Research progress on iron-based catalysts for CO2 hydrogenation to long-chain linear α-olefins
-
摘要: 长链线性α-烯烃(linear α-olefins, LAOs)是重要的化工原料,目前,主要通过石油化工路线获得。随着全球温升影响加剧,CO2控制与资源化利用技术研究受到持续关注。费托合成(Fischer-Tropsch synthesis, FTS)反应产生一定比例的LAOs,提供了制取LAOs可供选择的技术路线。本综述围绕CO2加氢制LAOs过程,结合其反应途径,分析了Fe基催化剂的研究进展,包括助催化剂和载体的作用,阐述了Fe基催化剂上链增长机理和影响LAOs选择性的关键因素,总结了该反应面临的挑战、可能的解决思路,对高效Fe基催化剂研究进行了展望。Abstract: Long-chain linear α-olefins (LAOs) are important industrial chemicals, which are mainly obtained from petrochemical process. With the increased impact of the greenhouse effect globally, research on CO2 control and mitigation has attracted much attention. Fischer-Tropsch synthesis (FTS) provides an alternative route to obtain LAOs. In this paper, research progress on iron-based catalysts including the roles of promoters and supports for the process of CO2 hydrogenation to LAOs are analyzed. Key factors affecting the selectivity of LAOs are discussed. Challenges and possible solutions of the reaction are summarized, and an outlook for designing high-efficient iron-based catalysts is thus presented.
-
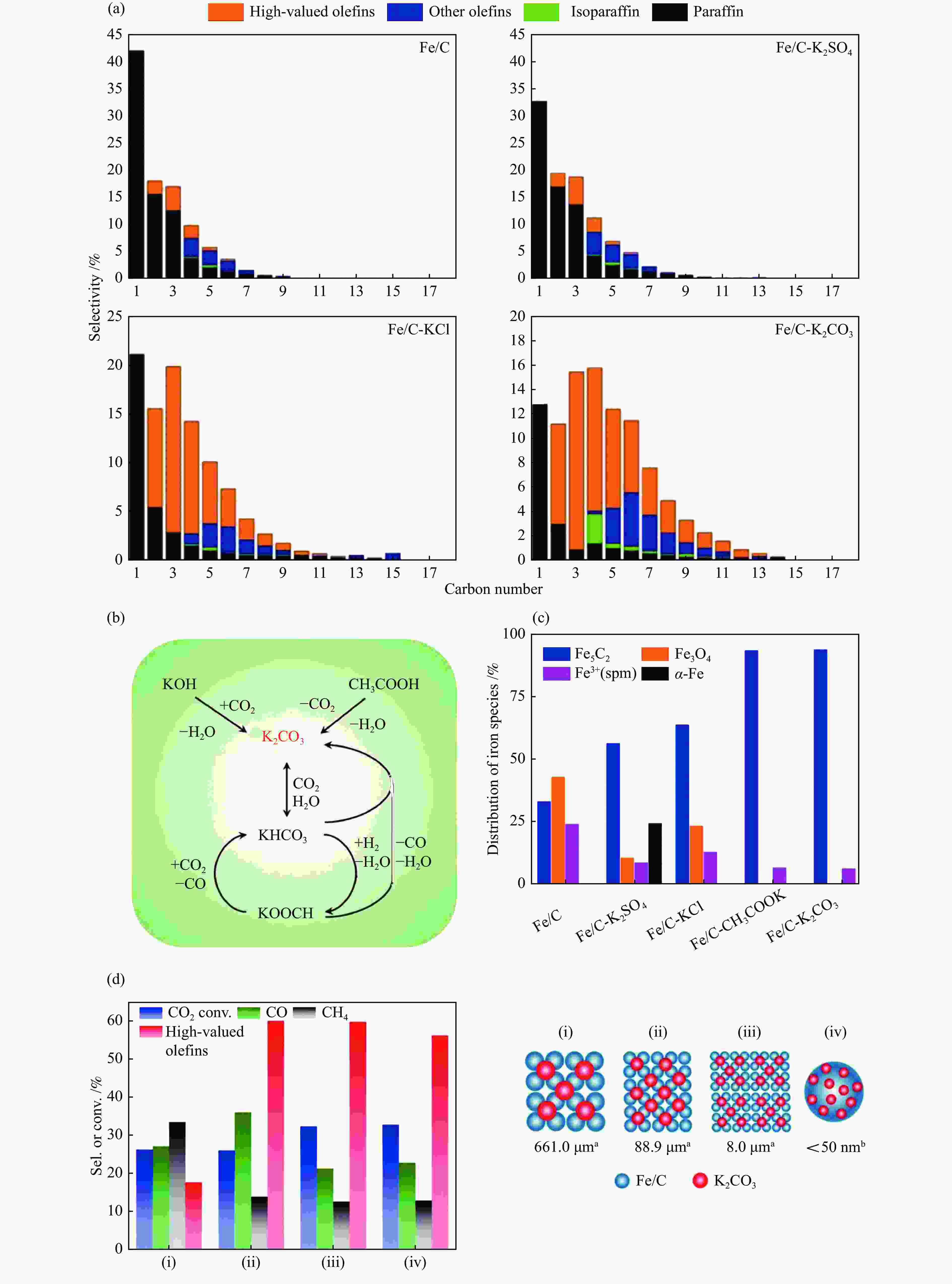
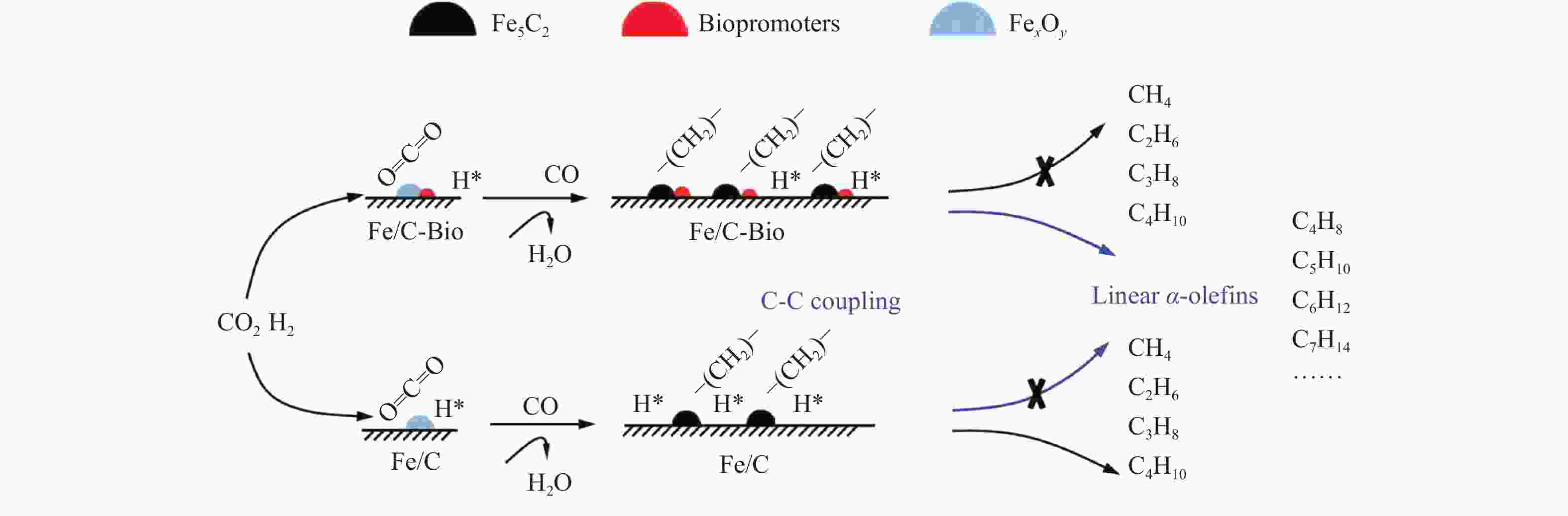
图 8 不同K助剂对Fe/C催化CO2加氢制长链烯烃的影响(a) 产物分布, (b) 含碳K助剂之间的相互转化过程, (c) 反应后催化剂中的Fe5C2含量, (d) 不同接触距离的Fe/C-K2CO3催化性能 (a:d50, b:TEM)[44]
Figure 8 Effect of different K promoters on Fe/C for CO2 hydrogenation to LAOs (a) product distribution, (b) interconversion of carbonaceous K-promoters, (c) Fe5C2 content of used catalysts, (d) catalytic performance on Fe/C-K2CO3 with different proximity (a:d50, b: estimated by TEM)[44](with permission from ACS Publications)
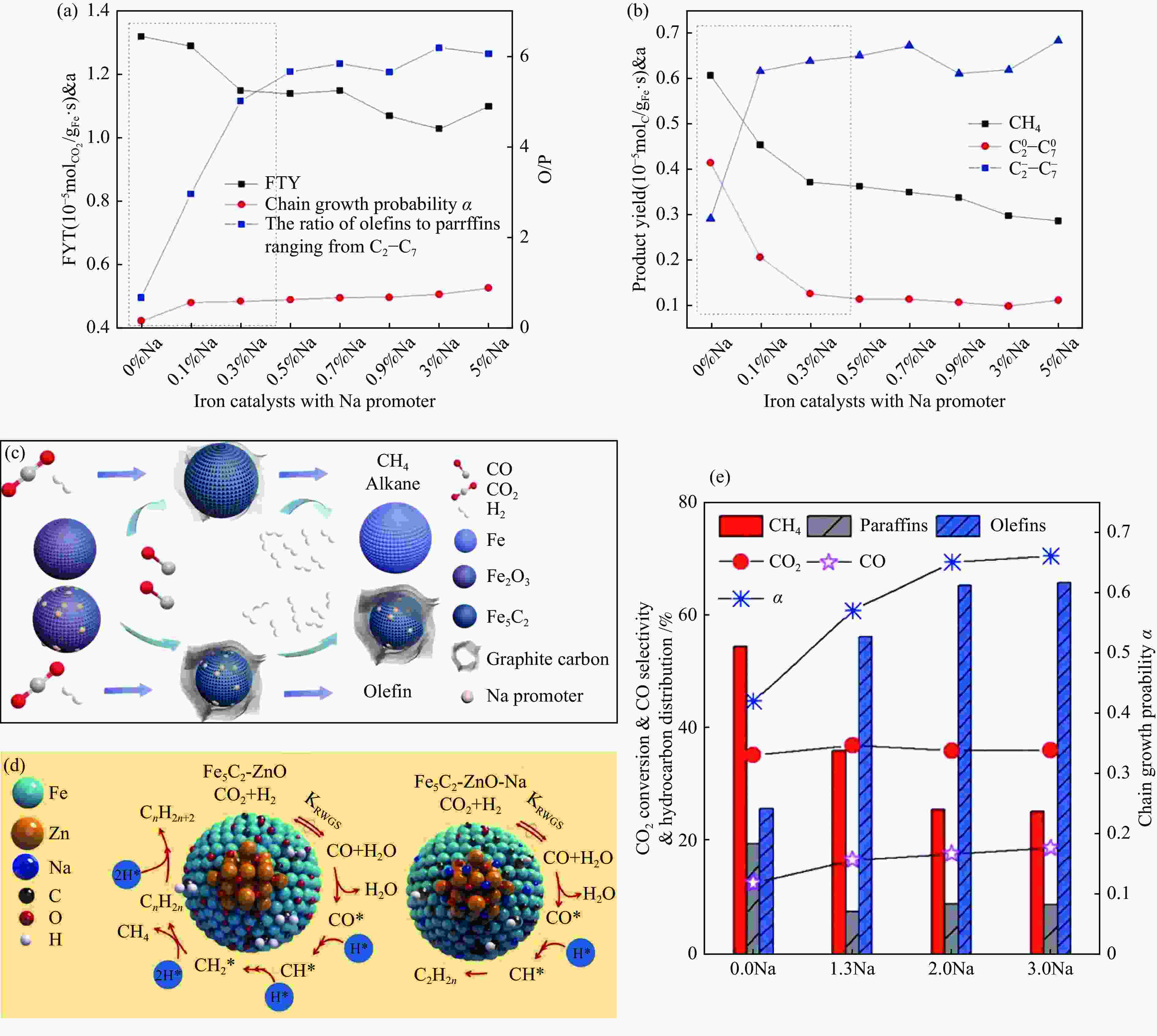
图 9 Na-Fe2O3催化CO2加氢制烯烃 (a) 时空收率、链增长因子、O/P值和(b) CH4、C2–C7烷烃、C2–C7烯烃产率与Na含量关系, (c) Na助剂在Fe、C物种演变中的作用[46],Fe5C2-ZnO-Na催化CO2加氢制烯烃,(d) Na促进作用机制, (e) Na含量对催化性能的影响[47]
