Theoretical study on thermal degradation mechanism of polybutylene terephthalate dimer
-
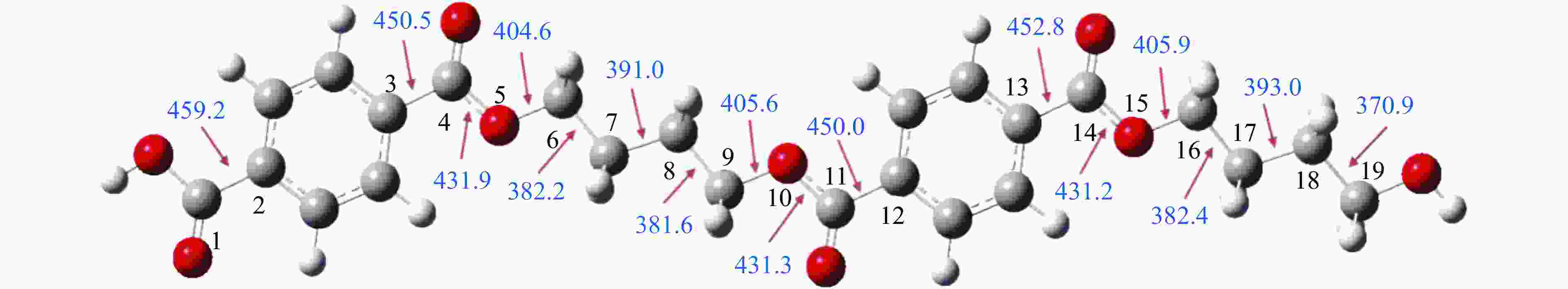
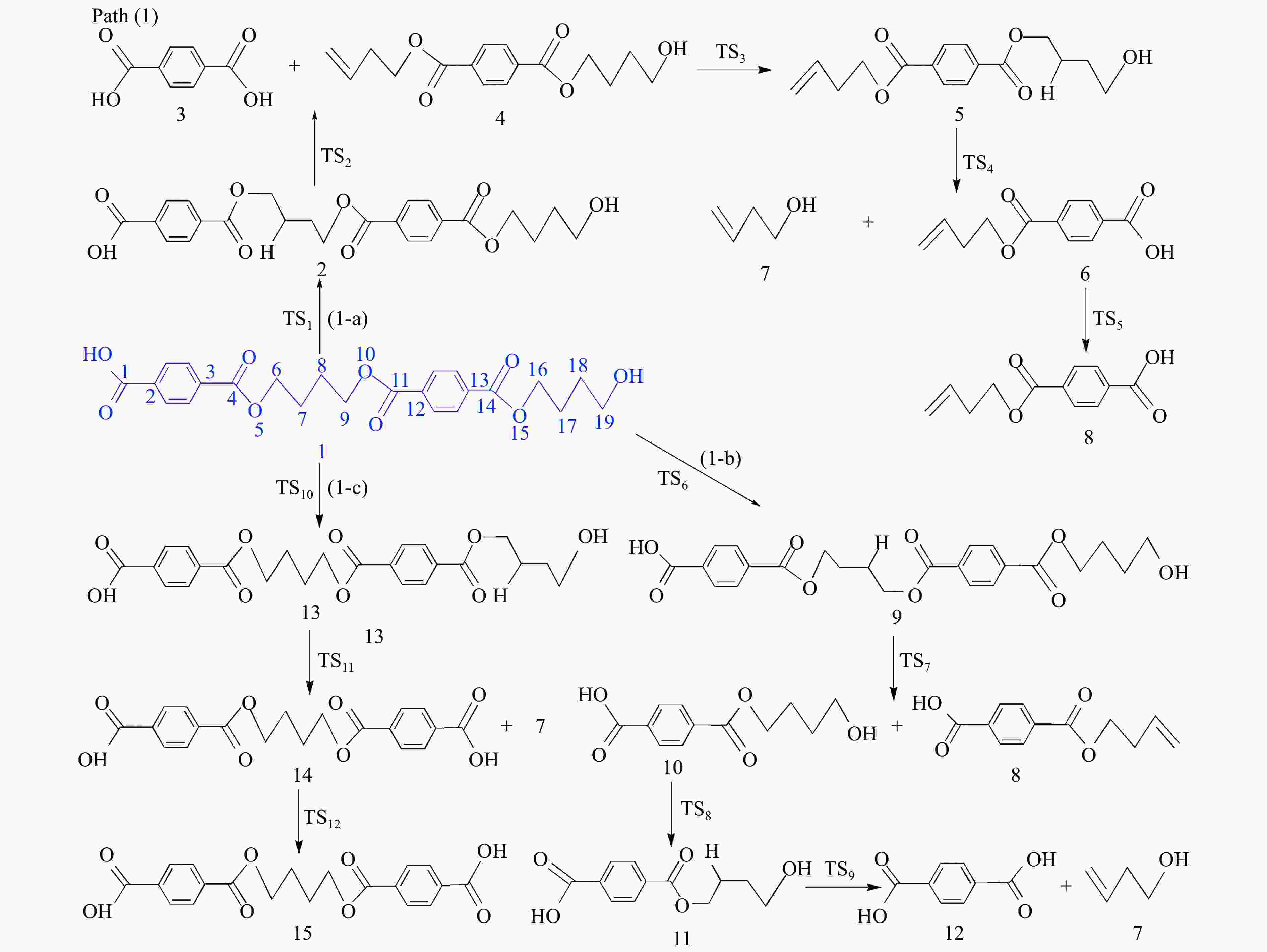
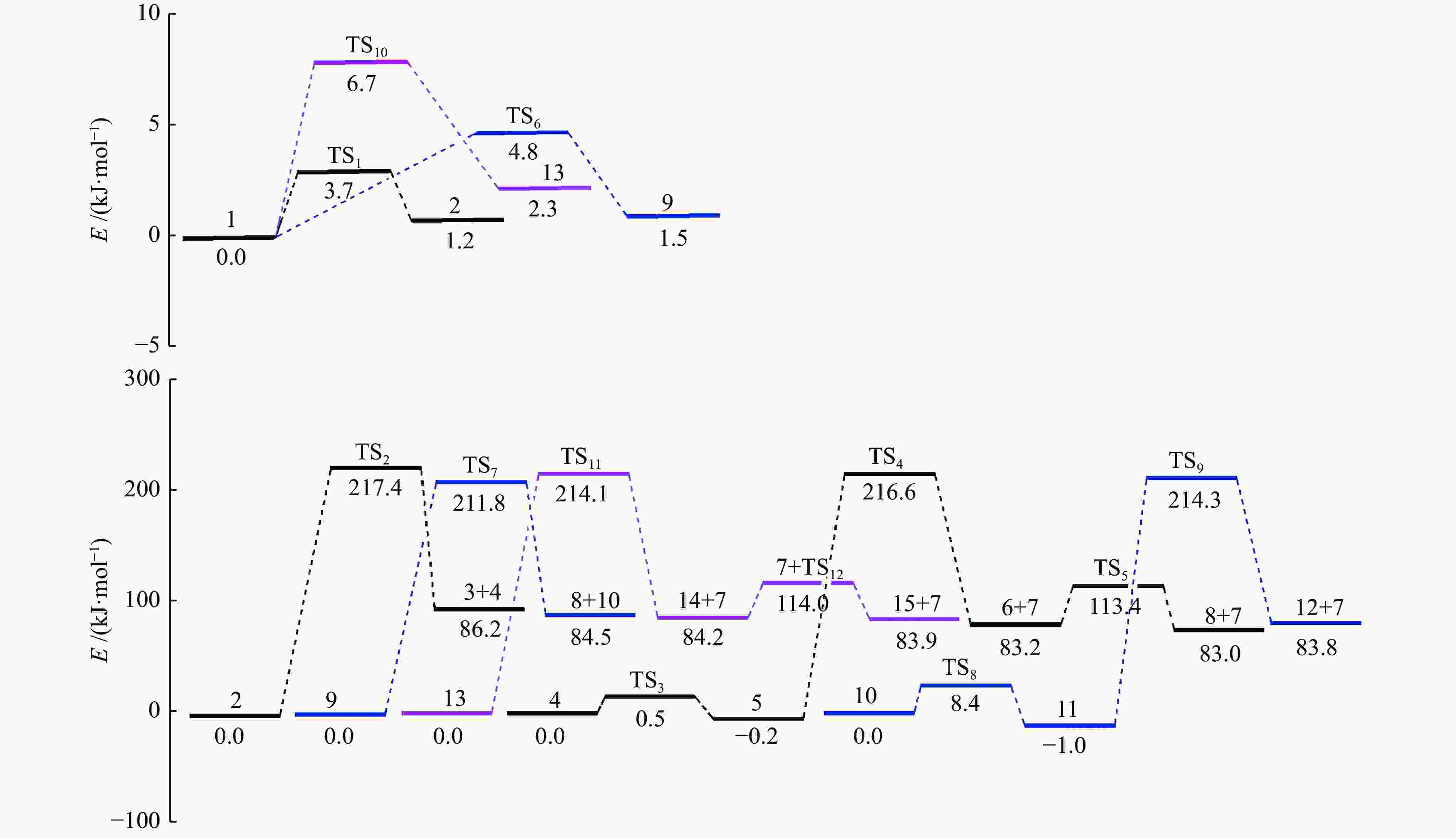
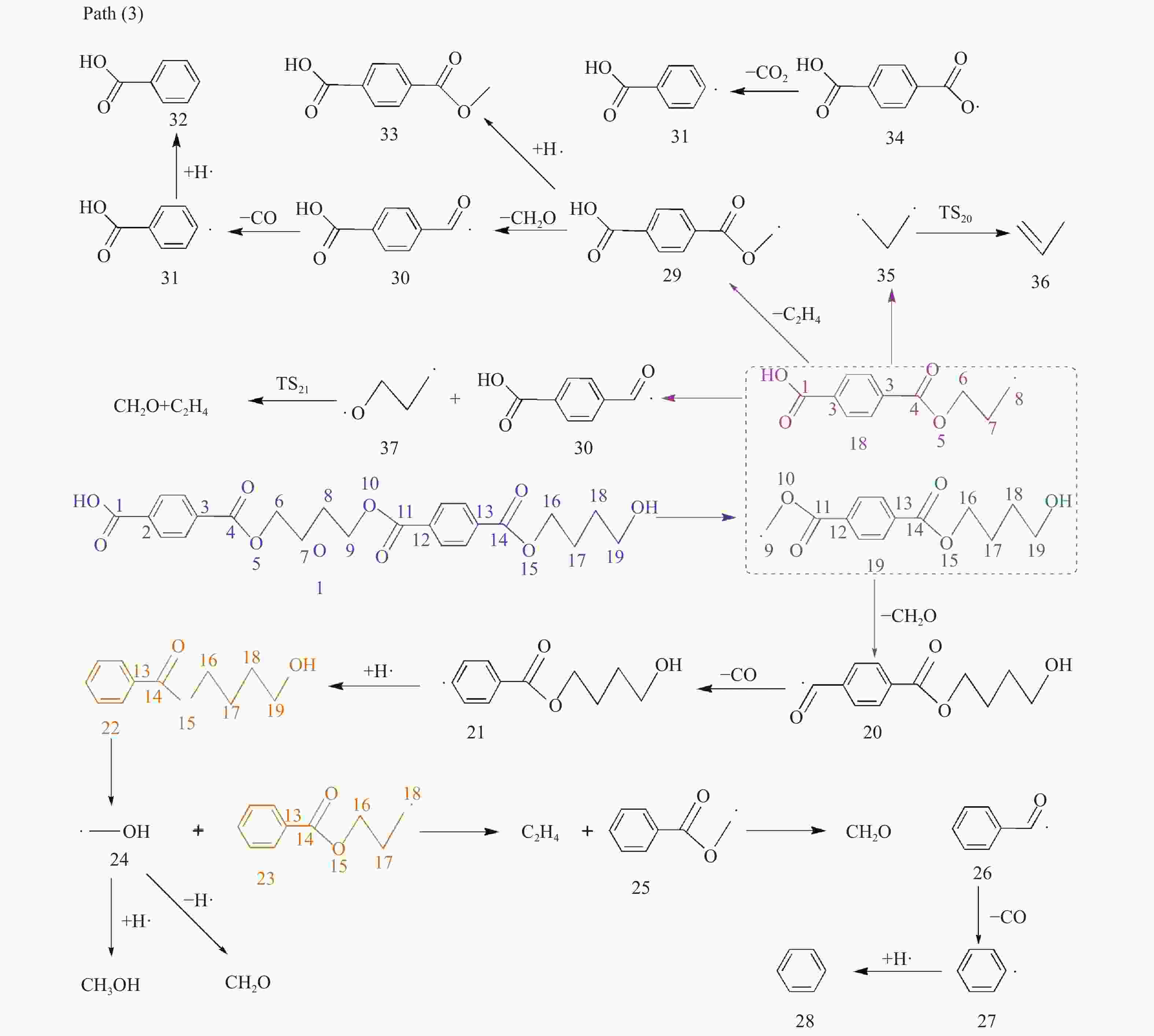
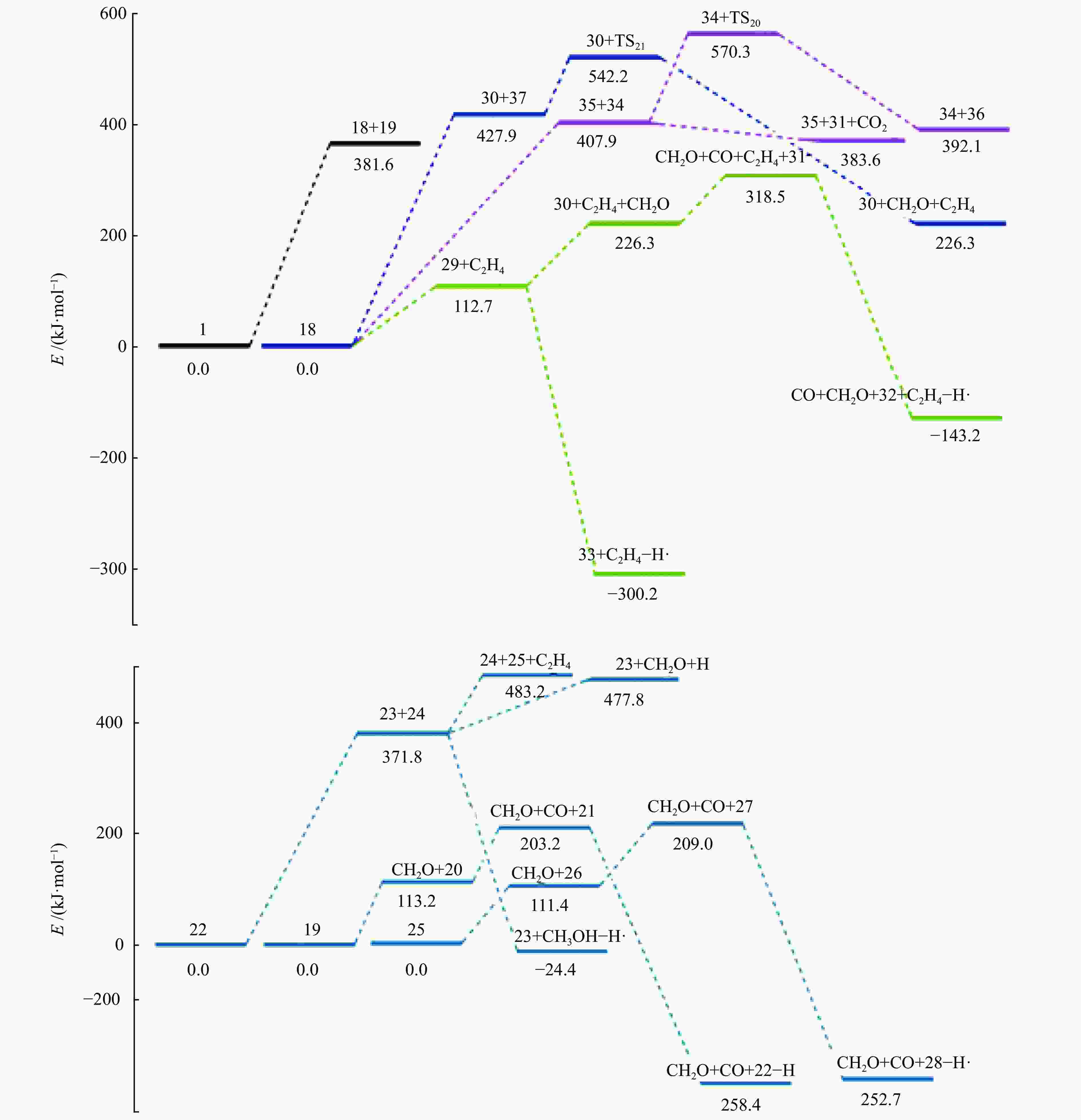
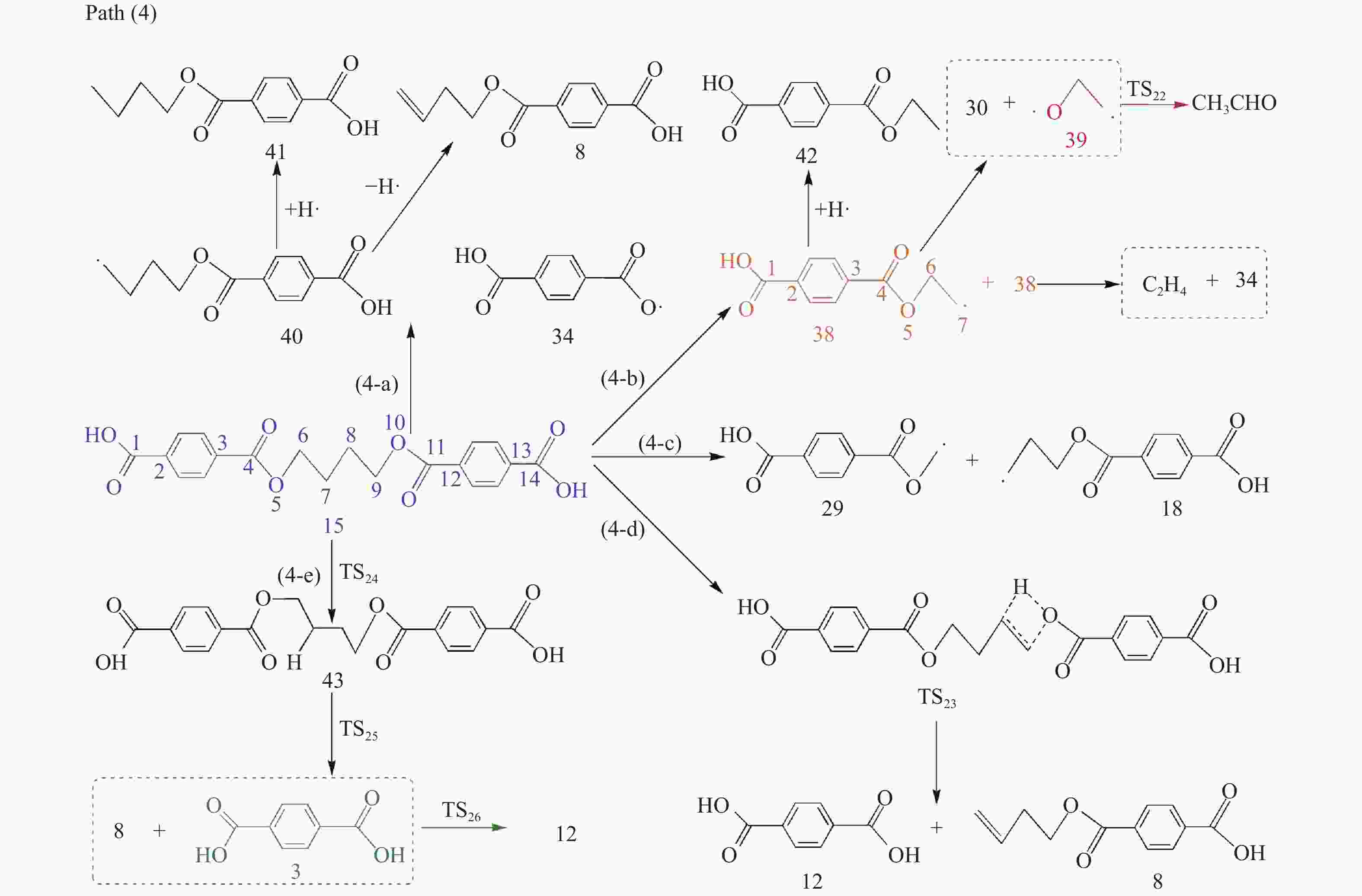
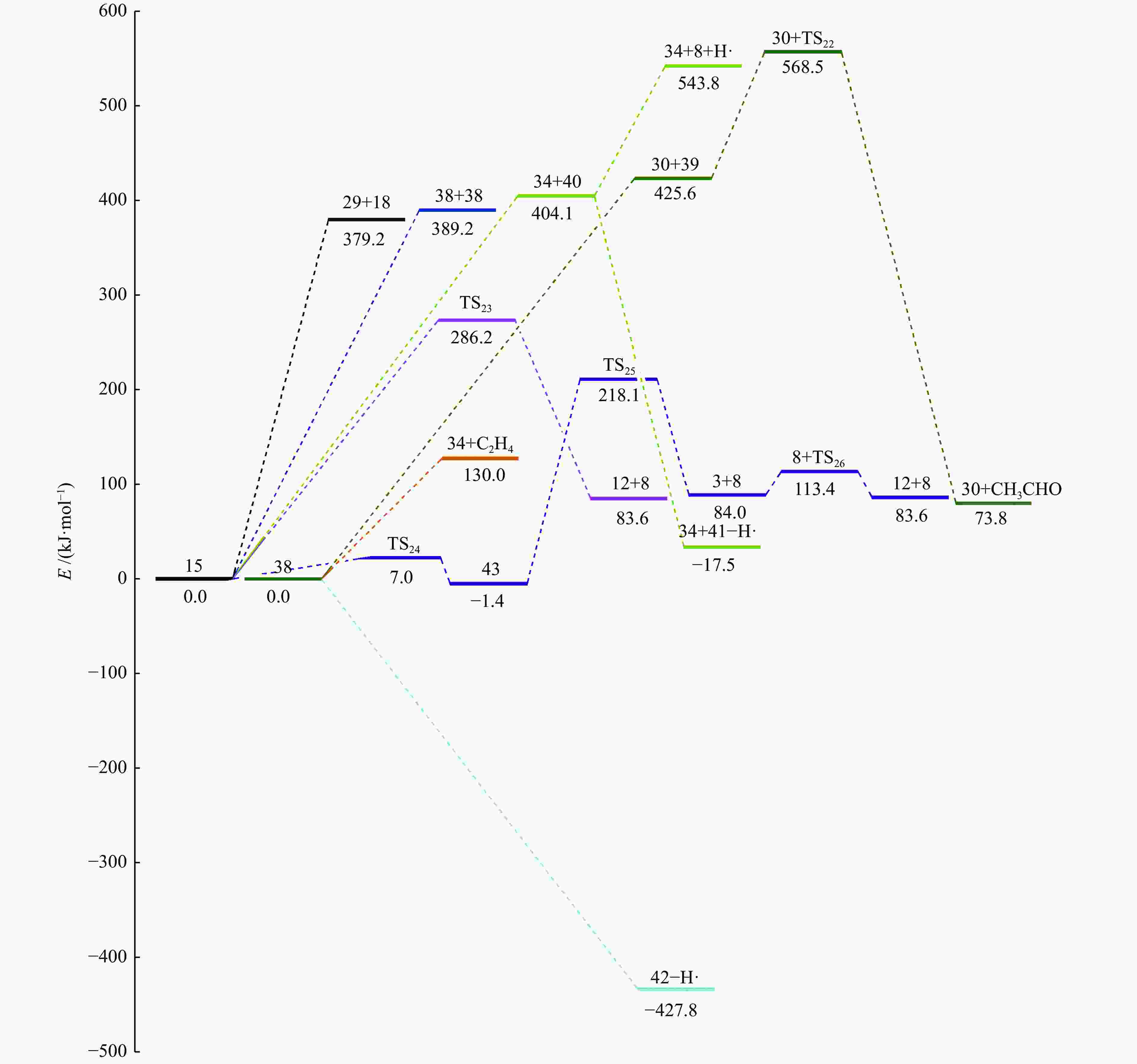
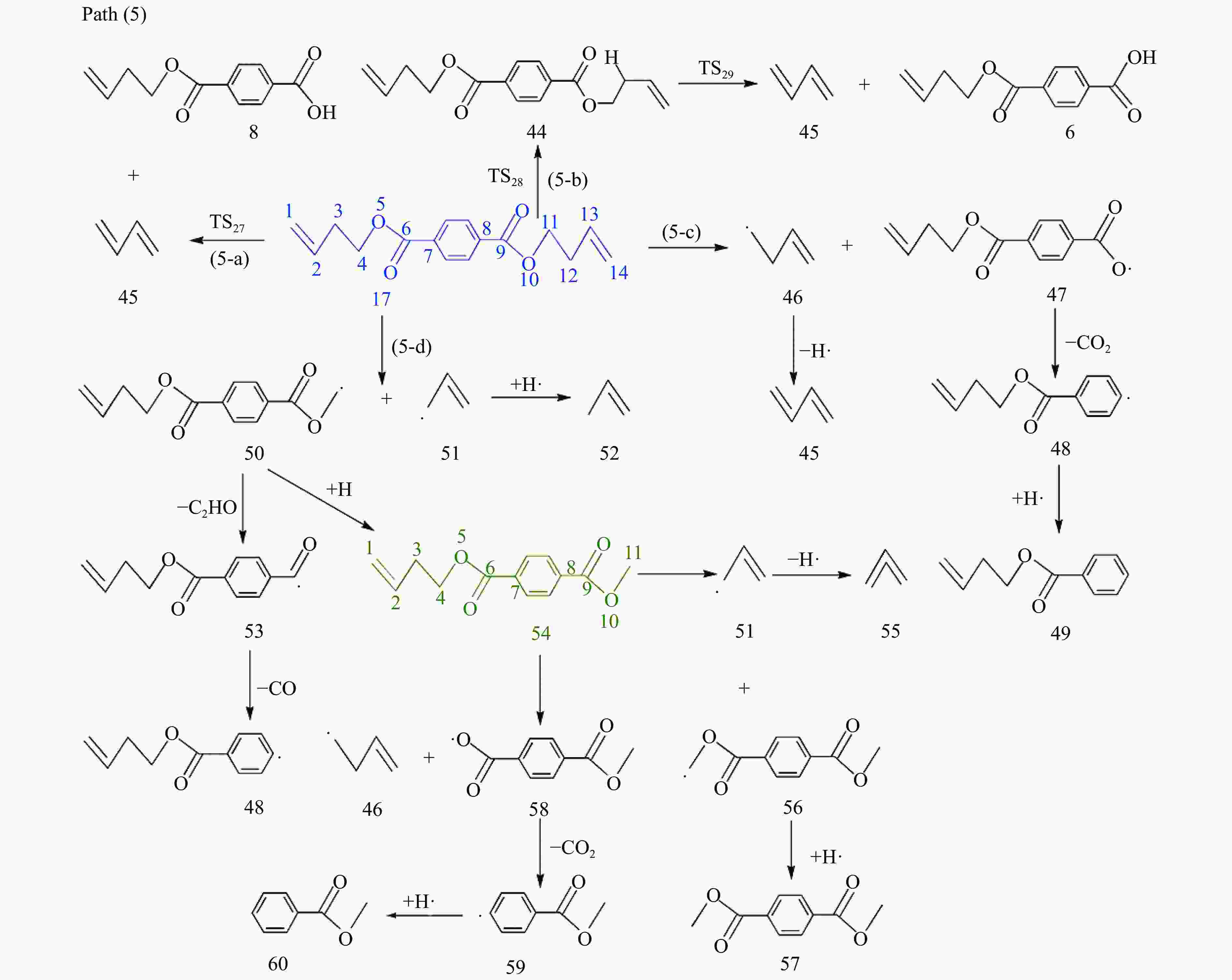
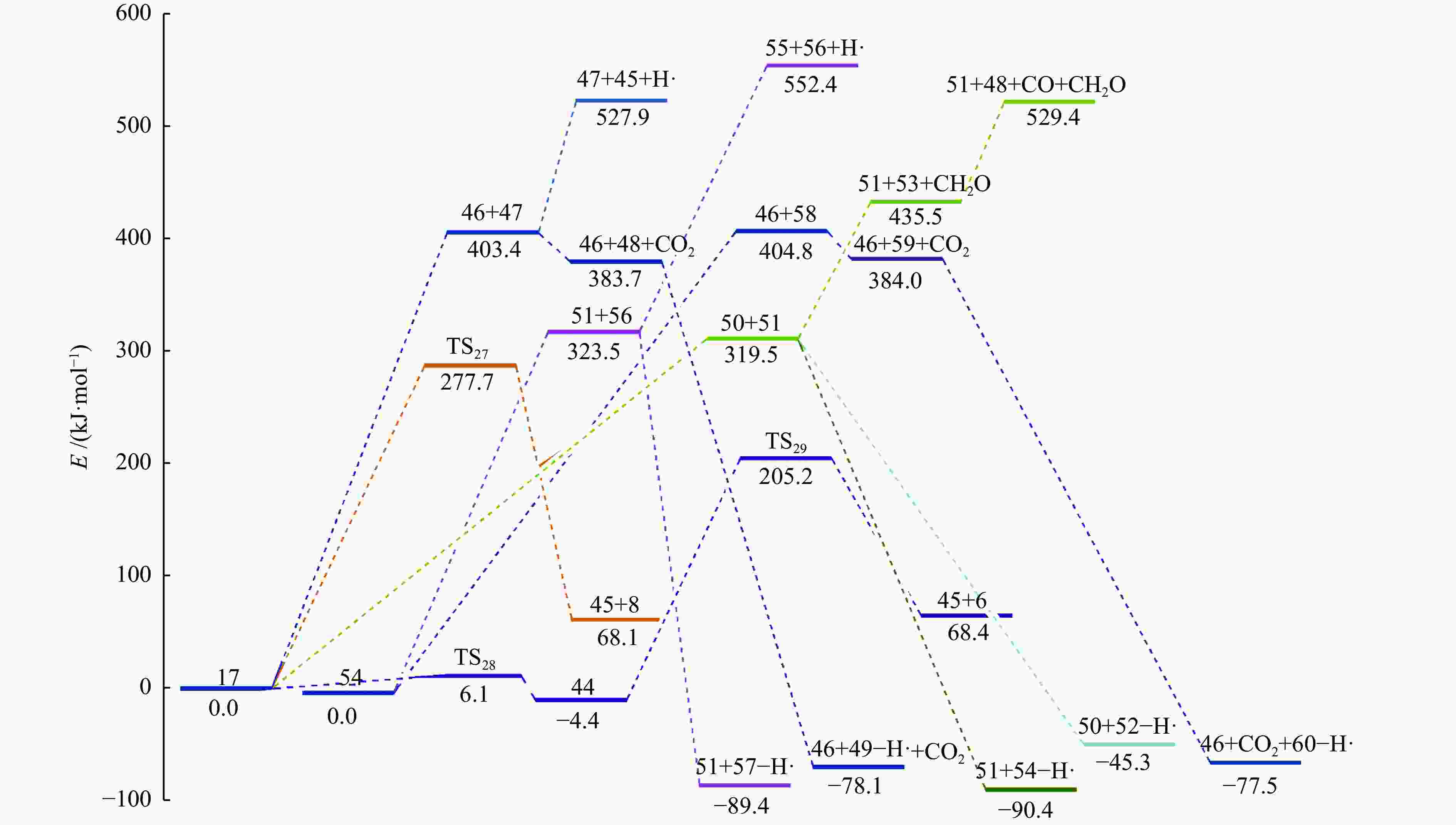
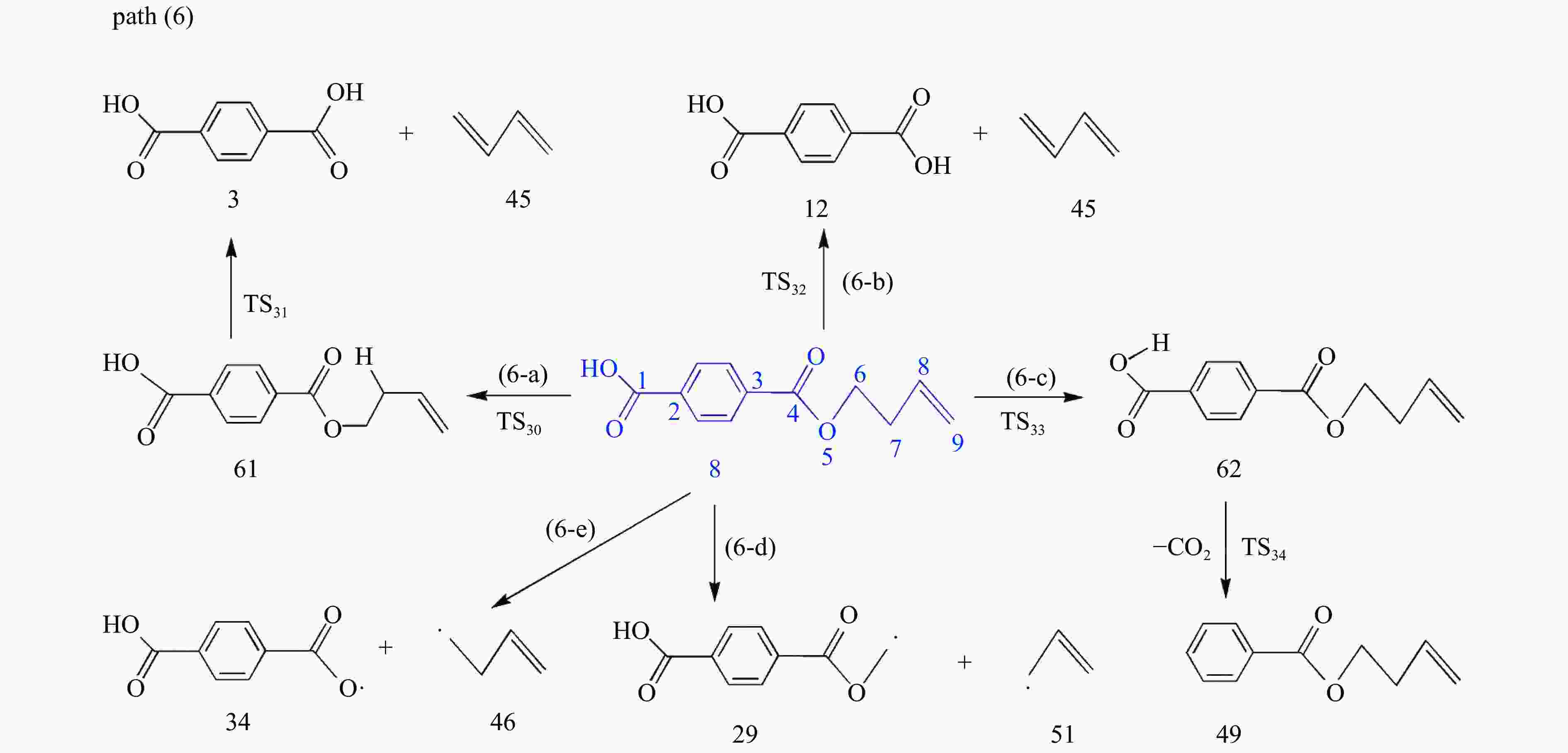
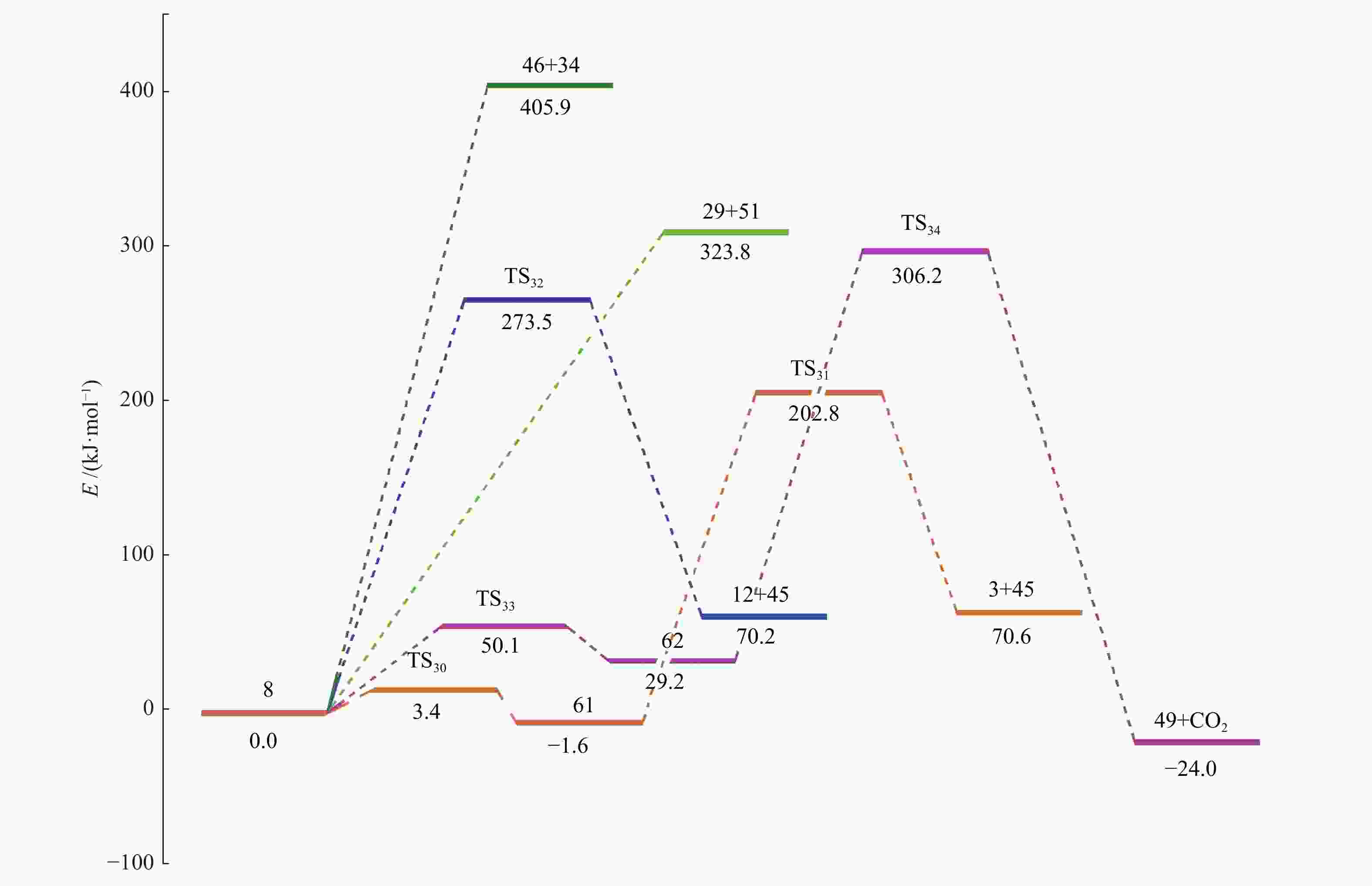
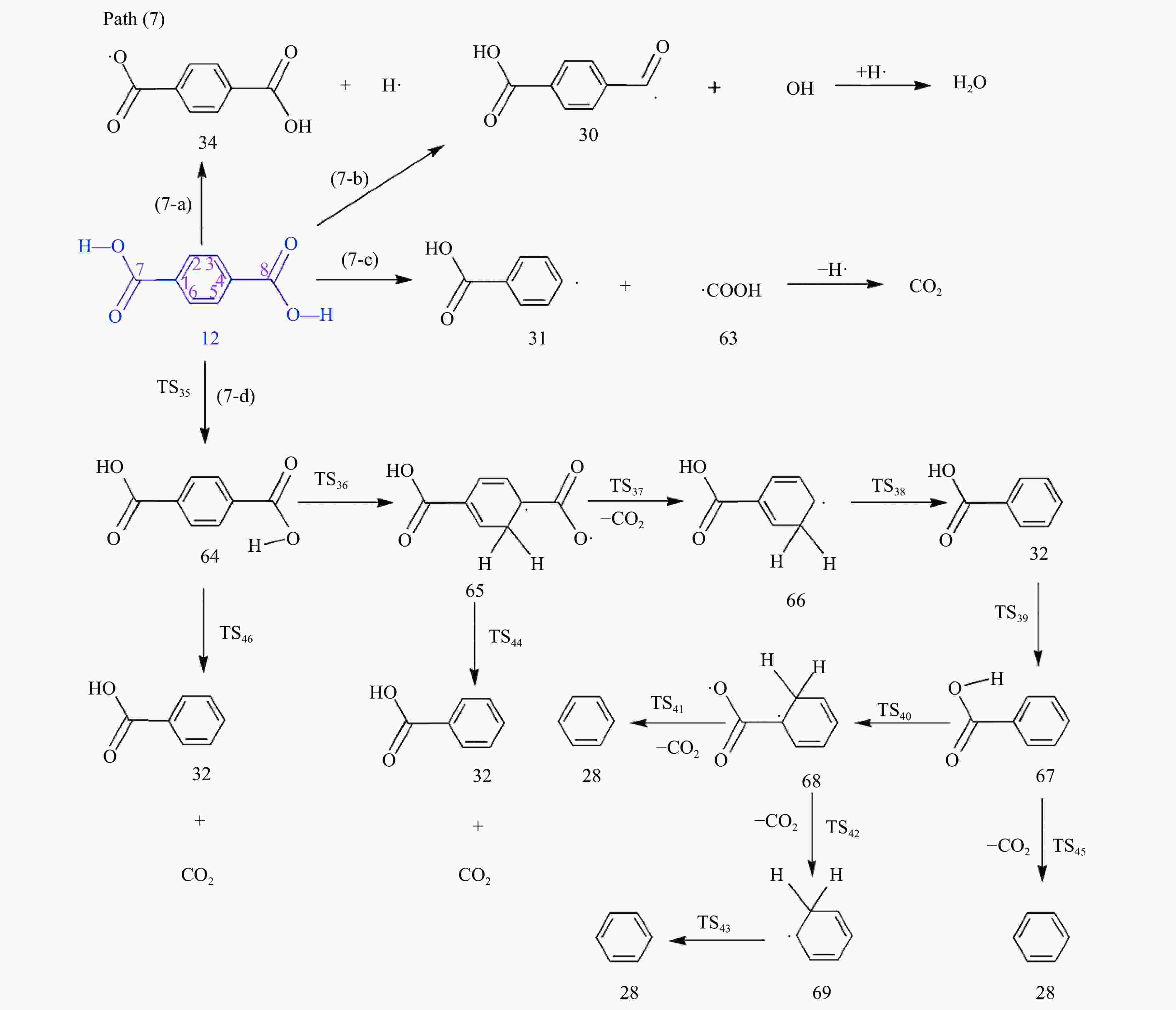
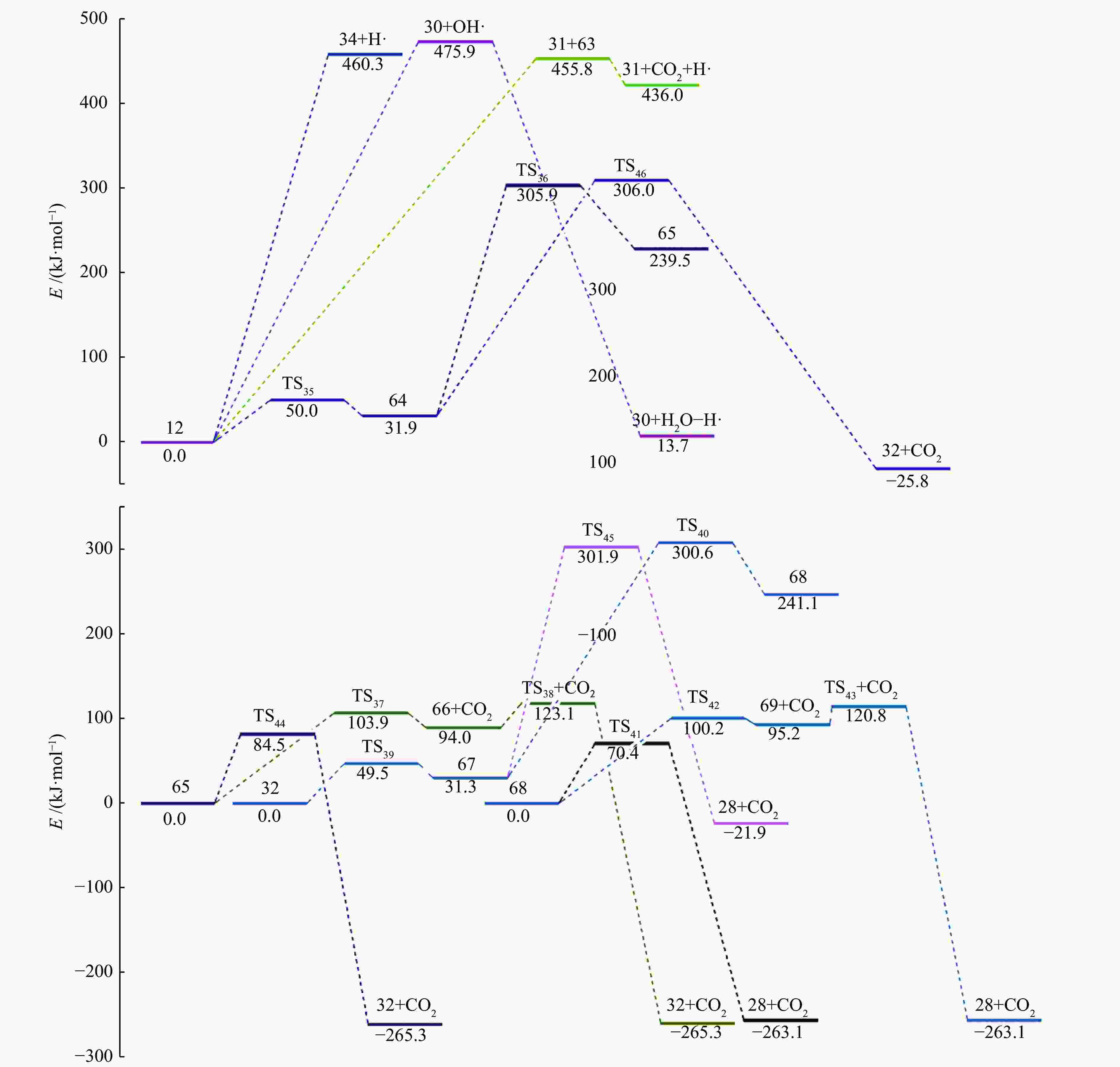
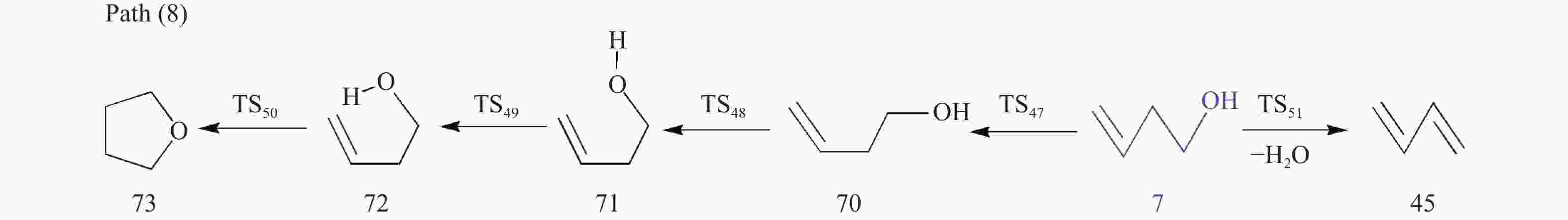
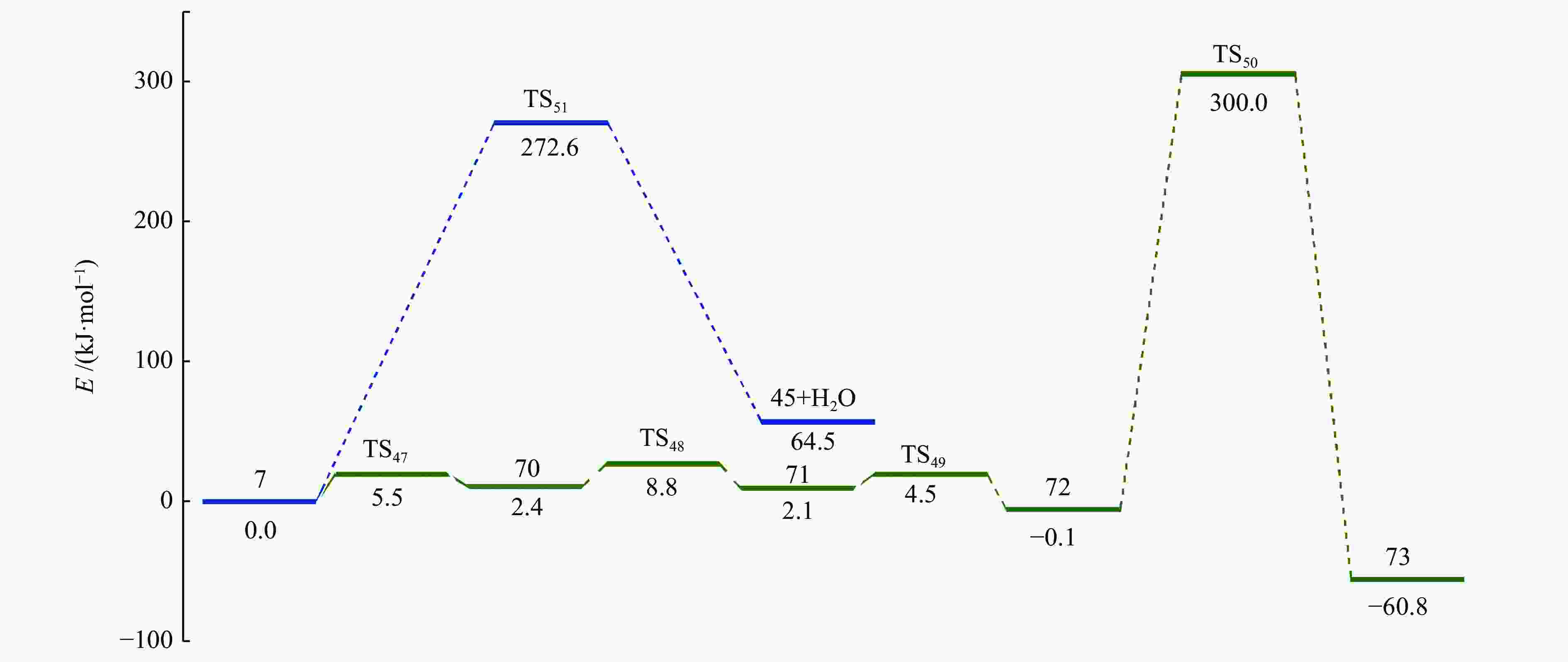
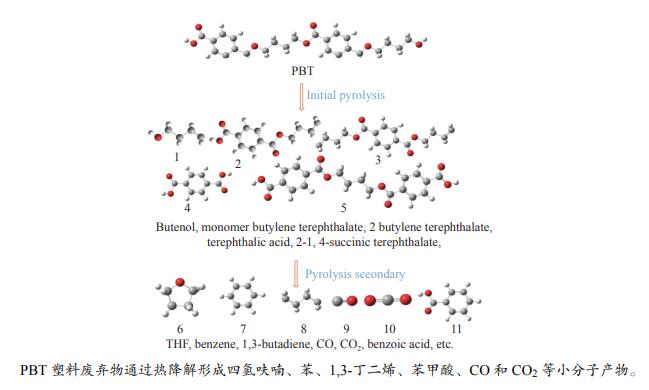
摘要: 采用密度泛函理论(DFT)方法M06-2X/6-311G(d)研究了聚对苯二甲酸丁二醇酯(PBT)二聚体的热降解机理,对PBT二聚体热解过程设计了八条可能反应路径,计算了每条反应路径的各基元反应步的热力学及动力学参数。计算结果表明,在PBT初始热解过程中,主链发生协同反应的反应能垒明显低于自由基反应的能垒,因此,通过协同反应生成的对苯二甲酸、对苯二甲酸单丁烯酯、对苯二甲酸二丁烯酯和二对苯二甲酸-1,4-丁二酯是PBT初始热解主要产物。主链通过六元环过渡态进行的协同反应的反应能垒低于通过四元环过渡态的,PBT主链的断裂主要通过六元环过渡态的协同反应而进行。此外,还讨论了PBT主要产物的二次降解反应,研究发现,在二次降解反应过程中主要以协同反应为主,生成1,3-丁二烯、四氢呋喃、苯、CO2、苯甲酸等主要产物。Abstract: Thermal degradation mechanism of polybutylene terephthalate (PBT) dimer was studied by density functional theory (DFT) method M06-2X/6-311G(d). Eight possible reaction paths were designed for the thermal decomposition of PBT dimer, and the thermodynamic and kinetic parameters of elementary reaction steps in each reaction path were calculated. Calculation results show that, in the initial pyrolysis process of PBT, the energy barrier of concerted reaction occurring on the main chain is significantly lower than that of the radical reaction, so terephthalic acid, monobutenyl terephthalate, dibutenyl terephthalate and diterephthalate-1,4-butadiester formed by concerted reaction are main products in PBT initial pyrolysis. The energy barrier of the main chain fracture through the six-membered cyclic transition state is lower than that through the four-membered cyclic transition state, and the main chain fracture of PBT is mainly through the concerted reaction with six-membered cyclic transition state. In addition, the secondary degradation reaction of the main products of PBT pyrolysis was also discussed. It is found that the main products such as 1,3-butadiene, tetrahydrofuran, benzene, CO2 and benzoic acid are mainly generated through concerted reaction in the processes of secondary degradation.
-
Key words:
- PBT dimer /
- density functional theory /
- pyrolysis /
- reaction mechanism
-
表 1 各热解路径中过渡态唯一的虚频
Table 1 Sole imaginary frequency of various transition states
Freq. i/cm−1 TS1 TS2 TS3 TS4 TS5 TS6 TS7 TS8 −89.58 −1443.48 −60.14 −1406.55 −89.01 −64.48 −1478.36 −76.92 TS9 TS10 TS11 TS12 TS13 TS14 TS15 TS16 −1426.85 −73.06 −1415.01 −86.88 −1017.01 −1135.73 −1015.16 −1908.39 TS17 TS18 TS19 TS20 TS21 TS22 TS23 TS24 −1106.42 −1909.65 −1017.91 −1859.80 −446.01 −1755.78 −1179.75 −58.67 TS25 TS26 TS27 TS28 TS29 TS30 TS31 TS32 −1461.59 −87.20 −1168.96 −65.87 −1535.27 −45.51 −1482.76 −1171.92 TS33 TS34 TS35 TS36 TS37 TS38 TS39 TS40 −516.58 −1897.99 −540.57 −859.71 −152.44 −467.65 −539.92 −885.34 TS41 TS42 TS43 TS44 TS45 TS46 TS47 TS48 −466.44 −154.37 −440.04 −608.36 −464.81 −1904.35 −125.15 −137.97 TS49 TS50 TS51 −234.88 −2034.71 −1800.56 -
[1] PASSALACQUA V, PILATI F, ZAMBONI V, FORTUNATO B, MANARESI P. Thermal degradation of poly (butylene terephthalate)[J]. Polymer,1976,17(12):1044−1048. doi: 10.1016/0032-3861(76)90004-5 [2] SABLONG R, DUCHATEAU R, KONING C E, POSPIECH D, KORWITZ A, KOMBER H, DER LANDWEHR M A. Incorporation of a flame retardancy enhancing phosphorus-containing diol into poly (butylene terephthalate) via solid state polycondensation: A comparative study[J]. Polym Degrad Stabil,2011,96(3):334−341. doi: 10.1016/j.polymdegradstab.2010.07.001 [3] NOGALES A, SANZ A, EZQUERRA T A, QUINTANA R, MUÑOZ-GUERRA S. Molecular dynamics of poly (butylene tert-butyl isophthalate) and its copolymers with poly (butylene terephthalate) as revealed by broadband dielectric spectroscopy[J]. Polymer,2006,47(20):7078−7084. doi: 10.1016/j.polymer.2006.07.044 [4] CHO M, YANG J, NOH S, JOE H, HAN M. Production of PBT (polybutylene terephthalate) oligomer from recycled PET (polyethylene terephthalate)[J]. Korean J Chem Eng,2016,54(4):437−442. doi: 10.9713/kcer.2016.54.4.437 [5] HUANG J, YANG J, CHYU M K, WANG Q, ZHU Z. Continuous-distribution kinetics for degradation of polybutylene terephthalate (PBT) in supercritical methanol[J]. Polym Degrad Stabil,2009,94(12):2142−2148. doi: 10.1016/j.polymdegradstab.2009.09.011 [6] ZHOU Z, YIN N, ZHANG Y, ZHANG Y. Properties of poly (butylene terephthalate) chain-extended by epoxycyclohexyl polyhedral oligomeric silsesquioxane[J]. J Appl Polym Sci,2008,107(2):825−830. doi: 10.1002/app.27106 [7] YOSHIOKA T, GRAUSE G, OTANI S, OKUWAKI A. Selective production of benzene and naphthalene from poly(butylene terephthalate) and poly(ethylene naphthalene-2, 6-dicarboxylate) by pyrolysis in the presence of calcium hydroxide[J]. Polym Degrad Stabil,2006,91(5):1002−1009. doi: 10.1016/j.polymdegradstab.2005.08.010 [8] 刘丽, 戴娟娟, 黄园园, 柳颖萍, 林春绵, 潘志彦. 聚对苯二甲酸丁二醇酯在亚临界水中的催化解聚[J]. 高校化学工程学报,2012,26(3):524−530. doi: 10.3969/j.issn.1003-9015.2012.03.027LIU Li, DAI Juan-juan, HUANG Yuan-yuan, LIU Ying-ping, LIN Chun-mian, PAN Zhi-yan. Catalytic depolymerization of polybutylene terephthalate in subcritical water[J]. J Chem Eng Chin Univ,2012,26(3):524−530. doi: 10.3969/j.issn.1003-9015.2012.03.027 [9] YANG W, SONG L, HU Y, LU H, YUEN R K. Investigations of thermal degradation behavior and fire performance of halogen-free flame retardant poly(1, 4-butylene terephthalate) composites[J]. J Appl Polym Sci,2011,122(3):1480−1488. doi: 10.1002/app.34119 [10] LAVILLA C, ALLA A, DE ILARDUYA A M, BENITO E, GARCÍA-MARTÍN M D G, GALBIS J A, MUÑOZ-GUERRA S. Bio-based poly (butylene terephthalate) copolyesters containing bicyclic diacetalized galactitol and galactaric acid: Influence of composition on properties[J]. Polymer,2012,53(16):3432−3445. doi: 10.1016/j.polymer.2012.05.048 [11] LÜDERWALD I, URRUTIA H. Strukturuntersuchung von polyestern durch direkten abbau im massenspektrometer, 2. aliphatische polyester[J]. Macromo Chem And Phys,1976,177(7):2093−2111. doi: 10.1002/macp.1976.021770716 [12] MA C, WANG J, YUAN Y, MU X, PAN Y, SONG L, HU Y. An insight into gas phase flame retardant mechanisms of AHP versus AlPi in PBT: Online pyrolysis vacuum ultraviolet photoionization time-of-flight mass spectrometry[J]. Combust Flame,2019,209:467−477. doi: 10.1016/j.combustflame.2019.08.020 [13] XIONG-FEI Y U. Identification of PET and PBT by pyrolysis gas chromatography[J]. C J S L,2005,22(2):319−321. [14] MANABE N, YOKOTA Y. The method for analyzing anhydride formed in poly (butylene terephthalate) during thermal and photo-degradation processes and applications for evaluation of the extent of degradation[J]. Polym Degrad Stabil,2000,69(2):183−190. doi: 10.1016/S0141-3910(00)00059-8 [15] SÁNCHEZ-JIMÉNEZ P E, PÉREZ-MAQUEDA L A, PEREJÓN A, CRIADO J M. A new model for the kinetic analysis of thermal degradation of polymers driven by random scission[J]. Polym Degrad Stabil,2010,95(5):733−739. doi: 10.1016/j.polymdegradstab.2010.02.017 [16] CARROCCIO S, RIZZARELLI P, SCALTRO G, PUGLISI C. Comparative investigation of photo-and thermal-oxidation processes in poly (butylene terephthalate)[J]. Polymer,2008,49(16):3371−3381. doi: 10.1016/j.polymer.2008.05.015 [17] VIJAYAKUMAR C T, FINK J K. Pyrolysis studies of aromatic polyesters[J]. Thermochim Acta,1982,59(1):51−61. doi: 10.1016/0040-6031(82)87092-5 [18] KOSHIDUKA T, OHKAWA T, TAKEDA K. Computer simulation of thermal degradation of poly(butylene terephthalate) and analytical problems of terephthalic acid in scission products[J]. Polym Degrad Stabil,2003,79(1):1−11. doi: 10.1016/S0141-3910(02)00228-8 [19] SAMPERI F, PUGLISI C, ALICATA R, MONTAUDO G. Thermal degradation of poly (butylene terephthalate) at the processing temperature[J]. Polym Degrad Stabil,2004,83(1):11−17. doi: 10.1016/S0141-3910(03)00167-8 [20] XU J L, MA B X, KANG C H, XU C C, CHEN Z, LIN Y G. Thermal decomposition kinetics of flame retardant polybutylene terephthalate matrix composites[J]. Mater Sci Forum, 2019, 956: 181−191. [21] MCNEILL I C, BOUNEKHEL M. Thermal degradation studies of terephthalate polyesters: 1. Poly(alkylene terephthalates)[J]. Polym Degrad Stabil,1991,34(1):187−204. [22] QU H, LIU X, XU J, MA H, JIAO Y, XIE J. Investigation on thermal degradation of poly (1, 4-butylene terephthalate) filled with aluminum hypophosphite and trimer by thermogravimetric analysis-fourier transform infrared spectroscopy and thermogravimetric analysis-mass spectrometry[J]. Ind Eng Chem Res,2014,53(20):8476−8483. doi: 10.1021/ie404297r [23] PELLOW-JARMAN M, HETEM M. Comparison of the thermal degradation products of poly (butylene terephthalate) and flame retardant poly (butylene terephthalate) formulations using a pyrolysis FTIR cell[J]. Polym Degrad Stabil,1995,47(3):413−421. doi: 10.1016/0141-3910(95)00006-2 [24] MONTAUDO G, PUGLISI C, SAMPERI F. Primary thermal degradation mechanisms of PET and PBT[J]. Polym Degrad Stabil,1993,42(1):13−28. doi: 10.1016/0141-3910(93)90021-A [25] LÜDERWALD I, URRUTIA H. Strukturuntersuchung von polyestern durch direkten abbau im massenspektrometer, 1. polyester der terephthalsure[J]. Macromol Chem Phys,1976,177(7):2079−2091. doi: 10.1002/macp.1976.021770715 [26] SONO M, KINASHI K, SAKAI W, TSUTSUMI N. Spin-trapping analysis of thermal degradation reaction of poly(butylene terephthalate)[J]. Macromolecules,2017,50(1):254−263. doi: 10.1021/acs.macromol.6b01942 [27] CHIU S J, WU Y S. A comparative study on thermal and catalytic degradation of polybutylene terephthalate[J]. J Anal Appl Pyrolysis,2009,86(1):22−27. doi: 10.1016/j.jaap.2009.03.003 [28] 钱和生. 聚对苯二甲酸丁二酯纤维热裂解分析[J]. 合成纤维工业,2007,30(4):60−62. doi: 10.3969/j.issn.1001-0041.2007.04.020QIAN He-sheng. Analysis of thermal cracking of polybutylene terephthalate fibers[J]. China Synt Fibe Ind,2007,30(4):60−62. doi: 10.3969/j.issn.1001-0041.2007.04.020 [29] FARROKHPOUR H, JOUYPAZADEH H. Description of adenine and cytosine on au (111) nano surface using different DFT functionals (PW91PW91, wB97XD, M06-2X, M06-L and CAM-B3LYP) in the framework of ONIOM scheme: Non-periodic calculations[J]. Chem Phys,2017,488:1−10. [30] WALKER M, HARVEY A J, SEN A, DESSENT C E. Performance of M06, M06-2X, and M06-HF density functionals for conformationally flexible anionic clusters: M06 functionals perform better than B3LYP for a model system with dispersion and ionic hydrogen-bonding interactions[J]. J Phys Chem A,2013,117(47):12590−12600. doi: 10.1021/jp408166m [31] FRISCH M J, TRUCKS G W, SCHLEGEL J, SCUSERIA G E, ROBB M A, CHEESEMAN J R. Gaussian 09[CP]. Revision C. 01, 2010. [32] HUANG J, MU X, LUO X, MENG H, WANG H, JIN L, LAI B. DFT studies on pyrolysis mechanisms of tetrabromobisphenol A (TBBPA)[J]. Environ Sci Pollut R,2021,28(48):68817−68833. doi: 10.1007/s11356-021-15426-9 [33] HUANG J, MENG H, LUO X, MU X, XU W, JIN L, LAI B. Insights into the thermal degradation mechanisms of polyethylene terephthalate dimer using DFT method[J]. Chemosphere,2022,291:133112. doi: 10.1016/j.chemosphere.2021.133112 [34] BROOKS C T, PEACOCK S J, REUBEN B G. Pyrolysis of benzene[J]. J Chem Soc, Faraday Trans 1,1979,75:652−662. doi: 10.1039/f19797500652 [35] YINHE S U, WANG X, JUNRONG Y U, WANG Y, ZHU J, ZUMING H U. Effect of initiator content on the polymerization kinetics of cyclic butylene terephthalate and resultant polymer[J]. J Donghua Univ (Nat Sci),2018,41(3):366−385. [36] VIJAYAKUMAR C T, PONNUSAMY E, BALAKRISHNAN T, KOTHANDARAMAN H. Thermal and pyrolysis studies of copolyesters[J]. J Polym Sci Part A Polym Chem,1982,20(9):2715−2725. doi: 10.1002/pol.1982.170200929 [37] BOTELHO G, QUEIRÓS A, LIBERAL S, GIJSMAN P. Studies on thermal and thermo-oxidative degradation of poly (ethylene terephthalate) and poly (butylene terephthalate)[J]. Polym Degrad Stabil,2001,74(1):39−48. doi: 10.1016/S0141-3910(01)00088-X [38] SAMPERI F, PUGLISI C, ALICATA R, MONTAUDO G. Thermal degradation of poly (ethylene terephthalate) at the processing temperature[J]. Polym Degrad Stabil,2004,83(1):3−10. doi: 10.1016/S0141-3910(03)00166-6 [39] KINOSHITA R, TERAMOTO Y, YOSHIDA H. Kinetic analysis of the thermal decomposition of polyesters by simultaneous TG-DTA/FT-IR[J]. Thermochim Acta,1993,222(1):45−52. doi: 10.1016/0040-6031(93)80537-K [40] LUM R M. Thermal decomposition of poly (butylene terephthalate)[J]. J Polym Sci Part A Polym Chem,1979,17(1):203−213. doi: 10.1002/pol.1979.170170120 [41] SATO H, KONDO K, TSUGE S, OHTANI H, SATO N. Mechanisms of thermal degradation of a polyester flame-retarded with antimony oxide/brominated polycarbonate studied by temperature-programmed analytical pyrolysis[J]. Polym Degrad Stabil,1998,62(1):41−48. doi: 10.1016/S0141-3910(97)00259-0 -





 下载:
下载: