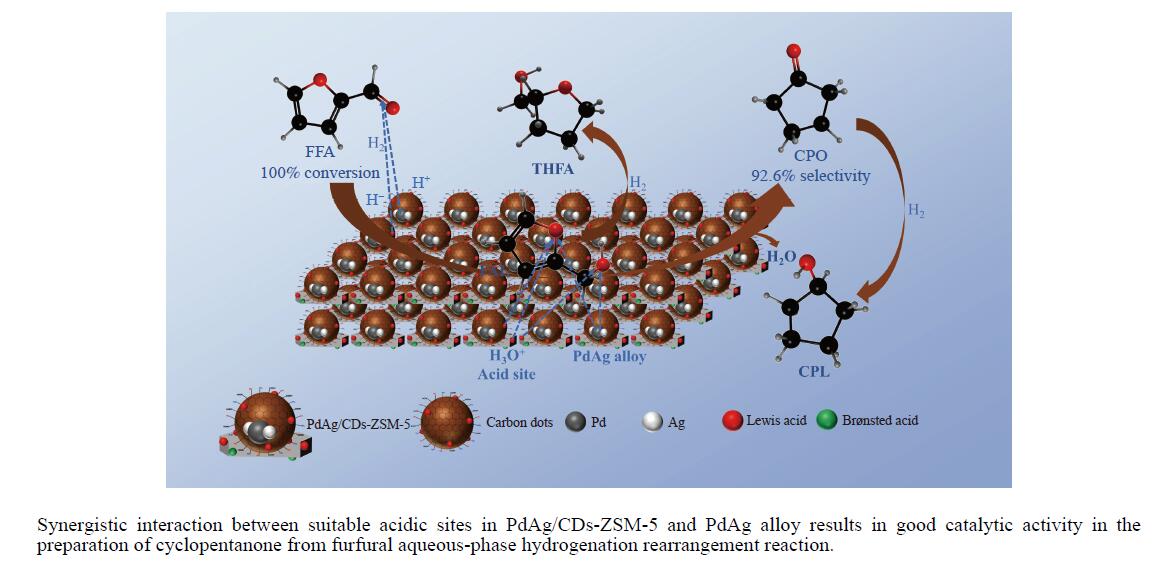
以沸石分子筛(ZSM-5)为载体,碳点(carbon dots, CDs)为还原剂和稳定剂,通过光照还原法制备了双金属PdAg/CDs-ZSM-5催化剂,用于糠醛(furfural, FFA)水相加氢-重排制备环戊酮(cyclopentanone, CPO)反应。采用X射线衍射(XRD)、X射线光电子能谱(XPS)、扫描电子显微镜(SEM)、透射电子显微镜(TEM)、氨气程序升温化学吸附(NH3-TPD)和吡啶红外(Py-FTIR)等手段对催化剂进行了表征。结果表明,CDs具有良好的还原性和丰富的Lewis酸性位点,能够将Pd2+、Ag+还原为金属单质并形成纳米合金结构,复合催化剂中适宜的酸性位点与PdAg合金之间的协同作用使得PdAg/CDs-ZSM-5催化剂在最优反应条件下,对FFA转化率达到100%,目标产物CPO选择性为92.6%。催化剂重复使用五次后仍能保持较高的活性与稳定性。
以沸石分子筛(ZSM-5)为载体,碳点(carbon dots, CDs)为还原剂和稳定剂,通过光照还原法制备了双金属PdAg/CDs-ZSM-5催化剂,用于糠醛(furfural, FFA)水相加氢-重排制备环戊酮(cyclopentanone, CPO)反应。采用X射线衍射(XRD)、X射线光电子能谱(XPS)、扫描电子显微镜(SEM)、透射电子显微镜(TEM)、氨气程序升温化学吸附(NH3-TPD)和吡啶红外(Py-FTIR)等手段对催化剂进行了表征。结果表明,CDs具有良好的还原性和丰富的Lewis酸性位点,能够将Pd2+、Ag+还原为金属单质并形成纳米合金结构,复合催化剂中适宜的酸性位点与PdAg合金之间的协同作用使得PdAg/CDs-ZSM-5催化剂在最优反应条件下,对FFA转化率达到100%,目标产物CPO选择性为92.6%。催化剂重复使用五次后仍能保持较高的活性与稳定性。
2024, 52(8): 1070-1078.
doi: 10.19906/j.cnki.JFCT.2024018
摘要:
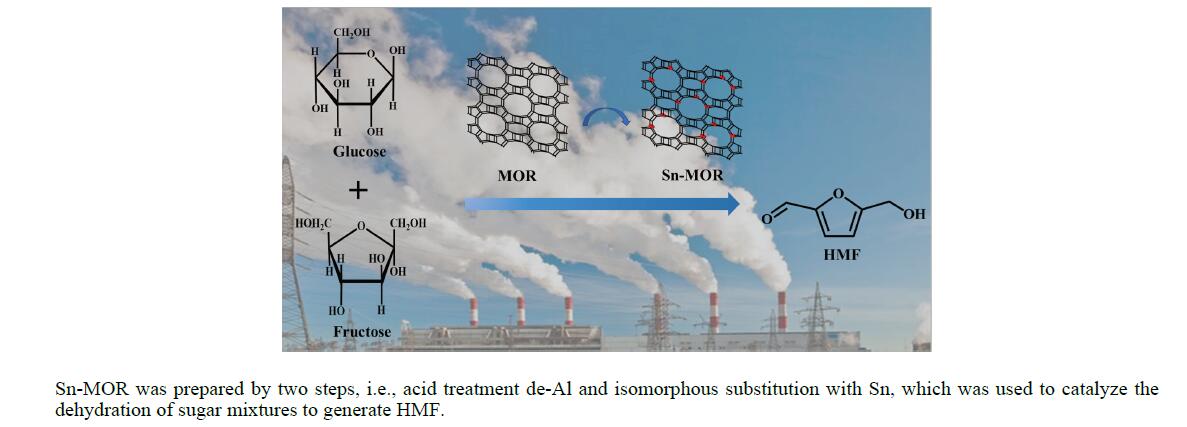
采用酸处理脱铝补位两步法对丝光沸石(MOR)锡改性制备了系列Sn-MOR催化剂,用X射线衍射(XRD)、紫外-可见漫反射光谱(UV-vis)、氨程序升温脱附(NH3-TPD)、X射线荧光光谱(XRF)对其进行表征。结果表明,改性后催化剂仍保持丝光沸石的晶体结构,酸中心强度与酸量有改变,同时在沸石骨架中引入了锡。研究了改性丝光沸石催化单糖(葡萄糖、果糖)及混合糖生成5-羟甲基糠醛(HMF),结果表明,改性后的Sn-MOR仍保持MOR对果糖脱水为HMF的较高催化活性,同时引入的锡物种对葡萄糖具有异构化活性,因此,Sn-MOR可以同时催化葡萄糖和果糖脱水生成HMF。以商品果葡糖浆作为反应底物,在果葡糖浆质量1.94 g,催化剂用量0.3 g,反应温度170 ℃,反应时间3 h的较优反应条件下,以3.76-Sn-MOR1为催化剂,果葡糖浆转化率91.82%,HMF产率63.76%,HMF选择性69.43%;催化剂循环使用五次,仍保持了一定的催化活性,HMF产率仍有49.5%。
采用酸处理脱铝补位两步法对丝光沸石(MOR)锡改性制备了系列Sn-MOR催化剂,用X射线衍射(XRD)、紫外-可见漫反射光谱(UV-vis)、氨程序升温脱附(NH3-TPD)、X射线荧光光谱(XRF)对其进行表征。结果表明,改性后催化剂仍保持丝光沸石的晶体结构,酸中心强度与酸量有改变,同时在沸石骨架中引入了锡。研究了改性丝光沸石催化单糖(葡萄糖、果糖)及混合糖生成5-羟甲基糠醛(HMF),结果表明,改性后的Sn-MOR仍保持MOR对果糖脱水为HMF的较高催化活性,同时引入的锡物种对葡萄糖具有异构化活性,因此,Sn-MOR可以同时催化葡萄糖和果糖脱水生成HMF。以商品果葡糖浆作为反应底物,在果葡糖浆质量1.94 g,催化剂用量0.3 g,反应温度170 ℃,反应时间3 h的较优反应条件下,以3.76-Sn-MOR1为催化剂,果葡糖浆转化率91.82%,HMF产率63.76%,HMF选择性69.43%;催化剂循环使用五次,仍保持了一定的催化活性,HMF产率仍有49.5%。
2024, 52(8): 1079-1087.
doi: 10.1016/S1872-5813(24)60448-2
摘要:
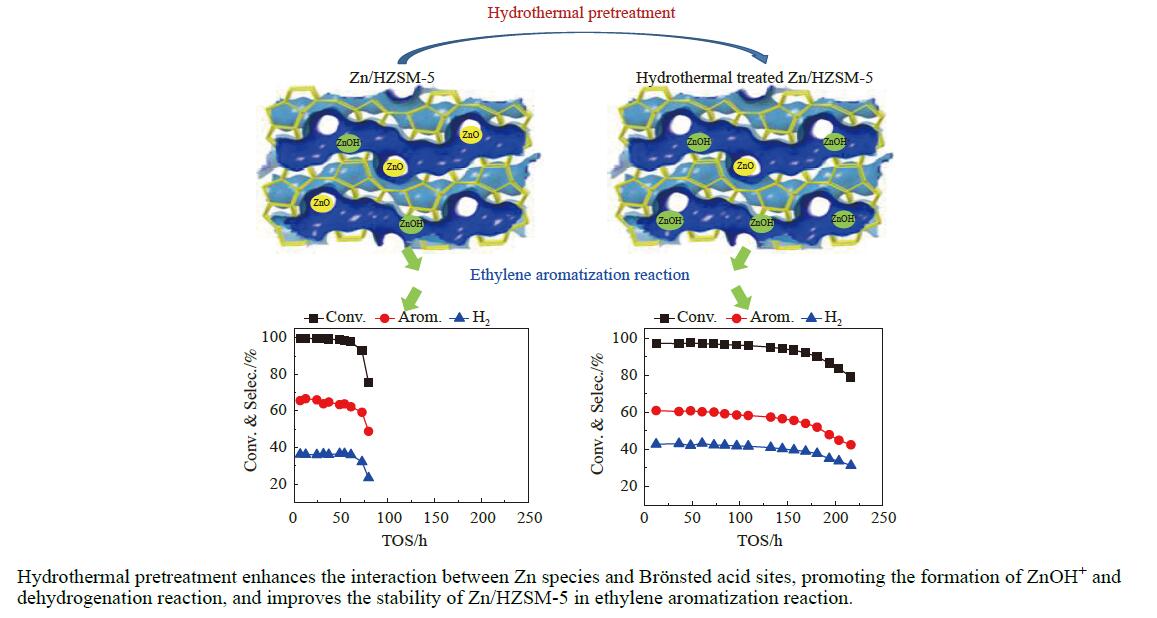
针对用于低碳烯烃芳构化的Zn/HZSM-5催化剂存在易于结焦失活的问题,采用高温水热的方法对催化剂进行预处理,通过XRD、N2物理吸附-脱附、NH3-TPD、Py-FTIR、XPS和TG等技术对样品进行表征,并以乙烯芳构化为探针反应考察了高温水热预处理对催化剂反应性能和稳定性的影响。结果表明,Zn/HZSM-5催化剂经高温水热预处理48 h后表现出优异的催化性能,虽然乙烯转化率略微降低,但是催化剂寿命显著延长,由72 h延长至216 h,同时芳烃选择性保持在60%以上;水热处理促进了ZnO物种与B酸中心的相互作用及ZnOH+物种的生成,在抑制氢转移反应的同时显著促进了催化剂的脱氢性能,提高了氢气选择性;此外,水热处理后催化剂容碳量明显增加、积炭速率降低,表现出优异的抗结焦积炭特性。
针对用于低碳烯烃芳构化的Zn/HZSM-5催化剂存在易于结焦失活的问题,采用高温水热的方法对催化剂进行预处理,通过XRD、N2物理吸附-脱附、NH3-TPD、Py-FTIR、XPS和TG等技术对样品进行表征,并以乙烯芳构化为探针反应考察了高温水热预处理对催化剂反应性能和稳定性的影响。结果表明,Zn/HZSM-5催化剂经高温水热预处理48 h后表现出优异的催化性能,虽然乙烯转化率略微降低,但是催化剂寿命显著延长,由72 h延长至216 h,同时芳烃选择性保持在60%以上;水热处理促进了ZnO物种与B酸中心的相互作用及ZnOH+物种的生成,在抑制氢转移反应的同时显著促进了催化剂的脱氢性能,提高了氢气选择性;此外,水热处理后催化剂容碳量明显增加、积炭速率降低,表现出优异的抗结焦积炭特性。
2024, 52(8): 1088-1094.
doi: 10.1016/S1872-5813(24)60439-1
摘要:
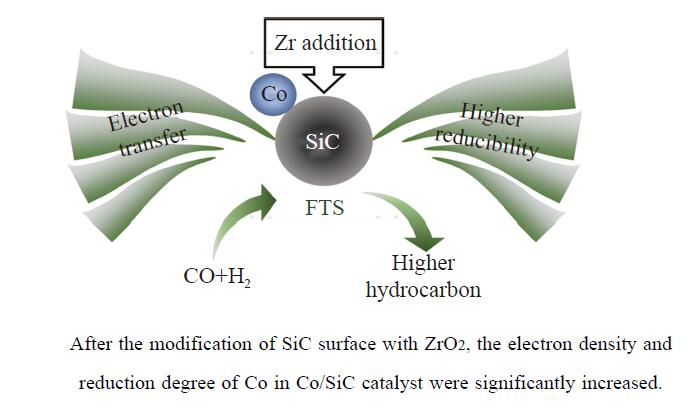
Co/SiC catalysts have exhibited excellent performance in Fischer-Tropsch synthesis reaction. However, few research focuses on investigating the effect of SiC supports surface properties of on catalyst performance. In this study, ZrO2 was utilized to modify the SiC surface, leading to the preparation of a series of Co-ZrO2/SiC catalysts. The physicochemical properties of the catalyst were comprehensively analyzed by using N2 adsorption, XRD, H2-TPR, XPS analyses. Catalytic performance was evaluated using a fixed bed reactor, shedding light on the effect of ZrO2 modified SiC support on cobalt-based Fischer-Tropsch synthesis catalysts. The results indicated that ZrO2 surface modification on SiC resulted in an enhanced reduction degree of Co/SiC catalysts. Additionally, ZrO2 exhibited strong interaction with the amorphous phase on the SiC surface, thereby weakening the interaction between Co and the amorphous phase. This led to an increase in the electron density of cobalt species, consequently improving the selectivity of Co/SiC catalysts towards long-chain hydrocarbons.
Co/SiC catalysts have exhibited excellent performance in Fischer-Tropsch synthesis reaction. However, few research focuses on investigating the effect of SiC supports surface properties of on catalyst performance. In this study, ZrO2 was utilized to modify the SiC surface, leading to the preparation of a series of Co-ZrO2/SiC catalysts. The physicochemical properties of the catalyst were comprehensively analyzed by using N2 adsorption, XRD, H2-TPR, XPS analyses. Catalytic performance was evaluated using a fixed bed reactor, shedding light on the effect of ZrO2 modified SiC support on cobalt-based Fischer-Tropsch synthesis catalysts. The results indicated that ZrO2 surface modification on SiC resulted in an enhanced reduction degree of Co/SiC catalysts. Additionally, ZrO2 exhibited strong interaction with the amorphous phase on the SiC surface, thereby weakening the interaction between Co and the amorphous phase. This led to an increase in the electron density of cobalt species, consequently improving the selectivity of Co/SiC catalysts towards long-chain hydrocarbons.
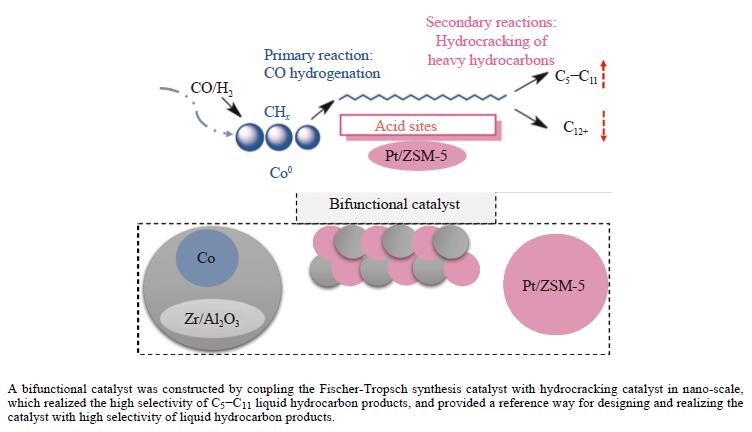
2024, 52(8): 1095-1102.
doi: 10.1016/S1872-5813(24)60438-X
摘要:
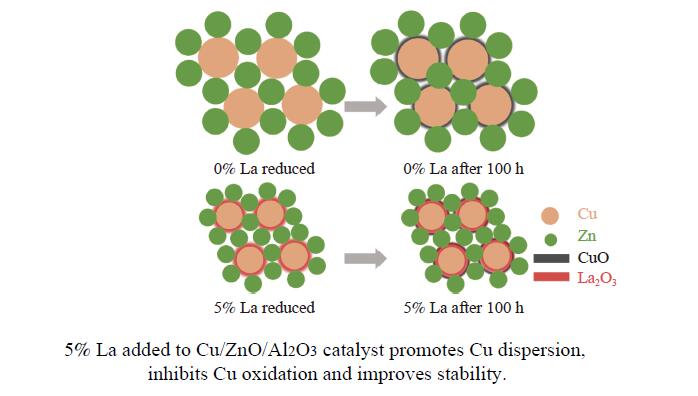
CO2加氢制甲醇反应中Cu/ZnO/Al2O3催化剂的失活是限制其应用的主要原因之一,实验通过向Cu/ZnO/Al2O3催化剂中添加不同含量的La,合成了一系列La改性的Cu/ZnO/Al2O3催化剂,以提高其对 CO2加氢制甲醇反应的催化稳定性。在温度200 ℃,压力3 MPa,空速12000 mL/(g·h)条件下进行的100 h短期稳定性测试,未改性的Cu/ZnO/Al2O3催化剂在100 h内活性衰减明显,添加La后催化剂稳定性逐渐得到提高,当La添加量为5% 时活性最佳(CO2转化率4%,甲醇选择性85%),并且该催化剂在1000 h长期稳定性测试中表现出较高的稳定性(在190−220 h失活 17% 后保持稳定)。通过X射线衍射(XRD)、X射线光电子能谱(XPS)表征发现,加入5% La提高了Cu/ZnO/Al2O3催化剂中Cu、ZnO的分散度,抑制了催化剂中Cu的烧结;同时稳定了Cu0/+,延缓了催化剂中Cu的氧化,从而提高了催化剂的稳定性。
CO2加氢制甲醇反应中Cu/ZnO/Al2O3催化剂的失活是限制其应用的主要原因之一,实验通过向Cu/ZnO/Al2O3催化剂中添加不同含量的La,合成了一系列La改性的Cu/ZnO/Al2O3催化剂,以提高其对 CO2加氢制甲醇反应的催化稳定性。在温度200 ℃,压力3 MPa,空速12000 mL/(g·h)条件下进行的100 h短期稳定性测试,未改性的Cu/ZnO/Al2O3催化剂在100 h内活性衰减明显,添加La后催化剂稳定性逐渐得到提高,当La添加量为5% 时活性最佳(CO2转化率4%,甲醇选择性85%),并且该催化剂在1000 h长期稳定性测试中表现出较高的稳定性(在190−220 h失活 17% 后保持稳定)。通过X射线衍射(XRD)、X射线光电子能谱(XPS)表征发现,加入5% La提高了Cu/ZnO/Al2O3催化剂中Cu、ZnO的分散度,抑制了催化剂中Cu的烧结;同时稳定了Cu0/+,延缓了催化剂中Cu的氧化,从而提高了催化剂的稳定性。
2024, 52(8): 1103-1114.
doi: 10.19906/j.cnki.JFCT.2024003
摘要:
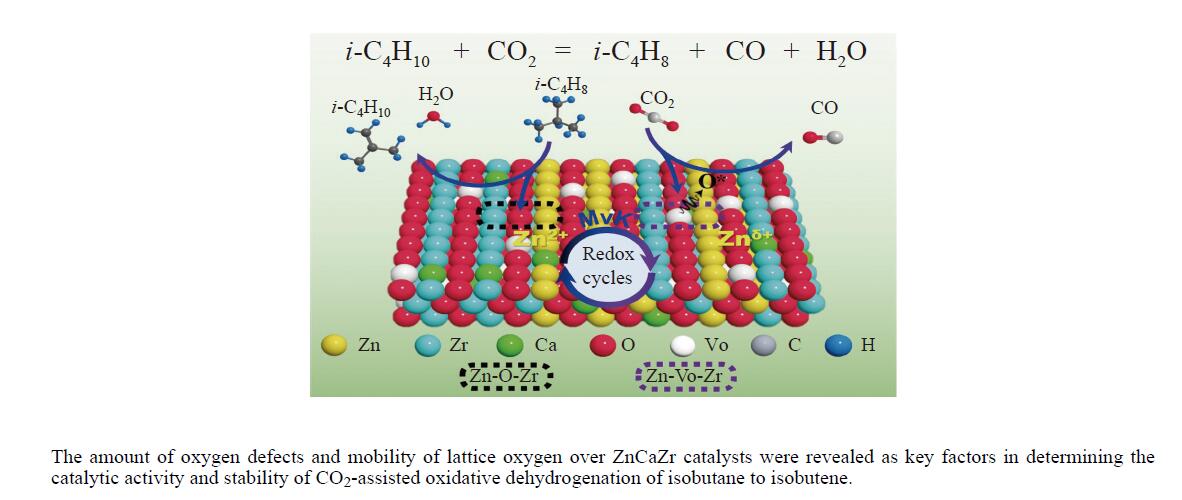
本研究采用一锅式共沉淀法制备了xZn-CaZr固溶体催化剂并将其应用于CO2-BDH反应,通过多种手段探明该系列催化剂的理化性质并结合催化性能阐述其构效关系及表面氧化还原机制。研究表明,xZn-CaZr催化剂在Zn含量为6%−12%的情况下形成了Zn物种高度分散的固溶体结构,且氧缺陷的数量与Zn的含量成正比。在xZn-CaZr催化剂上,晶格氧的数量和氧迁移率是决定催化性能的关键因素,其中,0.4Zn-CaZr催化剂展示出最佳的催化活性,而0.2Zn-CaZr催化剂展示出最佳的反应稳定性。该研究为进一步开发绿色高性能的CO2-BDH催化剂提供了参考价值。
本研究采用一锅式共沉淀法制备了xZn-CaZr固溶体催化剂并将其应用于CO2-BDH反应,通过多种手段探明该系列催化剂的理化性质并结合催化性能阐述其构效关系及表面氧化还原机制。研究表明,xZn-CaZr催化剂在Zn含量为6%−12%的情况下形成了Zn物种高度分散的固溶体结构,且氧缺陷的数量与Zn的含量成正比。在xZn-CaZr催化剂上,晶格氧的数量和氧迁移率是决定催化性能的关键因素,其中,0.4Zn-CaZr催化剂展示出最佳的催化活性,而0.2Zn-CaZr催化剂展示出最佳的反应稳定性。该研究为进一步开发绿色高性能的CO2-BDH催化剂提供了参考价值。
2024, 52(8): 1115-1130.
doi: 10.19906/j.cnki.JFCT.2024012
摘要:
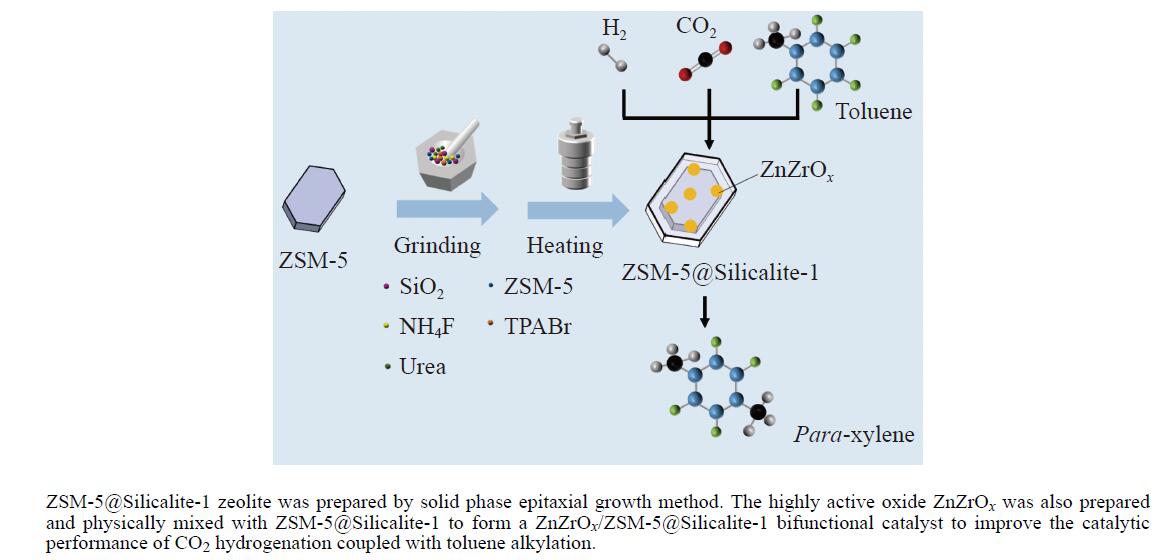
本研究采用固相法在ZSM-5表面外延生长Silicalite-1,制备出ZSM-5@Silicalite-1分子筛。同时制备高活性氧化物ZnZrOx,并与ZSM-5@Silicalite-1物理混合组成ZnZrOx/ ZSM-5@Silicalite-1双功能催化剂,研究了CO2加氢耦合甲苯烷基化催化性能。相比于ZnZrOx/ZSM-5催化剂,分子筛改性后的双功能催化剂提高了对二甲苯(PX)选择性。研究了晶化条件(硅源、晶化过程、晶化次数)对ZSM-5外延生长Silicalite-1的影响,以及Silicalite-1钝化层厚度对CO2加氢耦合甲苯烷基化反应性能的影响。在400 ℃、3 MPa反应条件下,ZZO/1:3.5Z5-Na-SiO2催化剂的甲苯转化率为12.0%,二甲苯选择性为77.4%,在二甲苯中对二甲苯选择性为73.4%。通过SEM、XRD、N2吸附-脱附、XPS、NH3-TPD、Py-FTIR等表征,研究了分子筛的结构和酸性质。结果表明,通过固相外延生长,延长ZSM-5的孔道,增加间二甲苯(MX)、邻二甲苯(OX)的扩散阻力,同时钝化外表面的酸性,可以有效提高对二甲苯(PX)的选择性。固相外延生长法改性ZSM-5分子筛,摒弃了以往堵塞孔以缩小孔口改性分子筛的缺点,在保证催化剂活性的同时提高了产物选择性。
本研究采用固相法在ZSM-5表面外延生长Silicalite-1,制备出ZSM-5@Silicalite-1分子筛。同时制备高活性氧化物ZnZrOx,并与ZSM-5@Silicalite-1物理混合组成ZnZrOx/ ZSM-5@Silicalite-1双功能催化剂,研究了CO2加氢耦合甲苯烷基化催化性能。相比于ZnZrOx/ZSM-5催化剂,分子筛改性后的双功能催化剂提高了对二甲苯(PX)选择性。研究了晶化条件(硅源、晶化过程、晶化次数)对ZSM-5外延生长Silicalite-1的影响,以及Silicalite-1钝化层厚度对CO2加氢耦合甲苯烷基化反应性能的影响。在400 ℃、3 MPa反应条件下,ZZO/1:3.5Z5-Na-SiO2催化剂的甲苯转化率为12.0%,二甲苯选择性为77.4%,在二甲苯中对二甲苯选择性为73.4%。通过SEM、XRD、N2吸附-脱附、XPS、NH3-TPD、Py-FTIR等表征,研究了分子筛的结构和酸性质。结果表明,通过固相外延生长,延长ZSM-5的孔道,增加间二甲苯(MX)、邻二甲苯(OX)的扩散阻力,同时钝化外表面的酸性,可以有效提高对二甲苯(PX)的选择性。固相外延生长法改性ZSM-5分子筛,摒弃了以往堵塞孔以缩小孔口改性分子筛的缺点,在保证催化剂活性的同时提高了产物选择性。
摘要:
利用CaO基吸附剂直接从高温烟气中捕集CO2因成本低、吸附性能好等优点成为CCUS(碳捕集、利用与封存)的重要技术之一。但CaO基吸附剂在碳酸化/煅烧吸脱附循环过程中存在易烧结的问题,导致其吸附性能急剧下降。本研究针对CaO基吸附剂全面总结了其吸附CO2的动力学、热力学及烧结机理,并重点综述了世界各国研究者在CaO基吸附剂抗烧结改性方面所做的研究,指出了各种方法的优点及其局限性。结果表明,水合作用改性可使吸附剂崩塌而获得更大的比表面积;酸溶液改性会在制备过程产生更多的气体和小分子物质提高吸附剂孔隙率;掺杂改性可以促进CaO对CO2的吸附和扩散,还可作为骨架分离CaO颗粒。经比较,掺杂改性工艺简单、性能好,是比较有前景的改性方法,以含钙固废制备抗烧结改性吸附剂是发展方向。
利用CaO基吸附剂直接从高温烟气中捕集CO2因成本低、吸附性能好等优点成为CCUS(碳捕集、利用与封存)的重要技术之一。但CaO基吸附剂在碳酸化/煅烧吸脱附循环过程中存在易烧结的问题,导致其吸附性能急剧下降。本研究针对CaO基吸附剂全面总结了其吸附CO2的动力学、热力学及烧结机理,并重点综述了世界各国研究者在CaO基吸附剂抗烧结改性方面所做的研究,指出了各种方法的优点及其局限性。结果表明,水合作用改性可使吸附剂崩塌而获得更大的比表面积;酸溶液改性会在制备过程产生更多的气体和小分子物质提高吸附剂孔隙率;掺杂改性可以促进CaO对CO2的吸附和扩散,还可作为骨架分离CaO颗粒。经比较,掺杂改性工艺简单、性能好,是比较有前景的改性方法,以含钙固废制备抗烧结改性吸附剂是发展方向。
摘要:
气流床气化过程中产生的细渣含碳量很高,目前多以填埋的方式进行处理,将细渣用于循环流化床锅炉掺烧有望为细渣处理提供有利的技术。本研究选用宁东能源化工基地典型气化工艺GE、OMB及GSP产生的气化细渣为研究对象,利用物理吸附仪、激光拉曼及热重分析仪等仪器,系统研究了气化细渣中残炭的结构特征与燃烧特性。结果表明,原始气化细渣中的物质可分为黏结球形颗粒、多孔不规则颗粒与孤立的大球形颗粒,而酸洗后的气化细渣多以疏松细小的颗粒和多孔不规则块状颗粒存在;细渣中残炭的孔径尺寸主要分布在4−8 nm,且比表面积与残炭的活性位点大小顺序均为:GE > OMB > GSP;GE渣中残炭结构有序度最低,无定形炭结构最多,GSP则相反;GE渣中残炭燃烧速率最快,主要是由于GE渣中残炭有较大的比表面积、较多的无定形炭结构及较高的的活性位点,且GE渣中残炭的综合燃烧指数为5.26 × 10−7%2/(min2·℃3)。
气流床气化过程中产生的细渣含碳量很高,目前多以填埋的方式进行处理,将细渣用于循环流化床锅炉掺烧有望为细渣处理提供有利的技术。本研究选用宁东能源化工基地典型气化工艺GE、OMB及GSP产生的气化细渣为研究对象,利用物理吸附仪、激光拉曼及热重分析仪等仪器,系统研究了气化细渣中残炭的结构特征与燃烧特性。结果表明,原始气化细渣中的物质可分为黏结球形颗粒、多孔不规则颗粒与孤立的大球形颗粒,而酸洗后的气化细渣多以疏松细小的颗粒和多孔不规则块状颗粒存在;细渣中残炭的孔径尺寸主要分布在4−8 nm,且比表面积与残炭的活性位点大小顺序均为:GE > OMB > GSP;GE渣中残炭结构有序度最低,无定形炭结构最多,GSP则相反;GE渣中残炭燃烧速率最快,主要是由于GE渣中残炭有较大的比表面积、较多的无定形炭结构及较高的的活性位点,且GE渣中残炭的综合燃烧指数为5.26 × 10−7%2/(min2·℃3)。
摘要:
Waste gasification has the potential to contribute to China’s transition towards carbon neutrality and zero waste cities via the recirculation of waste as secondary carbon feedstock for the production of chemicals with lower/and or zero carbon footprint, green hydrogen with zero carbon footprint and CO2-neutral synthetic liquid fuels. With China’s significant coal gasification capacity and associated experiences and expertise, Coal-to-X could act as a bridge to Waste-to-X for carbon intensive sectors such as the waste management, chemical production and mobility sectors. To illustrate the opportunities in these areas, this article presented highlights from dynamic global developments in waste gasification, focusing on pioneering industrial developments in Germany between 1980−2000’s as well as current international developments. Lessons learnt from previous and current waste gasification project deployment are shared and enabled the identification of problems which will have to be addressed in the transition from coal gasification towards mono-waste gasification technologies. Additionally, a qualitative evaluation of gasification technologies pointed to the strengths and weaknesses of fixed-bed, fluidized-bed and entrained-flow gasification principles in their application for waste gasification.
Waste gasification has the potential to contribute to China’s transition towards carbon neutrality and zero waste cities via the recirculation of waste as secondary carbon feedstock for the production of chemicals with lower/and or zero carbon footprint, green hydrogen with zero carbon footprint and CO2-neutral synthetic liquid fuels. With China’s significant coal gasification capacity and associated experiences and expertise, Coal-to-X could act as a bridge to Waste-to-X for carbon intensive sectors such as the waste management, chemical production and mobility sectors. To illustrate the opportunities in these areas, this article presented highlights from dynamic global developments in waste gasification, focusing on pioneering industrial developments in Germany between 1980−2000’s as well as current international developments. Lessons learnt from previous and current waste gasification project deployment are shared and enabled the identification of problems which will have to be addressed in the transition from coal gasification towards mono-waste gasification technologies. Additionally, a qualitative evaluation of gasification technologies pointed to the strengths and weaknesses of fixed-bed, fluidized-bed and entrained-flow gasification principles in their application for waste gasification.
摘要:
在NH3选择性催化还原(NH3-SCR)反应中,由于具有宽温度窗口和良好的水热稳定性,金属负载型分子筛是具有广泛应用潜力的脱硝催化剂。本文综述了Cu基和Fe基分子筛催化剂在NH3-SCR领域的研究进展,总结了催化剂的结构特征和NH3-SCR性能指标,并对相应的金属活性位点和反应机理进行了归纳。此外,系统介绍了密度泛函理论(DFT)计算在NH3-SCR反应机理中的应用及反应动力学的研究方法,并对比了不同催化剂体系下的表观动力学参数,为进一步研究金属负载型分子筛催化剂的NH3-SCR反应机理提供方法与思路。
在NH3选择性催化还原(NH3-SCR)反应中,由于具有宽温度窗口和良好的水热稳定性,金属负载型分子筛是具有广泛应用潜力的脱硝催化剂。本文综述了Cu基和Fe基分子筛催化剂在NH3-SCR领域的研究进展,总结了催化剂的结构特征和NH3-SCR性能指标,并对相应的金属活性位点和反应机理进行了归纳。此外,系统介绍了密度泛函理论(DFT)计算在NH3-SCR反应机理中的应用及反应动力学的研究方法,并对比了不同催化剂体系下的表观动力学参数,为进一步研究金属负载型分子筛催化剂的NH3-SCR反应机理提供方法与思路。
摘要:
The chemical and mineralogical characteristics of fly ash from a municipal solid waste incineration (MSWI) in China and the influence of processing parameters on heavy metals removal during leaching were investigated in this work. The fly ash particles had complex surface structure with limited specific surface area. The alkali chloride and metal salts in MSWI fly ash showed evidently impact on leaching efficiency. Metal leachability was related to their properties and speciation in fly ash. Water-soluble salts such as KCl, NaCl and CaCl2 in fly ash were easily washed out. In this study, removal efficiency by water washing was achieved to 93.1% for Cl, 41.4% for Na, 48.5% for K and 24.8% for Ca, respectively. Mineralogical analysis also revealed change of fly ash mineral phases and specification distribution after water washing. Under liquid to solid ratio of 40∶1 L/kg and treatment time of 120 min, the leaching process achieved high dropping yields of toxicity characteristic leaching procedure (TCLP) concentrations for Cu, Zn Cd and Pb (80%−100%), moderate dropping yields for As (30%−80%) and relatively low dropping yields of Ni (< 30%). In addition, heavy metals such as Pb and Zn in fly ash with twice water washing treatment at a low liquid-solid ratio could reach lower TCLP concentrations. The result indicated that the combination process of twice water washing and one acid washing could significantly reduce the environmental risk of MSWI fly ash.
The chemical and mineralogical characteristics of fly ash from a municipal solid waste incineration (MSWI) in China and the influence of processing parameters on heavy metals removal during leaching were investigated in this work. The fly ash particles had complex surface structure with limited specific surface area. The alkali chloride and metal salts in MSWI fly ash showed evidently impact on leaching efficiency. Metal leachability was related to their properties and speciation in fly ash. Water-soluble salts such as KCl, NaCl and CaCl2 in fly ash were easily washed out. In this study, removal efficiency by water washing was achieved to 93.1% for Cl, 41.4% for Na, 48.5% for K and 24.8% for Ca, respectively. Mineralogical analysis also revealed change of fly ash mineral phases and specification distribution after water washing. Under liquid to solid ratio of 40∶1 L/kg and treatment time of 120 min, the leaching process achieved high dropping yields of toxicity characteristic leaching procedure (TCLP) concentrations for Cu, Zn Cd and Pb (80%−100%), moderate dropping yields for As (30%−80%) and relatively low dropping yields of Ni (< 30%). In addition, heavy metals such as Pb and Zn in fly ash with twice water washing treatment at a low liquid-solid ratio could reach lower TCLP concentrations. The result indicated that the combination process of twice water washing and one acid washing could significantly reduce the environmental risk of MSWI fly ash.
摘要:
In order to improve the tar quality by decreasing the heavy tar content and ensuring high tar yield, in-situ catalytic upgrading of tar from the integrated process of coal pyrolysis coupled with steam reforming of methane was conducted over carbon (KD-9) based Ni catalyst. The results show that at 650 °C, the tar yield of CP-SRM over 5Ni/KD-9 is 24.4%, which is a little lower than that of without catalyst, while the light tar yield (i.e.,18.9%) is 1.4 times higher than that of without catalyst, and the content of C2, C3 and C4 alkyl used as a substitute for benzene significantly increases tar yields by 0.5, 0.6 and 4.0 times, respectively. The content of phenols and naphthalenes in tar also increases dramatically after upgrading. Isotope tracer approach combined with the mass spectra of typical components was employed in exploring the mechanism of the upgrading process. The results show that 5Ni/KD-9 catalyzes coal tar cracking and SRM at the same time. Small free radicals such as ·CHx, ·H and ·OH generated by SRM can combine with free radicals from tar cracking, thus avoiding excessive cracking of tar.
In order to improve the tar quality by decreasing the heavy tar content and ensuring high tar yield, in-situ catalytic upgrading of tar from the integrated process of coal pyrolysis coupled with steam reforming of methane was conducted over carbon (KD-9) based Ni catalyst. The results show that at 650 °C, the tar yield of CP-SRM over 5Ni/KD-9 is 24.4%, which is a little lower than that of without catalyst, while the light tar yield (i.e.,18.9%) is 1.4 times higher than that of without catalyst, and the content of C2, C3 and C4 alkyl used as a substitute for benzene significantly increases tar yields by 0.5, 0.6 and 4.0 times, respectively. The content of phenols and naphthalenes in tar also increases dramatically after upgrading. Isotope tracer approach combined with the mass spectra of typical components was employed in exploring the mechanism of the upgrading process. The results show that 5Ni/KD-9 catalyzes coal tar cracking and SRM at the same time. Small free radicals such as ·CHx, ·H and ·OH generated by SRM can combine with free radicals from tar cracking, thus avoiding excessive cracking of tar.
摘要:
采用溶胶-凝胶-超临界干燥法、水热法及共沉淀法分别合成了氧化铈气凝胶(CeO2-A)、纳米棒(CeO2-R)和纳米片(CeO2-F)。考察了不同形貌氧化铈的催化燃烧甲苯性能,通过多种方法分析表征了氧化铈样品的微观结构,讨论了不同方法制得的CeO2形貌结构对催化性能的影响。结果表明,CeO2-R和CeO2-F比表面积较低,并且仅暴露(111)晶面,催化燃烧甲苯活性较低。CeO2-A具有高比表面积和丰富的孔道结构,有利于反应物分子的吸附,而且同时暴露(100)和(111)两种活性晶面,增加了氧空位浓度(Osur/Olatt = 0.25)。此外,CeO2-A由于表面晶格氧移动性较强,有利于Ce3+/Ce4+氧化还原的循环,加快甲苯深度氧化反应的进行。因此,CeO2-A具有更加优异的催化燃烧甲苯活性,t50和t90分别为223 和239 ℃,这主要归因于其大比表面积、高暴露活性晶面以及强晶格氧迁移性。
采用溶胶-凝胶-超临界干燥法、水热法及共沉淀法分别合成了氧化铈气凝胶(CeO2-A)、纳米棒(CeO2-R)和纳米片(CeO2-F)。考察了不同形貌氧化铈的催化燃烧甲苯性能,通过多种方法分析表征了氧化铈样品的微观结构,讨论了不同方法制得的CeO2形貌结构对催化性能的影响。结果表明,CeO2-R和CeO2-F比表面积较低,并且仅暴露(111)晶面,催化燃烧甲苯活性较低。CeO2-A具有高比表面积和丰富的孔道结构,有利于反应物分子的吸附,而且同时暴露(100)和(111)两种活性晶面,增加了氧空位浓度(Osur/Olatt = 0.25)。此外,CeO2-A由于表面晶格氧移动性较强,有利于Ce3+/Ce4+氧化还原的循环,加快甲苯深度氧化反应的进行。因此,CeO2-A具有更加优异的催化燃烧甲苯活性,t50和t90分别为223 和239 ℃,这主要归因于其大比表面积、高暴露活性晶面以及强晶格氧迁移性。
摘要:
考察了Pb对Mn-Ce/TiO2低温选择性催化还原(SCR)脱硝活性的影响,并对Pb中毒的催化剂进行了再生;结合氮吸附、SEM、XRD、FT-IR、H2-TPR和NH3-TPD等表征结果,研究了Mn-Ce/TiO2催化剂Pb中毒和再生活性恢复的原因。结果表明,Pb对Mn-Ce/TiO2催化剂脱硝活性有明显的抑制作用;当Pb的含量为11%时,Mn-Ce/TiO2催化剂在180 ℃下的脱硝效率从原来100%下降至44%。Pb在Mn-Ce/TiO2中的掺杂使得催化剂的比表面积以及活性组分Mn4+和Ce3+的含量降低,影响了氧化还原循环反应(Mn4+ + Ce3+ ↔ Mn3+ + Ce4+)的进行;此外,Pb的加入破坏了催化剂的酸性位点,阻碍了催化剂对NH3的吸附和活化。经硝酸再生后的Mn-Ce/TiO2催化剂的脱硝活性几乎完全恢复,在80–150 ℃下其脱硝活性甚至超过新鲜未中毒的催化剂,其原因主要在于硝酸再生能恢复催化剂的氧化还原能力、增大比表面积、并形成新的酸位点。
考察了Pb对Mn-Ce/TiO2低温选择性催化还原(SCR)脱硝活性的影响,并对Pb中毒的催化剂进行了再生;结合氮吸附、SEM、XRD、FT-IR、H2-TPR和NH3-TPD等表征结果,研究了Mn-Ce/TiO2催化剂Pb中毒和再生活性恢复的原因。结果表明,Pb对Mn-Ce/TiO2催化剂脱硝活性有明显的抑制作用;当Pb的含量为11%时,Mn-Ce/TiO2催化剂在180 ℃下的脱硝效率从原来100%下降至44%。Pb在Mn-Ce/TiO2中的掺杂使得催化剂的比表面积以及活性组分Mn4+和Ce3+的含量降低,影响了氧化还原循环反应(Mn4+ + Ce3+ ↔ Mn3+ + Ce4+)的进行;此外,Pb的加入破坏了催化剂的酸性位点,阻碍了催化剂对NH3的吸附和活化。经硝酸再生后的Mn-Ce/TiO2催化剂的脱硝活性几乎完全恢复,在80–150 ℃下其脱硝活性甚至超过新鲜未中毒的催化剂,其原因主要在于硝酸再生能恢复催化剂的氧化还原能力、增大比表面积、并形成新的酸位点。
摘要:
近年来,利用生物质基平台化合物转化制备各种燃料及高值化学品引起研究人员的广泛关注。5-羟甲基糠醛(HMF)和糠醛(FFR)作为一类重要的生物质衍生平台化合物,分子结构中醛基和呋喃环等官能团赋予其独特的化学性质。本综述针对HMF和FFR在氢气、低碳醇、甲酸和硅烷等不同氢源中的催化加氢反应研究现状进行了阐述,对加氢转化过程中的主要影响因素如催化剂类型和反应条件以及反应机理等进行了详细分析,同时对HMF/FFR加氢转化应用研究前景进行了展望。
近年来,利用生物质基平台化合物转化制备各种燃料及高值化学品引起研究人员的广泛关注。5-羟甲基糠醛(HMF)和糠醛(FFR)作为一类重要的生物质衍生平台化合物,分子结构中醛基和呋喃环等官能团赋予其独特的化学性质。本综述针对HMF和FFR在氢气、低碳醇、甲酸和硅烷等不同氢源中的催化加氢反应研究现状进行了阐述,对加氢转化过程中的主要影响因素如催化剂类型和反应条件以及反应机理等进行了详细分析,同时对HMF/FFR加氢转化应用研究前景进行了展望。
摘要:
化石资源的大量使用导致CO2的大量排放,带来了严重的环境问题。与此同时,CO2又是一种清洁、无毒的含碳资源。将CO2作为原料,直接转化制备重要化学品,不仅可以减缓温室效应,同时也是一条有效利用含碳资源制备清洁燃料和化学品的新路线。本文概述了近年来关于CO2加氢制备一些烃类化合物(主要包括甲烷、烯烃和芳烃)的相关研究进展;重点分析了CO2加氢制烃类化合物相关过程催化剂的研发状态和对催化反应机理的认识,并对CO2加氢转化利用的未来发展进行了展望。
化石资源的大量使用导致CO2的大量排放,带来了严重的环境问题。与此同时,CO2又是一种清洁、无毒的含碳资源。将CO2作为原料,直接转化制备重要化学品,不仅可以减缓温室效应,同时也是一条有效利用含碳资源制备清洁燃料和化学品的新路线。本文概述了近年来关于CO2加氢制备一些烃类化合物(主要包括甲烷、烯烃和芳烃)的相关研究进展;重点分析了CO2加氢制烃类化合物相关过程催化剂的研发状态和对催化反应机理的认识,并对CO2加氢转化利用的未来发展进行了展望。
摘要:
以生物质(葡萄糖、蔗糖、淀粉)为模板剂,通过水热合成法制备具有核壳结构的CuO-ZnO-Al2O3@Al2O3复合催化剂,该催化剂以甲醇合成催化剂CuO-ZnO-Al2O3为核,甲醇脱水催化剂Al2O3为壳.SEM-EDS对催化剂核壳结构的表征发现,通过改变水热合成温度和合成时间可以调变催化剂中Al2O3壳层的厚度.将该复合催化剂用于合成气直接制备二甲醚的反应,在空速1 500 mL/(h·gcat)、温度260 ℃、压力5.0 MPa的条件下,CO转化率和二甲醚选择性分别达到35.2%和61.1%.
以生物质(葡萄糖、蔗糖、淀粉)为模板剂,通过水热合成法制备具有核壳结构的CuO-ZnO-Al2O3@Al2O3复合催化剂,该催化剂以甲醇合成催化剂CuO-ZnO-Al2O3为核,甲醇脱水催化剂Al2O3为壳.SEM-EDS对催化剂核壳结构的表征发现,通过改变水热合成温度和合成时间可以调变催化剂中Al2O3壳层的厚度.将该复合催化剂用于合成气直接制备二甲醚的反应,在空速1 500 mL/(h·gcat)、温度260 ℃、压力5.0 MPa的条件下,CO转化率和二甲醚选择性分别达到35.2%和61.1%.
摘要:
通过10℃/min时的热重分析,对五种不同性质污泥分别在氮气和氧气气氛下的热解和燃烧特性进行了研究。结果表明,污水处理工艺的“好氧+厌氧”、“厌氧+好氧”过程以及污泥的厌氧消化均使污泥中的有机物结构复杂,导致污泥热解时有机物的分解和析出温度升高,且“好氧+厌氧”过程使污泥中有机物结构更复杂;而对污泥的燃烧过程和燃尽点无影响,但使着火温度升高。利用 šatava-šesták 方程对污泥热解、燃烧的反应机理进行了研究。结果表明,五种污泥热解时均呈现为挥发分扩散和随后的化学反应机理函数,而燃烧时则为化学反应和随后的扩散过程。
通过10℃/min时的热重分析,对五种不同性质污泥分别在氮气和氧气气氛下的热解和燃烧特性进行了研究。结果表明,污水处理工艺的“好氧+厌氧”、“厌氧+好氧”过程以及污泥的厌氧消化均使污泥中的有机物结构复杂,导致污泥热解时有机物的分解和析出温度升高,且“好氧+厌氧”过程使污泥中有机物结构更复杂;而对污泥的燃烧过程和燃尽点无影响,但使着火温度升高。利用 šatava-šesták 方程对污泥热解、燃烧的反应机理进行了研究。结果表明,五种污泥热解时均呈现为挥发分扩散和随后的化学反应机理函数,而燃烧时则为化学反应和随后的扩散过程。