Figure 9 CO2 hydrogenation to olefins on Na-Fe2O3 (a) FTY, chain growth probability, O/P value and (b) CH4,
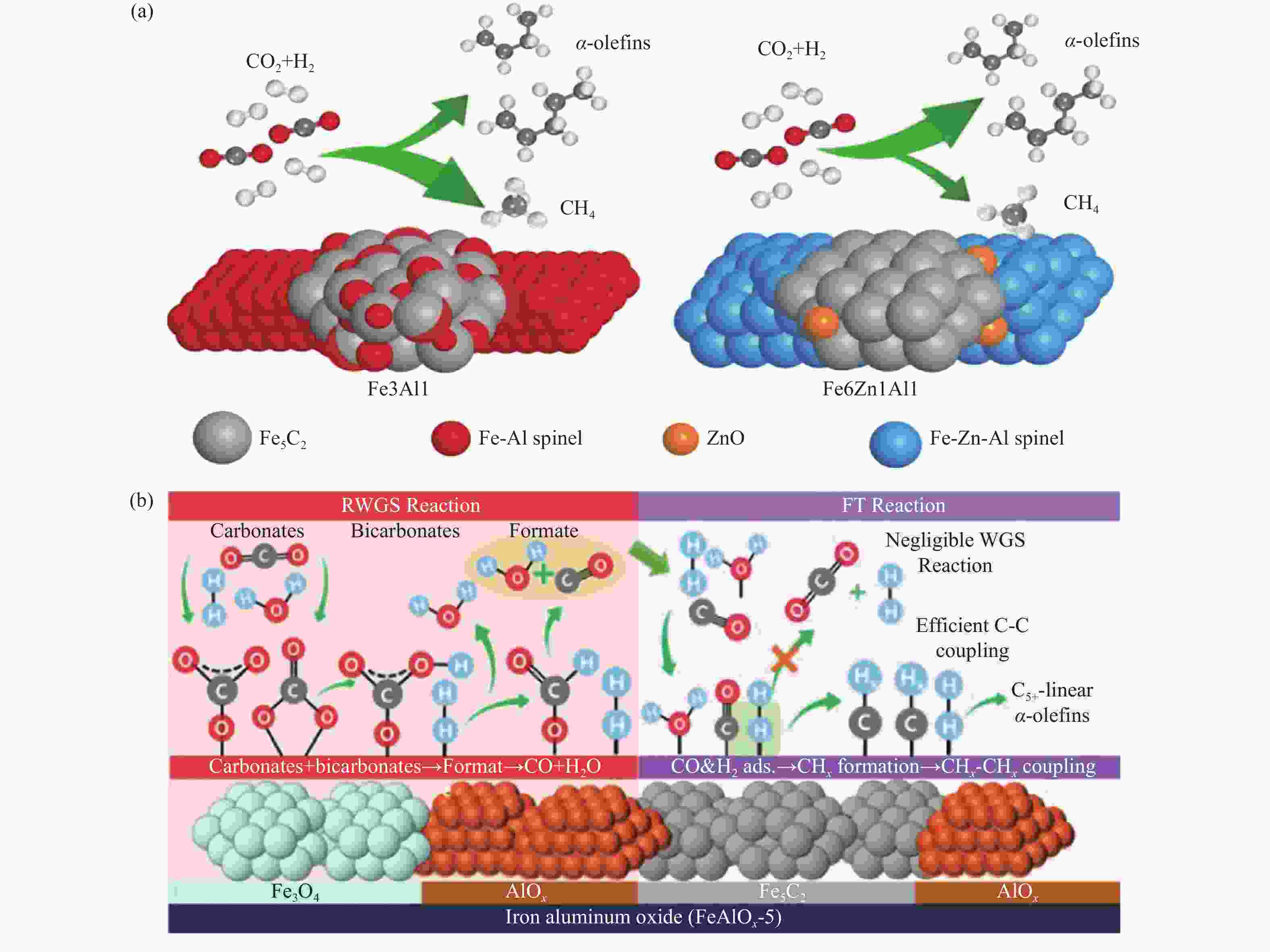
${\rm{C}}^0_2 -{\rm{C}}^0_7 $ ,${\rm{C}}^=_2 -{\rm{C}}^=_7 $ yields as a function of Na content, (c) role of Na promoter in the evolution of iron and carbon species[46], CO2 hydrogenation to olefins on Fe5C2-ZnO-Na, (d) promotion mechanism of Na, (e) effect of Na content on catalytic performance[47](with permission from Elsevier)图 10 Zn助剂对Fe基催化剂CO2加氢制LAOs影响:(a) Fe5C2-ZnO在CO2加氢过程中结构变化[53],(b) Fe2O3和Fe2Zn1催化稳定性[54]
Figure 10 Effect of Zn promoter on Fe-based catalysts for CO2 hydrogenation to LAOs: (a) structure evolution of Fe5C2-ZnO during CO2 hydrogenation[53] (b) catalytic stability of Fe2O3 and Fe2Zn1[54](with permission from Elsevier and ACS Publications)
表 1 Fe基催化剂CO2加氢制LAOs性能
Table 1 Performance of Fe-based catalysts for CO2 hydrogenation to LAOs
Catalyst Reaction conditions CO2 conv.
/%CO sel.
/%Hydrocarbon distribution/% LAOs
sel. /%Ref. H2/CO2 t/℃ p/MPa GHSV/(mL·g−1·h−1) CH4 ${\rm{C}}^=_2 -{\rm{C}}^=_4 $ ${\rm{C}}^=_{5+} $ Fe-K2CO3 3 320 1 2400 32.4 21.4 12.7 26.5 60.8 71.8 [44] Fe-Zn-Na 3 330 1.5 15000 43.5 9.2 11.5 16.6 60.7 89.3 [53] Fe-Mn-Na 3 340 2 12000 35 18.1 10.7 31.7 39.5 − [57] Fe-Cu-Na 3 300 1 1800 16.7 31.4 2.4 28.9 64.9 − [59] Fe-Zn-Al 3 350 1.5 15000 39.1 22.5 16 24.7 45.6 88.7 [60] FeAlOx 1 330 3.5 2000 20.2 16.8 5.4 11.7 66.8 78.4 [61] Fe/C-bio 3 320 3 2400 31 23.2 11.8 24.4 50.3 80 [62] Fe-K/Al2O3 3 320 3 3600 24.1 23.4 7.5 21.9 40.4 − [71] FeK/SWNTs 3 340 2 9000 52.7 9.6 13.5 30.7 39.8 − [88] 表 2 碱金属对Fe/ZrO2催化CO2加氢影响[43]
Table 2 Effect of alkali metals on Fe/ZrO2 for CO2 hydrogenation[43](with permission from Elsevier)
Alkali
metalCO2
conv.
/%Selectivity/% Hydrocarbon
distribution/%CO HC CH4 ${\rm{C}}^=_2 - {\rm{C}}^=_4 $ ${\rm{C}}^0_2 - {\rm{C}}^0_4 $ ${\rm{C}}^=_{5+} $ ${\rm{C}}^0_{5+} $ − 32 25 75 70 0.1 29 0.4 0.5 Li + 26 42 56 68 1.4 30 0.1 0.5 Na + 39 21 59 21 49 8.8 15 6.2 K + 43 15 66 18 44 9.2 19 9.8 Rb + 31 15 68 19 43 8.0 19 11 Cs + 39 16 67 26 43 9.6 14 7.4 -
[1] ZOU C N, XIONG B, XUE H Q, ZHENG D W, GE Z X, WANG Y, JIANG L Y, PAN S Q, WU S T. The role of new energy in carbon neutral[J]. Pet Explor Dev,2021,48(2):480−491. doi: 10.1016/S1876-3804(21)60039-3 [2] FANG K, LI C L, TANG Y Q, HE J J, SONG J N. China’s pathways to peak carbon emissions: New insights from various industrial sectors[J]. Appl Energy,2022,306:118039. doi: 10.1016/j.apenergy.2021.118039 [3] LANG X D, HE X, LI Z M, HE L N. New routes for CO2 activation and subsequent conversion[J]. Curr Opin Green Sustainable Chem,2017,7:31−38. doi: 10.1016/j.cogsc.2017.07.001 [4] SATTHAWONG R, KOIZUMI N, SONG C, PRASASSARAKICH P. Bimetallic Fe-Co catalysts for CO2 hydrogenation to higher hydrocarbons[J]. J CO2 Util,2013,3−4:102−106. doi: 10.1016/j.jcou.2013.10.002 [5] XUE L, ZHANG C J, WU J F, FAN Q Y, LIU Y, WU Y X, LI J X, ZHANG H, LIU F R, ZENG S H. Unveiling the reaction pathway on Cu/CeO2 catalyst for electrocatalytic CO2 reduction to CH4[J]. Appl Catal B: Environ,2022,304:120951. doi: 10.1016/j.apcatb.2021.120951 [6] MIAO Z R, WANG Q L, ZHANG Y F, MENG L P, WANG X X. In situ construction of S-scheme AgBr/BiOBr heterojunction with surface oxygen vacancy for boosting photocatalytic CO2 reduction with H2O[J]. Appl Catal B: Environ,2022,301:120802. doi: 10.1016/j.apcatb.2021.120802 [7] GUO L S, CUI Y, LI H J, FANG Y, PRASERT R, WU J, YANG G, YONEYAMA Y, TSUBAKI N. Selective formation of linear-alpha olefins (LAOS) by CO2 hydrogenation over bimetallic Fe/Co-Y catalyst[J]. Catal Commun,2019,130:105759. doi: 10.1016/j.catcom.2019.105759 [8] RA E C, KIM K Y, KIM E H, LEE H, AN K, LEE J S. Recycling carbon dioxide through catalytic hydrogenation: Recent key developments and perspectives[J]. ACS Catal,2020,10(19):11318−11345. doi: 10.1021/acscatal.0c02930 [9] ROY S, CHEREVOTAN A, PETER S C. Thermochemical CO2 hydrogenation to single carbon products: Scientific and technological challenges[J]. ACS Energy Lett,2018,3(8):1938−1966. doi: 10.1021/acsenergylett.8b00740 [10] GUO L S, SUN J, GE Q J, TSUBAKI N. Recent advances in direct catalytic hydrogenation of carbon dioxide to valuable C2 + hydrocarbons[J]. J Mater Chem A,2018,6(46):23244−23262. doi: 10.1039/C8TA05377D [11] WEI J, YAO R W, HAN Y, GE Q J, SUN J. Towards the development of the emerging process of CO2 heterogenous hydrogenation into high-value unsaturated heavy hydrocarbons[J]. Chem Soc Rev,2021,50(19):10764−10805. doi: 10.1039/D1CS00260K [12] WANG D, XIE Z H, POROSOFF M D, CHEN J G. Recent advances in carbon dioxide hydrogenation to produce olefins and aromatics[J]. Chem,2021,7(9):2277−2311. doi: 10.1016/j.chempr.2021.02.024 [13] 王晗, 樊升, 王森, 董梅, 秦张峰, 樊卫斌, 王建国. 二氧化碳加氢制一些烃类化合物的研究进展[J]. 燃料化学学报,2021,49(11):1609−1619. doi: 10.1016/S1872-5813(21)60122-6WANG Han, FAN Sheng, WANG Sen, DONG Mei, QIN Zhang-feng, FAN Wei-bin, WANG Jian-guo. Research progresses in the hydrogenation of carbon dioxide to certain hydrocarbon products[J]. J Fuel Chem Technol,2021,49(11):1609−1619. doi: 10.1016/S1872-5813(21)60122-6 [14] MA Z, POROSOFF M D. Development of tandem catalysts for CO2 hydrogenation to olefins[J]. ACS Catal,2019,9(3):2639−2656. doi: 10.1021/acscatal.8b05060 [15] PAWELEC B, GUIL-L PEZ R, MOTA N, FIERRO J L G, NAVARRO YERGA R M. Catalysts for the conversion of CO2 to low molecular weight olefins-A review[J]. Materials,2021,14(22):6952. doi: 10.3390/ma14226952 [16] GAO P, DANG S S, LI S G, BU X N, LIU Z Y, QIU M H, YANG C G, WANG H, ZHONG L S, HAN Y, LIU Q, WEI W, SUN Y H. Direct production of lower olefins from CO2 conversion via bifunctional catalysis[J]. ACS Catal,2017,8(1):571−578. [17] LI Z L, WANG J J, QU Y Z, LIU H L, TANG C Z, MIAO S, FENG Z C, AN H Y, LI C. Highly selective conversion of carbon dioxide to lower olefins[J]. ACS Catal,2017,7(12):8544−8548. doi: 10.1021/acscatal.7b03251 [18] GAO P, LI S G, BU X N, DANG S S, LIU Z Y, WANG H, ZHONG L S, QIU M H, YANG C G, CAI J, WEI W, SUN Y H. Direct conversion of CO2 into liquid fuels with high selectivity over a bifunctional catalyst[J]. Nat Chem,2017,9(10):1019−1024. doi: 10.1038/nchem.2794 [19] 纪学玮, 葛庆杰, 孙剑. 协同高效的铁催化剂构建及其在 CO2 加氢制高碳烃中的应用[J]. 燃料化学学报,2019,47(4):438−445.JI Xue-wei, GE Qing-jie, SUN Jian. Construction of synergistic and efficient iron-based catalysts for hydrogenation of CO2 to higher hydrocarbons[J]. J Fuel Chem Technol,2019,47(4):438−445. [20] ALBRECHT M, RODEMERCK U, SCHNEIDER M, BR RING M, BAABE D, KONDRATENKO E V. Unexpectedly efficient CO2 hydrogenation to higher hydrocarbons over non-doped Fe2O3[J]. Appl Catal B: Environ,2017,204:119−126. doi: 10.1016/j.apcatb.2016.11.017 [21] WANG Q, CHEN Y, LI Z H. Research progress of catalysis for low-carbon olefins synthesis through hydrogenation of CO2[J]. J Nanosci Nanotechnol,2019,19(6):3162−3172. doi: 10.1166/jnn.2019.16586 [22] 张超, 张玉龙, 朱明辉, 孟博, 涂维峰, 韩一帆. CO2高值化利用新途径: 铁基催化剂CO2加氢制烯烃研究进展[J]. 化工进展,2021,40(2):577−593.ZHANG Chao, ZHANG Yu-long, ZHU Ming-hui, MENG Bo, TU Wei-feng, HAN Yi-fan. New pathway for CO2 high-valued utilization: Fe-based catalysts for CO2 hydrogenation to low olefins[J]. Chem Ind Eng Prog,2021,40(2):577−593. [23] AMOYAL M, VIDRUK-NEHEMYA R, LANDAU M V, HERSKOWITZ M. Effect of potassium on the active phases of Fe catalysts for carbon dioxide conversion to liquid fuels through hydrogenation[J]. J Catal,2017,348:29−39. doi: 10.1016/j.jcat.2017.01.020 [24] WEBER D, HE T, WONG M, MOON C, ZHANG A, FOLEY N, RAMER N J, ZHANG C. Recent advances in the mitigation of the catalyst deactivation of CO2 hydrogenation to light olefins[J]. Catalysts,2021,11(12):1447. doi: 10.3390/catal11121447 [25] 李梦青, 邓国才, 陈荣悌, 刘崇微, 朱起明. FeCoMnK/BeO催化剂上二氧化碳加氢合成低碳烯烃的反应性能和原位FT-IR研究[J]. 催化学报,2000,21(1):71−74. doi: 10.3321/j.issn:0253-9837.2000.01.020LI Meng-qing, DENG Guo-cai, CHEN Rong-ti, LIU Chong-wei, ZHU Qi-ming. Synthesis of light olefins from CO2 hydrogenation on FeCoMnK/BeO catalysts and in situ FT-IR studies[J]. Chin J Catal,2000,21(1):71−74. doi: 10.3321/j.issn:0253-9837.2000.01.020 [26] RIEDEL T, SCHAUB G, JUN K-W, LEE K-W. Kinetics of CO2 hydrogenation on a K-promoted Fe catalyst[J]. Ind Eng Chem Res,2001,40(5):1355−1363. doi: 10.1021/ie000084k [27] LIU J H, ZHANG G H, JIANG X, WANG J H, SONG C S, GUO X W. Insight into the role of Fe5C2 in CO2 catalytic hydrogenation to hydrocarbons[J]. Catal Today,2021,371:162−170. doi: 10.1016/j.cattod.2020.07.032 [28] FISCHER F, TROPSCH H. The synthesis of petroleum at atmospheric pressures from gasification products of coal[J]. Brennst-Chem,1926,7:97−104. [29] LEE S C, JANG J H, LEE B Y, KIM J S, KANG M, LEE S B, CHOI M J, CHOUNG S J. Promotion of hydrocarbon selectivity in CO2 hydrogenation by Ru component[J]. J Mol Catal A: Chem,2004,210(1/2):131−141. doi: 10.1016/j.molcata.2003.09.013 [30] SAEIDI S, NAJARI S, HESSEL V, WILSON K, KEIL F J, CONCEPCI N P, SUIB S L, RODRIGUES A E. Recent advances in CO2 hydrogenation to value-added products-current challenges and future directions[J]. Prog Energy Combust Sci,2021,85:100905. doi: 10.1016/j.pecs.2021.100905 [31] SCHULZ H, RIEDEL T, SCHAUB G. Fischer-Tropsch principles of co-hydrogenation on iron catalysts[J]. Top Catal,2005,32(3):117−124. [32] SCHULZ H, SCHAUB G, CLAEYS M, RIEDEL T. Transient initial kinetic regimes of Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,1999,186(1):215−227. [33] PANZONE C, PHILIPPE R, CHAPPAZ A, FONGARLAND P, BENGAOUER A. Power-to-liquid catalytic CO2 valorization into fuels and chemicals: Focus on the Fischer-Tropsch route[J]. J CO2 Util,2020,38:314−347. doi: 10.1016/j.jcou.2020.02.009 [34] SCHULZ H. Selforganization in Fischer-Tropsch synthesis with iron- and cobalt catalysts[J]. Catal Today,2014,228:113−122. doi: 10.1016/j.cattod.2013.11.060 [35] LI W H, WANG H Z, JIANG X, ZHU J, LIU Z M, GUO X W, SONG C S. A short review of recent advances in CO2 hydrogenation to hydrocarbons over heterogeneous catalysts[J]. RSC Adv,2018,8(14):7651−7669. doi: 10.1039/C7RA13546G [36] LIU Y Y, CHEN B J, LIU R, LIU W Q, GAO X H, TAN Y S, ZHANG Z Z, TU W F. CO2 hydrogenation to olefins on supported iron catalysts: effects of support properties on carbon-containing species and product distribution[J]. Fuel,2022,324:124649. doi: 10.1016/j.fuel.2022.124649 [37] LEE S C, JANG J H, LEE B Y, KANG M C, KANG M, CHOUNG S J. The effect of binders on structure and chemical properties of Fe-K/γ-Al2O3 catalysts for CO2 hydrogenation[J]. Appl Catal A: Gen,2003,253(1):293−304. doi: 10.1016/S0926-860X(03)00540-4 [38] ZHOU W, CHENG K, KANG J C, ZHOU C, SUBRAMANIAN V, ZHANG Q H, WANG Y. New horizon in C1 chemistry: Breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels[J]. Chem Soc Rev,2019,48(12):3193−3228. doi: 10.1039/C8CS00502H [39] JIANG J D, WEN C Y, TIAN Z P, WANG Y C, ZHAI Y P, CHEN L G, LI Y P, LIU Q Y, WANG C G, MA L L. Manganese-promoted Fe3O4 microsphere for efficient conversion of CO2 to light olefins[J]. Ind Eng Chem Res,2020,59(5):2155−2162. doi: 10.1021/acs.iecr.9b05342 [40] 吴大凯, 王旭, 高新华, 马清祥, 张建利, 范素兵, 赵天生. 层状K-Fe-Zn-Ti催化剂的制备及其对二氧化碳加氢制烯烃反应的催化性能[J]. 燃料化学学报,2019,47(8):949−956. doi: 10.1016/S1872-5813(19)30040-4WU Da-kai, WANG Xu, GAO Xin-hua, MA Qing-xiang, ZHANG Jian-li, FAN Su-bing, ZHAO Tian-sheng. Preparation of layered K-Fe-Zn-Ti catalyst and its performance in the hydrogenation of carbon dioxide to light olefins[J]. J Fuel Chem Technol,2019,47(8):949−956. doi: 10.1016/S1872-5813(19)30040-4 [41] GUPTA S, JAIN V K, JAGADEESAN D. Fine tuning the composition and nanostructure of Fe-based core-shell nanocatalyst for efficient CO2 hydrogenation[J]. ChemNanoMat,2016,2(10):989−996. doi: 10.1002/cnma.201600234 [42] ELISHAV O, SHENER Y, BEILIN V, LANDAU M V, HERSKOWITZ M, SHTER G E, GRADER G S. Electrospun Fe-Al-O nanobelts for selective CO2 hydrogenation to light olefins[J]. ACS Appl Mater Interfaces,2020,12(22):24855−24867. doi: 10.1021/acsami.0c05765 [43] WANG J J, YOU Z Y, ZHANG Q H, DENG W P, WANG Y. Synthesis of lower olefins by hydrogenation of carbon dioxide over supported iron catalysts[J]. Catal Today,2013,215:186−193. doi: 10.1016/j.cattod.2013.03.031 [44] HAN Y, FANG C Y, JI X W, WEI J, GE Q J, SUN J. Interfacing with carbonaceous potassium promoters boosts catalytic CO2 hydrogenation of iron[J]. ACS Catal,2020,10(20):12098−12108. doi: 10.1021/acscatal.0c03215 [45] WANG S W, JI Y S, LIU X C, YAN S R, XIE S H, PEI Y, LI H X, QIAO M H, ZONG B N. Potassium as a versatile promoter to tailor the distribution of the olefins in CO2 hydrogenation over iron-based catalyst[J]. ChemCatChem,2022,14(6):e202101535. [46] WEI C Y, TU W F, JIA L Y, LIU Y Y, LIAN H L, WANG P, ZHANG Z Z. The evolutions of carbon and iron species modified by Na and their tuning effect on the hydrogenation of CO2 to olefins[J]. Appl Surf Sci,2020,525:146622. doi: 10.1016/j.apsusc.2020.146622 [47] TU W F, SUN C, ZHANG Z Z, LIU W Q, MALHI H S, MA W, ZHU M H, HAN Y F. Chemical and structural properties of Na decorated Fe5C2-ZnO catalysts during hydrogenation of CO2 to linear alpha-olefins[J]. Appl Catal B: Environ,2021,298:120567. doi: 10.1016/j.apcatb.2021.120567 [48] HESKETT D. The interaction range in alkali metal-promoted systems[J]. Surf Sci,1988,199(1):67−86. [49] LI J F, CHENG X F, ZHANG C H, CHANG Q, WANG J, WANG X P, LV Z G, DONG W S, YANG Y, LI Y W. Effect of alkalis on iron-based Fischer-Tropsch synthesis catalysts: Alkali-FeOx interaction, reduction, and catalytic performance[J]. Appl Catal A: Gen,2016,528:131−141. doi: 10.1016/j.apcata.2016.10.006 [50] LI J F, CHENG X F, ZHANG C H, WANG J, DONG W S, YANG Y, LI Y W. Alkalis in iron-based Fischer-Tropsch synthesis catalysts: distribution, migration and promotion[J]. J Chem Technol Biotechnol,2017,92(6):1472−1480. doi: 10.1002/jctb.5152 [51] GAO X H, ZHANG J L, CHEN N, MA Q X, FAN S B, ZHAO T S, TSUBAKI N. Effects of zinc on Fe-based catalysts during the synthesis of light olefins from the Fischer-Tropsch process[J]. Chin J Catal,2016,37(4):510−516. doi: 10.1016/S1872-2067(15)61051-8 [52] ZHANG J L, LU S P, SU X J, FAN S B, MA Q X, ZHAO T S. Selective formation of light olefins from CO2 hydrogenation over Fe-Zn-K catalysts[J]. J CO2 Util,2015,12:95−100. doi: 10.1016/j.jcou.2015.05.004 [53] ZHANG C, XU M J, YANG Z X, ZHU M H, GAO J, HAN Y F. Uncovering the electronic effects of zinc on the structure of Fe5C2-ZnO catalysts for CO2 hydrogenation to linear alpha-olefins[J]. Appl Catal B: Environ,2021,295:120287. doi: 10.1016/j.apcatb.2021.120287 [54] ZHANG C, CAO C X, ZHANG Y L, LIU X L, XU J, ZHU M H, TU W F, HAN Y F. Unraveling the role of zinc on bimetallic Fe5C2-ZnO catalysts for highly selective carbon dioxide hydrogenation to high carbon alpha-olefins[J]. ACS Catal,2021,11(4):2121−2133. doi: 10.1021/acscatal.0c04627 [55] ZHU J, WANG P, ZHANG X B, ZHANG G H, LI R T, LI W H, SENFTLE THOMAS P, LIU W, WANG J Y, WANG Y L, ZHANG A F, FU Q, SONG C S, GUO X W. Dynamic structural evolution of iron catalysts involving competitive oxidation and carburization during CO2 hydrogenation[J]. Sci Adv,2022,8(5):eabm3629. doi: 10.1126/sciadv.abm3629 [56] DE SMIT E, WECKHUYSEN B M. The renaissance of iron-based Fischer-Tropsch synthesis: on the multifaceted catalyst deactivation behaviour[J]. Chem Soc Rev,2008,37(12):2758−2781. doi: 10.1039/b805427d [57] XU Y, ZHAI P, DENG Y C, XIE J L, LIU X, WANG S, MA D. Highly selective olefin production from CO2 hydrogenation on iron catalysts: a subtle synergy between manganese and sodium additives[J]. Angew Chem Int Ed Eng,2020,59(48):21736−21744. doi: 10.1002/anie.202009620 [58] AL-DOSSARY M, ISMAIL A A, FIERRO J L G, BOUZID H, AL-SAYARI S A. Effect of Mn loading onto MnFeO nanocomposites for the CO2 hydrogenation reaction[J]. Appl Catal B: Environ,2015,165:651−660. doi: 10.1016/j.apcatb.2014.10.064 [59] CHOI Y H, JANG Y J, PARK H, KIM W Y, LEE Y H, CHOI S H, LEE J S. Carbon dioxide Fischer-Tropsch synthesis: A new path to carbon-neutral fuels[J]. Appl Catal B: Environ,2017,202:605−610. doi: 10.1016/j.apcatb.2016.09.072 [60] XU M J, LIU X L, CAO C X, SUN Y, ZHANG C, YANG Z X, ZHU M H, DING X X, LIU Y T, TONG Z F, XU J. Ternary Fe-Zn-Al spinel catalyst for CO2 hydrogenation to linear alpha-olefins: Synergy effects between Al and Zn[J]. ACS Sustainable Chem Eng,2021,9(41):13818−13830. doi: 10.1021/acssuschemeng.1c04629 [61] KHAN M K, BUTOLIA P, JO H, IRSHAD M, HAN D, NAM K W, KIM J. Selective conversion of carbon dioxide into liquid hydrocarbons and long-chain α-olefins over Fe-Amorphous AlOx bifunctional catalysts[J]. ACS Catal,2020,10(18):10325−10338. doi: 10.1021/acscatal.0c02611 [62] GUO L S, SUN J, JI X W, WEI J, WEN Z Y, YAO R W, XU H Y, GE Q J. Directly converting carbon dioxide to linear α-olefins on bio-promoted catalysts[J]. Commun Chem,2018,1(1):11. doi: 10.1038/s42004-018-0012-4 [63] WANG L C, TAHVILDAR KHAZANEH M, WIDMANN D, BEHM R J. TAP reactor studies of the oxidizing capability of CO2 on a Au/CeO2 catalyst-a first step toward identifying a redox mechanism in the reverse water-gas shift reaction[J]. J Catal,2013,302:20−30. doi: 10.1016/j.jcat.2013.02.021 [64] YIN X L, WANG S, SUN R X, JIANG S X, SHEN L H. A Ce-Fe oxygen carrier with a core-shell structure for chemical looping steam methane reforming[J]. Ind Eng Chem Res,2020,59(21):9775−9786. doi: 10.1021/acs.iecr.0c00055 [65] WATANABE S, MA X L, SONG C S. Characterization of structural and surface properties of nanocrystalline TiO2-CeO2 mixed oxides by XRD, XPS, TPR, and TPD[J]. J Phy Chem C,2009,113(32):14249−14257. doi: 10.1021/jp8110309 [66] ZHANG J L, SU X J, WANG X, MA Q X, FAN S B, ZHAO T S. Promotion effects of Ce added Fe-Zr-K on CO2 hydrogenation to light olefins[J]. React Kinet Mech Catal,2018,124(2):575−585. doi: 10.1007/s11144-018-1377-1 [67] DORNER R W, HARDY D R, WILLIAMS F W, WILLAUER H D. Effects of ceria-doping on a CO2 hydrogenation iron-manganese catalyst[J]. Catal Commun,2010,11(9):816−819. doi: 10.1016/j.catcom.2010.02.024 [68] DORNER R W, HARDY D R, WILLIAMS F W, WILLAUER H D. C2-C5 + olefin production from CO2 hydrogenation using ceria modified Fe/Mn/K catalysts[J]. Catal Commun,2011,15(1):88−92. doi: 10.1016/j.catcom.2011.08.017 [69] MACLACHLAN M J, GINZBURG M, COOMBS N, COYLE T W, RAJU N P, GREEDAN J E, OZIN G A, MANNERS I I. Shaped ceramics with tunable magnetic properties from metal-containing polymers[J]. Science,2000,287(5457):1460−1463. doi: 10.1126/science.287.5457.1460 [70] DING F S, ZHANG A F, LIU M, ZUO Y, LI K Y, GUO X W, SONG C S. CO2 hydrogenation to hydrocarbons over iron-based catalyst: Effects of physicochemical properties of Al2O3 supports[J]. Ind Eng Chem Res,2014,53(45):17563−17569. doi: 10.1021/ie5031166 [71] LIU J H, ZHANG A F, JIANG X, LIU M, ZHU J, SONG C S, GUO X W. Direct transformation of carbon dioxide to value-added hydrocarbons by physical mixtures of Fe5C2 and K-modified Al2O3[J]. Ind Eng Chem Res,2018,57(28):9120−9126. doi: 10.1021/acs.iecr.8b02017 [72] NUMPILAI T, CHANLEK N, POO-ARPORN Y, WANNAPAIBOON S, CHENG C K, SIRI-NGUAN N, SORNCHAMNI T, KONGKACHUICHAY P, CHAREONPANICH M, RUPPRECHTER G, LIMTRAKUL J, WITOON T. Pore size effects on physicochemical properties of Fe-Co/K-Al2O3 catalysts and their catalytic activity in CO2 hydrogenation to light olefins[J]. Appl Surf Sci,2019,483:581−592. doi: 10.1016/j.apsusc.2019.03.331 [73] ZHU J, ZHANG G H, LI W H, ZHANG X B, DING F S, SONG C S, GUO X W. Deconvolution of the particle size effect on CO2 Hydrogenation over iron-based catalysts[J]. ACS Catal,2020,10(13):7424−7433. doi: 10.1021/acscatal.0c01526 [74] RODEMERCK U, HOLEŇA M, WAGNER E, SMEJKAL Q, BARKSCHAT A, BAERNS M. Catalyst development for CO2 hydrogenation to fuels[J]. ChemCatChem,2013,5(7):1948−1955. doi: 10.1002/cctc.201200879 [75] BORERIBOON N, JIANG X, SONG C, PRASASSARAKICH P. Higher hydrocarbons synthesis from CO2 hydrogenation over K- and La-promoted Fe-Cu/TiO2 catalysts[J]. Top Catal,2018,61(15/17):1551−1562. doi: 10.1007/s11244-018-1023-1 [76] BORERIBOON N, JIANG X, SONG C, PRASASSARAKICH P. Fe-based bimetallic catalysts supported on TiO2 for selective CO2 hydrogenation to hydrocarbons[J]. J CO2 Util,2018,25:330−337. doi: 10.1016/j.jcou.2018.02.014 [77] LIN Y P, ZHU Y F, PAN X L, BAO X H. Modulating the methanation activity of Ni by the crystal phase of TiO2[J]. Catal Sci Technol,2017,7(13):2813−2818. doi: 10.1039/C7CY00124J [78] YU J, YU J H, SHI Z P, GUO Q S, XIAO X Z, MAO H F, MAO D S. The effects of the nature of TiO2 supports on the catalytic performance of Rh-Mn/TiO2 catalysts in the synthesis of C2 oxygenates from syngas[J]. Catal Sci Technol,2019,9(14):3675−3685. doi: 10.1039/C9CY00406H [79] LI W H, ZHANG G H, JIANG X, LIU Y, ZHU J, DING F S, LIU Z M, GUO X W, SONG C S. CO2 hydrogenation on unpromoted and M-promoted Co/TiO2 catalysts (M=Zr, K, Cs): Effects of crystal phase of supports and metal-support interaction on tuning product distribution[J]. ACS Catal,2019,9(4):2739−2751. doi: 10.1021/acscatal.8b04720 [80] TORRENTE-MURCIANO L, CHAPMAN R S, NARVAEZ-DINAMARCA A, MATTIA D, JONES M D. Effect of nanostructured ceria as support for the iron catalysed hydrogenation of CO2 into hydrocarbons[J]. Phys Chem Chem Phys,2016,18(23):15496−15500. doi: 10.1039/C5CP07788E [81] LóPEZ J M, GILBANK A L, GARCíA T, SOLSONA B, AGOURAM S, TORRENTE-MURCIANO L. The prevalence of surface oxygen vacancies over the mobility of bulk oxygen in nanostructured ceria for the total toluene oxidation[J]. Appl Catal B: Environ,2015,174−175:403−412. doi: 10.1016/j.apcatb.2015.03.017 [82] CHERNYAK S A, IVANOV A S, STOLBOV D N, MAKSIMOV S V, SAVILOV S V. Sintered Fe/CNT framework catalysts for CO2 hydrogenation into hydrocarbons[J]. Carbon,2020,168:475−484. doi: 10.1016/j.carbon.2020.06.067 [83] ZHANG Z P, ZHANG J, XU W, RUI S, HAN Y F. Promotional effects of multiwalled carbon nanotubes on iron catalysts for Fischer-Tropsch to olefins[J]. J Catal,2018,365:71−85. doi: 10.1016/j.jcat.2018.05.021 [84] DAI L Y, CHEN Y, LIU R J, LI X, ULLAH N, LI Z H. CO2 hydrogenation to C5 + hydrocarbons over K-promoted Fe/CNT catalyst: Effect of potassium on structure-activity relationship[J]. Appl Organomet Chem,2021,35(7):e6253. [85] WU T J, LIN J, CHENG Y, TIAN J, WANG S W, XIE S H, PEI Y, YAN S R, QIAO M H, XU H L, ZONG B N. Porous graphene-confined Fe-K as highly efficient catalyst for CO2 direct hydrogenation to light olefins[J]. ACS Appl Mater Interfaces,2018,10(28):23439−23443. doi: 10.1021/acsami.8b05411 [86] ZHANG P Z, HAN F, YAN J Y, QIAO X L, GUAN Q X, LI W. N-doped ordered mesoporous carbon (N-OMC) confined Fe3O4-FeCx heterojunction for efficient conversion of CO2 to light olefins[J]. Appl Catal B: Environ,2021,299:120639. doi: 10.1016/j.apcatb.2021.120639 [87] WITOON T, NUMPILAI T, NUEANGNORAJ K, CHENG C K, CHAREONPANICH M, LIMTRAKUL J. Light olefins synthesis from CO2 hydrogenation over mixed Fe-Co-K supported on micro-mesoporous carbon catalysts[J]. Int J Hydrogen Energy, 2021. https://doi.org/10.1016/j.ijhydene.2021.10.265 [88] WANG S W, WU T J, LIN J, JI Y S, YAN S R, PEI Y, XIE S H, ZONG B N, QIAO M H. Iron-potassium on single-walled carbon nanotubes as efficient catalyst for CO2 hydrogenation to heavy olefins[J]. ACS Catal,2020,10(11):6389−6401. doi: 10.1021/acscatal.0c00810 -





 下载:
下载: