Investigate of the optimum process conditions for Co/HZSM-5 catalyzed propane dehydrogenation by a response surface method
-
摘要: 采用浸渍法制备Co/HZSM-5催化剂用于丙烷催化脱氢制丙烯反应,对其进行预处理,使反应可在低温下进行,然后采用响应曲面考察工艺条件对该反应的影响。利用XRD、SEM、NH3-TPD、H2-TPR、氮气物理吸附-脱附等手段对催化剂进行表征,并在固定床反应器上对催化剂性能进行评价。结合实际实验条件可得最佳工艺条件为:反应温度461 ℃、Co负载量2.4%、GHSV为4300 h−1;此时,丙烯收率为27.7%,对应的丙烯选择性93.8%。
-
关键词:
- 响应曲面 /
- 工艺条件 /
- 丙烷脱氢 /
- Co/HZSM-5催化剂
Abstract: Co/HZSM-5 catalyst was fabricated for catalytic dehydrogenation of propane to propylene, which was pretreated to allow the reaction to react at low temperatures. A response surface approach was employed to examine the effect of process conditions on the reaction. The morphological and oxidative performance of Co/HZSM-5 was characterized by XRD, XPS, SEM, NH3-TPD, H2-TPR, and nitrogen physical absorption-desorption. Besides, the in-situ catalyst performance was evaluated by a fixed-bed reactor. Combining the actual experimental conditions, the optimal process conditions parameters obtained by the response surface method were as follows: a reaction temperature of 461 °C, a Co loading of 2.4%, and a GHSV of 4300 h−1. At this point, the propylene yield reached 27.7% and the corresponding propylene selectivity was up to 93.8%.-
Key words:
- Co/HZSM-5 catalyst /
- response surface /
- propane dehydrogenation /
- process conditions
-
Figure 1 Schematic illustration of catalyst performance evaluation for propane dehydrogenation
1: Pressure reducing valves; 2: Flow meters; 3: Stop valves; 4: Mixer; 5: Quartz reactor; 6: Resistance furnaces; 7: Temperature controllers; 8: Catalyst bed; 9: Gas chromatography model SP-3420A; 10: Online analysis computers
Table 2 Influence of reaction temperature on propane dehydrogenation performance
Reaction temperature/°C Selectivity /% Propane conversion /% Propylene yield /% CH4 C2H6 C2H4 C3H6 350 6.184 0.762 8.772 84.282 8.566 7.220 400 5.978 0.555 8.325 85.142 14.415 12.273 450 2.243 0.301 7.565 89.891 24.821 22.312 500 4.171 0.456 8.011 87.362 21.736 18.989 550 18.275 2.642 22.376 56.707 19.833 11.247 GHSV=4500 h−1, Co loading= 2.0% Table 1 Pore structure properties of HA and HZ-x catalysts
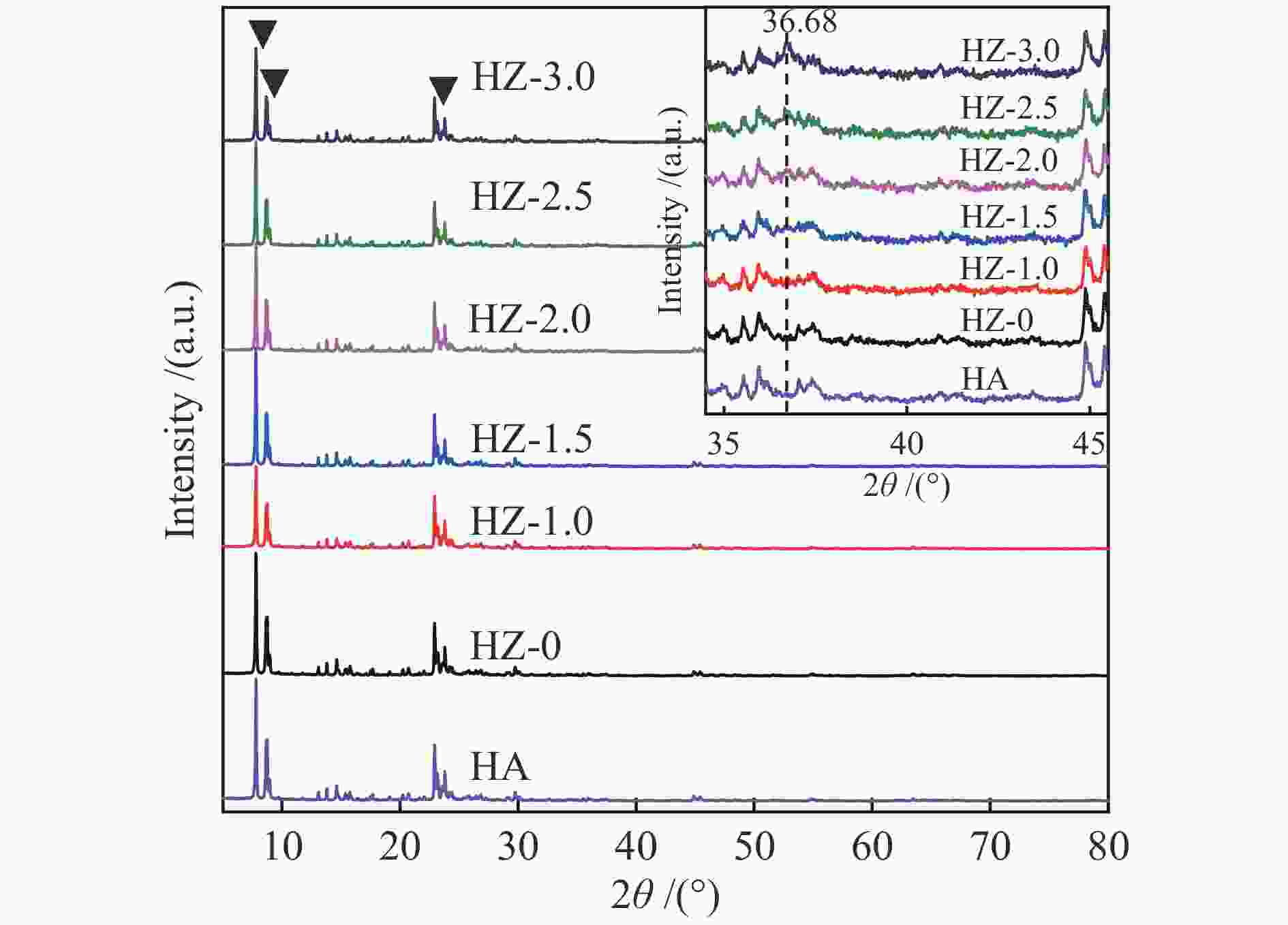
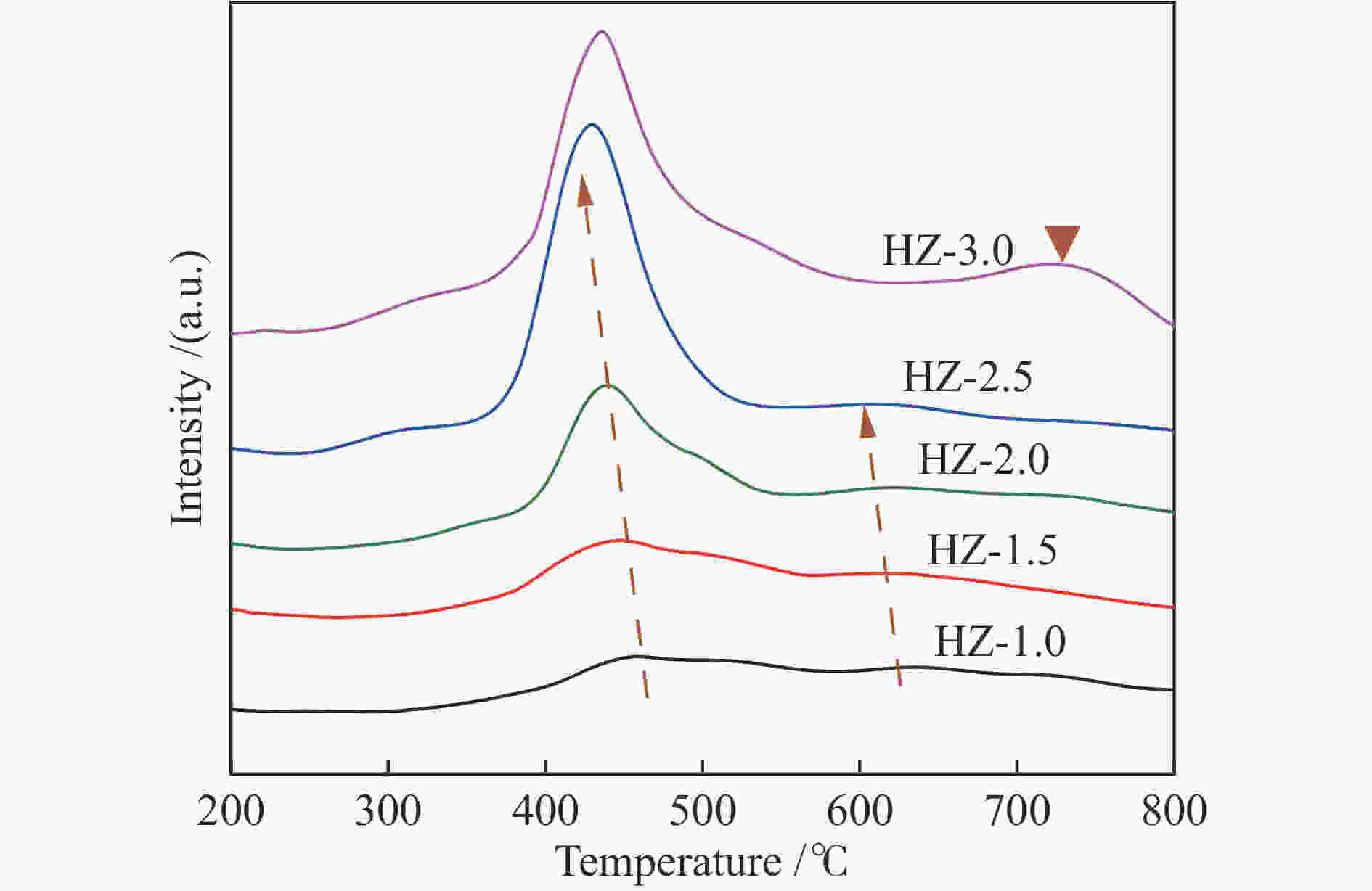
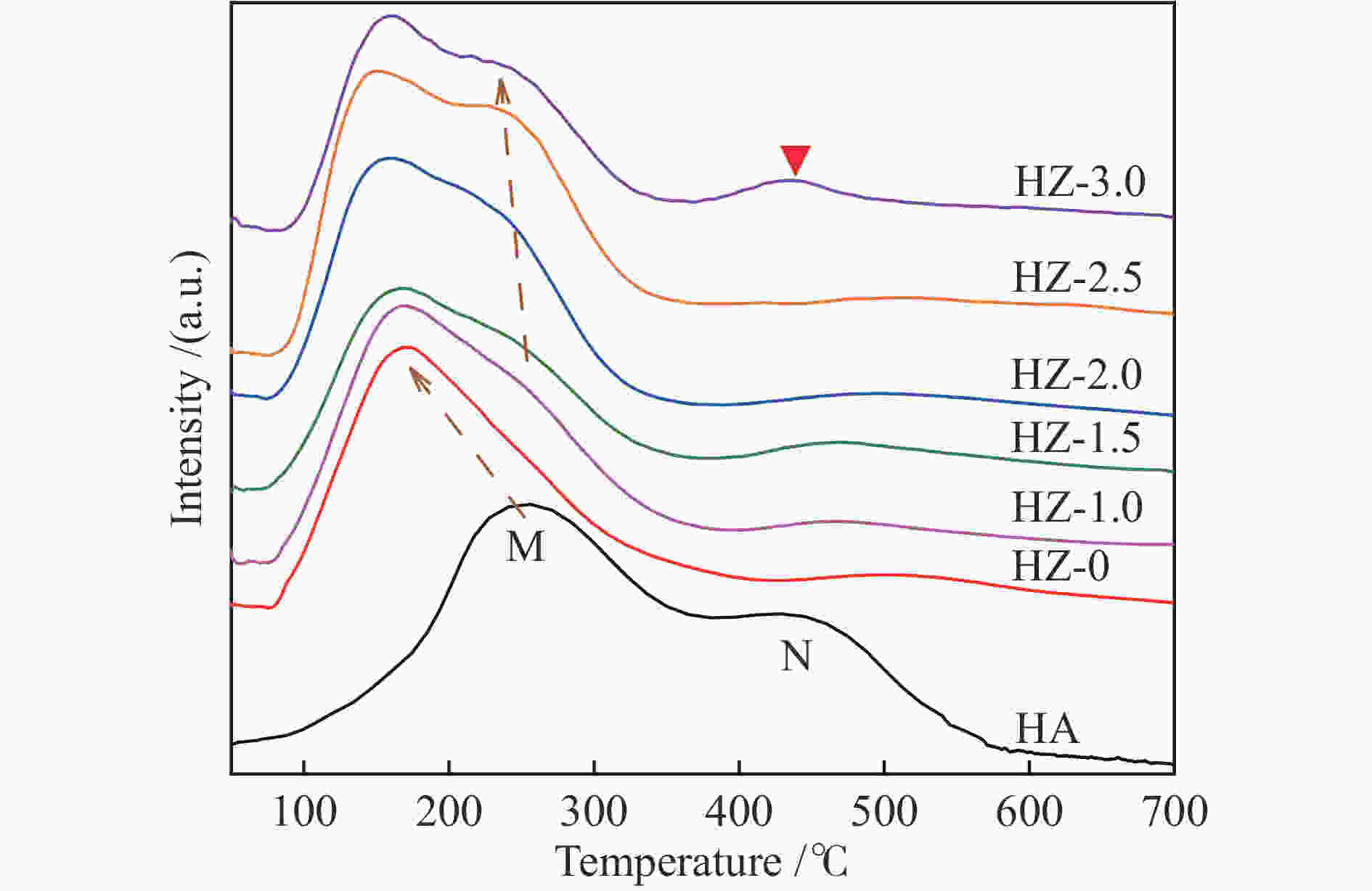
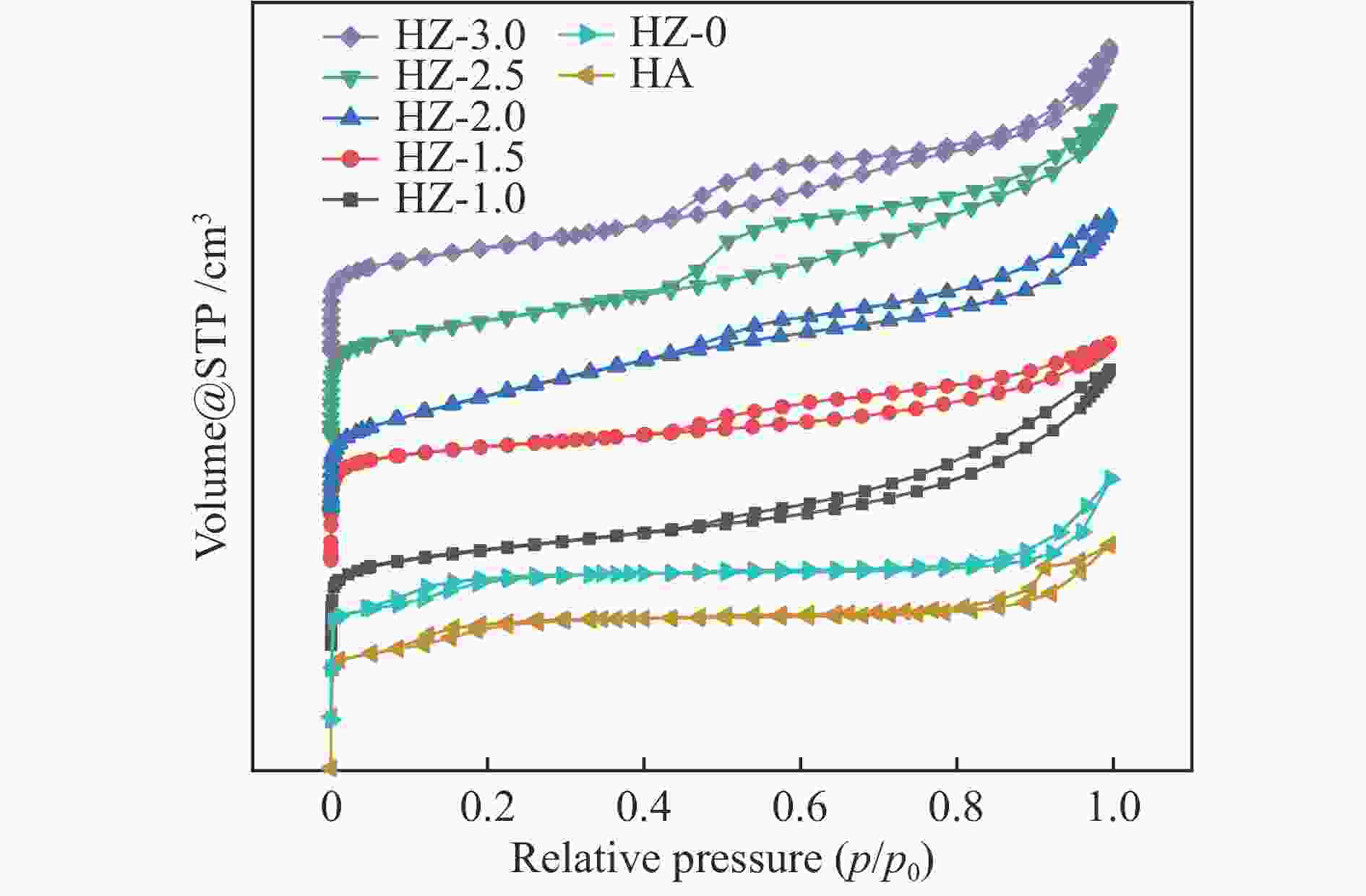
Catalyst SSA/(m2·g−1) SSAmicro/(m2·g−1) Dp/nm Pore volume /(cm3·g−1) Vmicro Vext Vtotal HA 260.372 215.339 3.977 0.1200 0.0613 0.1813 HZ-0 266.881 218.708 4.997 0.1207 0.0675 0.1882 HZ-1.0 163.960 57.719 6.358 0.0249 0.1930 0.2179 HZ-1.5 182.639 105.009 5.389 0.0514 0.1231 0.1745 HZ-2.0 195.881 60.881 4.780 0.0096 0.2142 0.2238 HZ-2.5 192.717 41.643 5.567 0.0203 0.2395 0.2598 HZ-3.0 175.157 40.827 5.664 0.0199 0.2207 0.2406 Table 3 Influence of GHSV on propane dehydrogenation performance
GHSV/h−1 Selectivity /% Propane conversion /% Propylene yield /% CH4 C2H6 C2H4 C3H6 1500 8.324 0.703 12.306 78.667 13.316 10.475 3000 7.865 0.679 10.113 81.343 17.411 14.163 4500 2.243 0.301 7.565 89.891 24.821 22.312 6000 2.198 0.292 7.477 90.033 19.725 17.759 7500 2.036 0.228 6.939 90.797 10.724 9.737 450 °C, Co loading= 2.0% Table 4 Influence of Co loading on propane dehydrogenation performance
Co loading/% Selectivity /% Propane conversion /% Propylene yield /% CH4 C2H6 C2H4 C3H6 1.0 11.039 1.014 17.559 70.388 12.654 8.907 1.5 9.759 0.762 16.426 73.053 14.953 10.924 2.0 2.243 0.301 7.565 89.891 24.821 22.312 2.5 1.438 0.120 5.427 93.015 26.532 24.679 3.0 2.202 0.269 7.513 90.016 23.532 21.183 450 °C, GHSV=4500 h−1 Table 5 Experimental design of three factors and three levels of response surface method
Factor Symbol Level −1 0 1 Reaction temperature /°C A 400 450 500 GHSV/h−1 B 3000 4500 6000 Co loading /% C 2.0 2.5 3.0 Table 6 Box-Behnken response surface experimental design and results
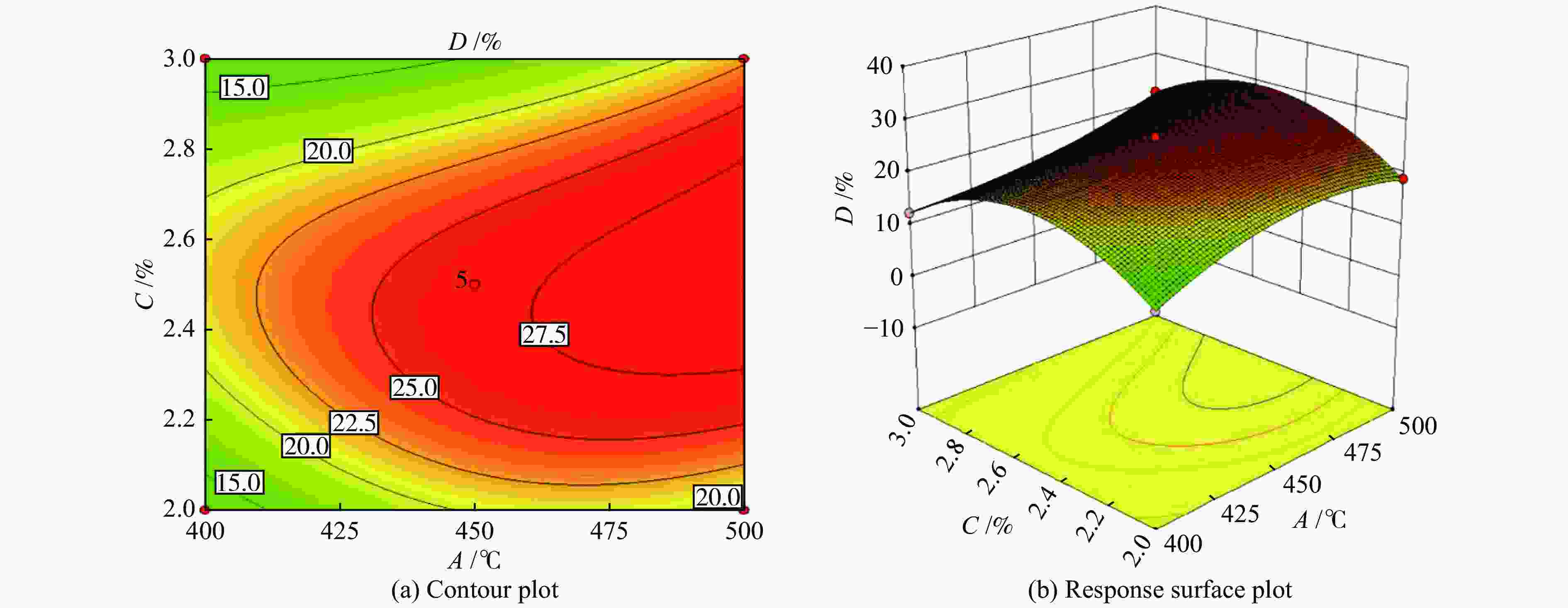
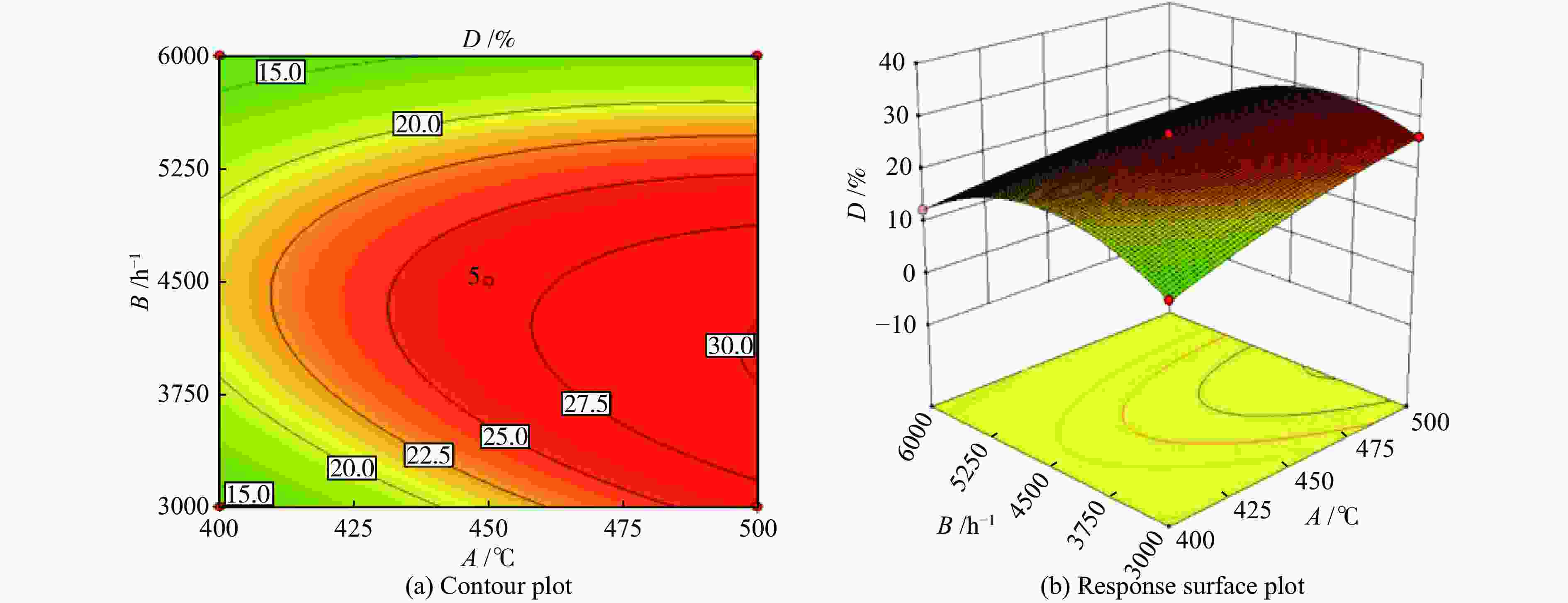
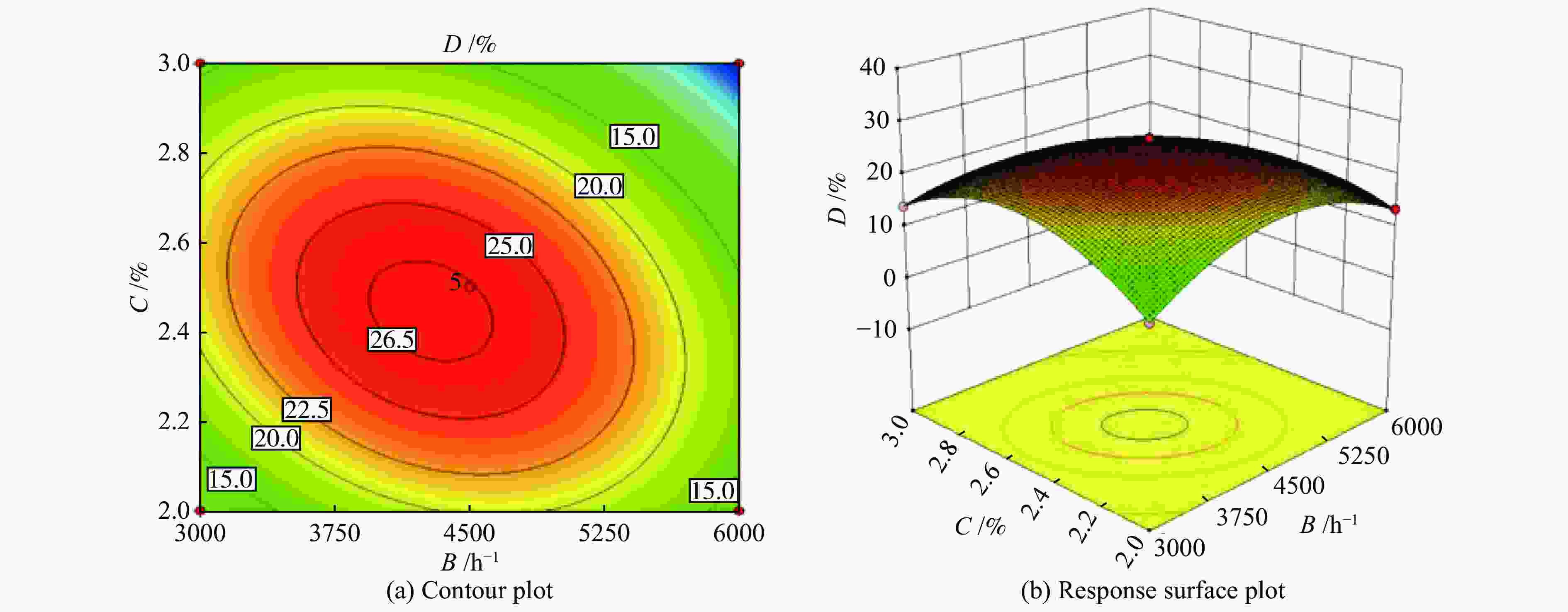
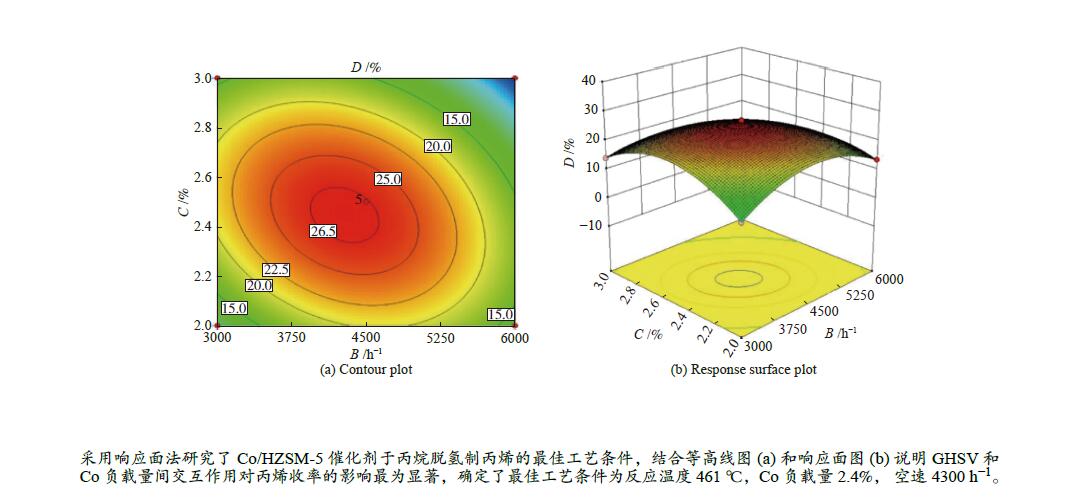
Number A/°C B/h−1 C/% Yield of propylene (D)/% Actual error/% experiment value actual equation prediction 1 400 3000 2.5 13.71 13.51 1.48 2 500 3000 2.5 26.31 26.29 0.06 3 400 6000 2.5 12.53 12.55 −0.14 4 500 6000 2.5 15.66 15.86 −1.30 5 400 4500 2.0 12.28 12.37 −0.77 6 500 4500 2.0 19.04 18.95 0.49 7 400 4500 3.0 12.48 12.57 −0.73 8 500 4500 3.0 22.18 22.09 0.42 9 450 3000 2.0 10.45 10.56 −1.02 10 450 6000 2.0 13.51 13.40 0.81 11 450 3000 3.0 14.07 14.18 −0.77 12 450 6000 3.0 0.054 0.055 −1.85 13 450 4500 2.5 26.97 26.58 1.43 14 450 4500 2.5 26.29 26.58 −1.09 15 450 4500 2.5 27.06 26.58 1.79 16 450 4500 2.5 27.88 26.58 1.13 17 450 4500 2.5 26.67 26.58 −3.54 Table 7 Analysis of variance (ANOVA)
Source Sum of squares df Mean square F-Value P-Value Significance Model 986.68 10 98.67 382.36 < 0.0001 significant A 129.53 1 129.53 501.96 < 0.0001 B 64.93 1 64.93 251.63 < 0.0001 C 24.19 1 24.19 93.75 < 0.0001 AB 22.42 1 22.42 86.86 < 0.0001 AC 2.16 1 2.16 8.35 0.0277 BC 72.89 1 72.89 282.46 < 0.0001 A2 6.85 1 6.85 26.53 0.0021 B2 286.43 1 286.43 1109.96 < 0.0001 C2 326.44 1 326.44 1265.01 < 0.0001 A2C 21.72 1 21.72 84.16 < 0.0001 Residual 1.55 6 0.26 Lack of fit 0.17 2 0.08 0.24 0.7977 not significant Pure error 1.38 4 0.35 Cor total 988.23 16 R2=0.9984 CV(%)=2.83 Table 8 Experimental and predicted values of optimal process parameters
Number Experimental conditions Experiment value/% Actual equation prediction/% Actual error/% temperature/°C GHSV/h−1 Co loading/% 1 461 4300 2.4 27.719 27.666 0.19 2 461 4300 2.4 27.598 27.666 −0.25 3 461 4300 2.4 27.689 27.666 0.08 -
[1] SPEIGHT J. G. Ullmann's encyclopedia of industrial chemistry[J]. Petrol Sci Technol,1999,17(3/4):445−445. doi: 10.1080/10916469908949727 [2] ZHAO D, YUAN M, ZHANG Y, JIANG G, SUN H. Incorporation of Cr in ZSM-5 zeolite framework as bifunctional catalysts for n-butane catalytic cracking[J]. CIESC J,2016,67(08):3400−3407. [3] XU Z, YUE Y, BAO X, XIE Z, ZHU H. Propane dehydrogenation over Pt clusters localized at the Sn single-site in zeolite framework[J]. ACS Catal,2020,10:818−828. doi: 10.1021/acscatal.9b03527 [4] FRICKE C, RAJBANSHI B, WALKER E A, TEREJANU G, HEYDEN A. Propane dehydrogenation on platinum catalysts: Identifying the active sites through Bayesian analysis[J]. ACS Catal,2022,12(4):2487−2498. doi: 10.1021/acscatal.1c04844 [5] HU Z P, WANG Y, YANG D, YUAN Z Y. CrO supported on high-silica HZSM-5 for propane dehydrogenation[J]. J Energy Chem,2020,47:225−233. doi: 10.1016/j.jechem.2019.12.010 [6] LIOTTA L F, PANTALEO G, MACALUSO A, CARLO G D, DEGANELLO G. CoOx catalysts supported on alumina and alumina-baria: influence of the support on the cobalt species and their activity in NO reduction by C3H6 in lean conditions[J]. Appl Catal A: Gen,2003,245(1):167−177. doi: 10.1016/S0926-860X(02)00652-X [7] ZHANG Y, ZHOU Y, SHI J, ZHOU S, SHENG X, ZHANG Z, XIANG S. Comparative study of bimetallic Pt-Sn catalysts supported on different supports for propane dehydrogenation[J]. J Mol Catal A: Chem,2014,381:138−147. doi: 10.1016/j.molcata.2013.10.007 [8] DEWANGAN N, ASHOK J, SETHIA M, DAS S, PATI S, KUS H, KAWI S. Cobalt‐Based catalyst supported on different morphologies of alumina for non‐oxidative propane dehydrogenation: Effect of metal support interaction and lewis acidic sites[J]. ChemCatChem,2019,11(19):4923−4934. doi: 10.1002/cctc.201900924 [9] XU Y, YU W, ZHANG H, XIN J, HE X, LIU B, JIANG F, LIU X. Suppressing C–C bond dissociation for efficient ethane dehydrogenation over the isolated Co(II) sites in SAPO-34[J]. ACS Catal,2021,13001−13019. [10] LEI, LIU, QING-FANG, DENG, BAO, AGULA, XU, ZHAO, TIE-ZHEN, REN. Ordered mesoporous carbon catalyst for dehydrogenation of propane to propylene[J]. Chem Commun,2011,47(29):8334−8336. doi: 10.1039/c1cc12806j [11] LIU L, DENG Q F, BAO A, REN T Z, LIU Y P, BAO Z, YUAN Z Y. Synthesis of ordered mesoporous carbon materials and their catalytic performance in dehydrogenation of propane to propylene[J]. Catal Today,2012,186(1):35−41. doi: 10.1016/j.cattod.2011.08.022 [12] LIU L, DENG Q F, LIU Y P, REN T Z, YUAN Z Y. HNO3-activated mesoporous carbon catalyst for direct dehydrogenation of propane to propylene[J]. Catal Commun,2011,16(1):81−85. doi: 10.1016/j.catcom.2011.09.005 [13] CAO T, DAI X, LI F, LIU W, BAI Y, FU Y, QI W. Efficient non-precious metal catalyst for propane dehydrogenation: Atomically dispersed cobalt-nitrogen compounds on carbon nanotubes[J]. ChemCatChem,2021,13(13):3067−3073. doi: 10.1002/cctc.202100410 [14] WANG Q, XU W, MA Z, YU F, CHEN Y, LIAO H, WANG X, ZHOU J. Highly effective direct dehydrogenation of propane to propylene by microwave catalysis at low temperature over Co-Sn/NC microwave catalyst[J]. ChemCatChem,2020,13(3):1009−1022. [15] ZHANG Y, ZHOU Y, QIU A, YU W, YI X, WU P. Propane dehydrogenation on PtSn/ZSM-5 catalyst: Effect of tin as a promoter[J]. Catal Commun,2006,7(11):860−866. doi: 10.1016/j.catcom.2006.03.016 [16] ZHANG Y, ZHOU Y, HUANG L, ZHOU S, SHENG X, WANG Q, ZHANG C. Structure and catalytic properties of the Zn-modified ZSM-5 supported platinum catalyst for propane dehydrogenation[J]. Chem Eng J,2015,270:352−361. doi: 10.1016/j.cej.2015.01.008 [17] GONG J J, LI S C, ZHOU H L, XU B L, FAN Y N. Promotional effect of Nain the supported PtSnNa/SUZ-4 catalysts for propane dehydrogenation[J]. J Fuel Chem Technol,2015,43(7):857−861. [18] HUANG C, HAN D, GUAN L, ZHU L, MEI Y, HE D, ZU Y. Bimetallic Ni-Zn site anchored in siliceous zeolite framework for synergistically boosting propane dehydrogenation[J]. Fuel,2022,307:121790. doi: 10.1016/j.fuel.2021.121790 [19] GUO-HAO X U, JIN-PENG Y U, HUA-SHENG X U, CHUN-CHENG L I, WANG P F. Catalystic performance of HZSM-5 zeolite treated by CH3COONa[J]. J Inorg Mater,2019,34(5):546. doi: 10.15541/jim20180336 [20] YANG S H, CUI Y D, CHEN X J, TU X. Development of synthesis and surface modification of ZSM-5 zeolite molecular sieves[J]. Adv Fine Fetrochem,2003,(4):47−50. [21] TIAN H F, HE H H, LIAO J K, ZHA F, GUO X J, TANG X H. Catalytic performance of different zeolites for propane and CO2 coupling to propylene[J]. J Fuel Chem Technol,2021,49(4):495−503. doi: 10.1016/S1872-5813(21)60029-4 [22] OKUHARA S T. Change in pore structure of MFI zeolite by treatment with NaOH aqueous solution[J]. Microporous Mesoporous Mater,2001,43(1):83−89. doi: 10.1016/S1387-1811(00)00349-8 [23] LAPIDUS A, KRYLOVA A, KAZANSKII V. Hydrocarbon synthesis from carbon monoxide and hydrogen on impregnated cobalt catalysts Part I. Physico-chemical properties of 10% cobalt/alumina and 10% cobalt/silica[J]. Appl Catal,1991,73(1):849−850. [24] WANG W J, CHEN Y W. Influence of metal loading on the reducibility and hydrogenation activity of cobalt/alumina catalysts[J]. Appl Catal,1991,77(2):223−233. doi: 10.1016/0166-9834(91)80067-7 [25] ARNOLDY P, FRANKEN M C, SCHEFFER B, MOULIJN J A. Temperature-programmed reduction of CoOMoO3Al2O3 catalysts[J]. J Catal,1985,96(2):381−395. doi: 10.1016/0021-9517(85)90308-2 [26] MARTÍNEZ A, L PEZ C, M RQUEZ F, DÍAZ I. Fischer-Tropsch synthesis of hydrocarbons over mesoporous Co/SBA-15 catalysts: The influence of metal loading, cobalt precursor, and promoters[J]. J Catal,2003,220(2):486−499. doi: 10.1016/S0021-9517(03)00289-6 [27] HYMAN M P, VOHS J M. Reaction of ethanol on oxidized and metallic cobalt surfaces[J]. Surf Sci,2011,605(3/4):383−389. doi: 10.1016/j.susc.2010.11.005 [28] SUN Y, WU Y, SHAN H, LI C. Studies on the nature of active cobalt species for the production of methane and propylene in catalytic dehydrogenation of propane[J]. Catal Lett,2015,145(7):1413−1419. doi: 10.1007/s10562-015-1533-4 [29] SARTIPI S, ALBERTS M, SANTOS V P, NASALEVICH M, GASCON J, KAPTEIJN F. Insights into the catalytic performance of mesoporous H-ZSM-5-supported cobalt in Fischer-Tropsch synthesis[J]. ChemCatChem,2014,6(1):142−151. doi: 10.1002/cctc.201300635 [30] YU S Y, YU G J, LI W, IGLESIA E. Kinetics and reaction pathways for propane dehydrogenation and aromatization on Co/H-ZSM5 and H-ZSM5[J]. J Phys Chem B,2002,106(18):4714−4720. doi: 10.1021/jp013245m [31] GAJARDO P, PIROTTE D, DEFOSSE C, GRANGE P, DELMON B. XPS study of the supported phase — SiO2 interaction in Mo/SiO2 and CoMo/SiO2 hydrodesulphurization catalysts in their oxidic precursor form[J]. J Electron Spectrosc,1979,17(2):121−135. doi: 10.1016/0368-2048(79)85033-1 [32] MU S F, SHANG R J, ZHANG J L, CHEN J G. Pretreating Co/SiO2 to generate highly active Fischer-Tropsch synthesis catalyst with low CH4 selectivity[J]. J Fuel Chem Technol,2021,49(11):1592−1597. doi: 10.1016/S1872-5813(21)60163-9 [33] TAN B J, KLABUNDE K J, SHERWOOD P M A. XPS studies of solvated metal atom dispersed (SMAD) catalysts. Evidence for layered cobalt-manganese particles on alumina and silica[J]. J Am Chem Soc,1991,113(3):855−861. doi: 10.1021/ja00003a019 [34] MOSELAGE M, JIE L, ACKERMANN L. Cobalt-catalyzed C−H activation[J]. ACS Catal,2015,6(2):498−525. [35] B H B, A G, M S N. Selective propane dehydrogenation with single-site Co-II on SiO2 by a non-redox mechanism[J]. J Catal,2015,322:24−37. doi: 10.1016/j.jcat.2014.10.018 [36] YANRAO C, MAO T, ZHENG C, UNIVERSITY G. Microwave synthesis technique and properties of octadecylmethyldihydroxyethyl ammonium bromide[J]. CIESC J,2011,62(2):566−573. [37] SONG Y Q, FENG Y L, FENG L, KANG C L, ZHOU X L, XU L Y, YU G X. Effect of variations in pore structure and acidity of alkali treated ZSM-5 on the isomerization performance[J]. J Mol Catal A: Chem,2009,310(1/2):130−137. doi: 10.1016/j.molcata.2009.06.006 [38] STRA ‐EIFERT A, WAL L I, HERN NDEZ MEJ A C, WEBER L J, YOSHIDA H, ZEČEVIĆ J, JONG K P, G TTEL R. Bifunctional Co-based catalysts for fischer-tropsch synthesis: Descriptors affecting the product distribution[J]. ChemCatChem,2021,13(11):2726−2742. doi: 10.1002/cctc.202100270 [39] S C, J X, R Y Y, D W J, P Z X, X Z K. Properties of ultramicroscopic beta zeolites synthesized from mesoporous molecular sieves as silica source and their hydrocracking performance[J]. J Catal,2006,27(003):255−258. [40] KHODAKOV A Y, BECHARA R, GRIBOVAL-CONSTANT A. Fischer-Tropsch synthesis over silica supported cobalt catalysts: Mesoporous structure versus cobalt surface density[J]. Appl Catal A: Gen,2003,254(2):273−288. doi: 10.1016/S0926-860X(03)00489-7 [41] KHODAKOV A Y, GRIBOVAL-CONSTANT A, BECHARA R, ZHOLOBENKO V L. Pore size effects in Fischer Tropsch synthesis over cobalt-supported mesoporous silicas[J]. J Catal,2002,206(2):230−241. doi: 10.1006/jcat.2001.3496 [42] LI X, JIA R, LI P, ANG S. Response surface analysis for enzymatic decolorization of Congo red by manganese peroxidase[J]. J Mol Catal B: Enzym,2009,56(1):1−6. doi: 10.1016/j.molcatb.2008.03.013 [43] HILL W J, HUNTER W G. A review of response surface methodology: A literature survey[J]. Technometrics,1966,8(4):571−590. doi: 10.2307/1266632 [44] LONG Y F, TAN F X, YANG K D, GE L, LV X Y, WEN Y X. Response surface methodology to optimize the preparation of LiFePO-4/C by low-temperature carbon thermal reduction[J]. J Chem Eng High Schools,2013,(1):125−130. [45] JIANG S J, FENG X R, LI X E, JIANG S L. Optimized preparation and characterization of PAC-PDMDAAC hybrid flocculants by response surface methodology[J]. J Chem Eng,2014,(2):731−736. [46] M Z Z, L Z H. Optimization for decolorization of azo dye acid green 20 by ultrasound and H2O2 using response surface methodology[J]. J Hazard Mater,2009,172(2/3):1388−1393. doi: 10.1016/j.jhazmat.2009.07.146 -





 下载:
下载: