Nanosized amorphous nickel-boron alloy electrocatalysts for hydrogen evolution reaction under alkaline conditions
-
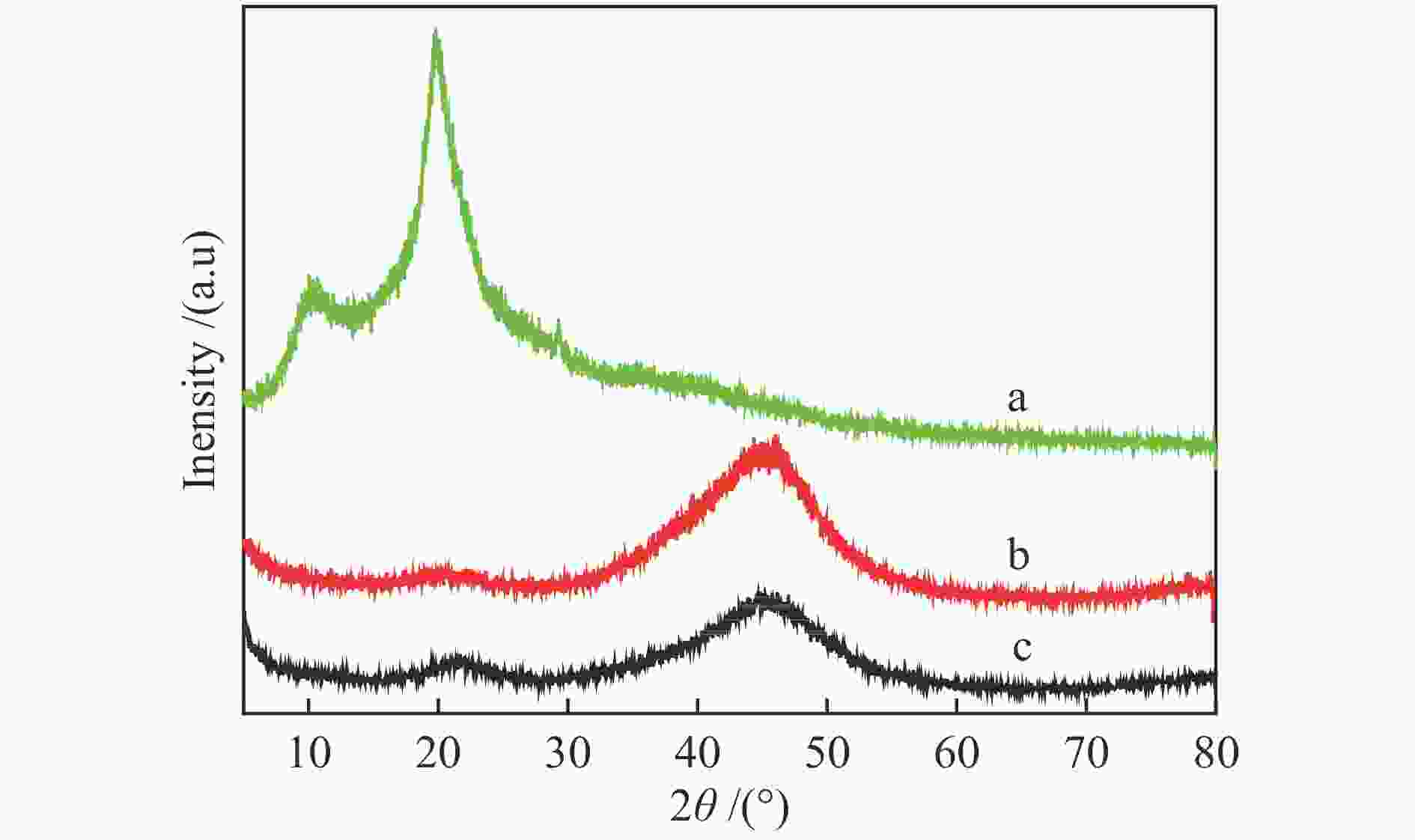
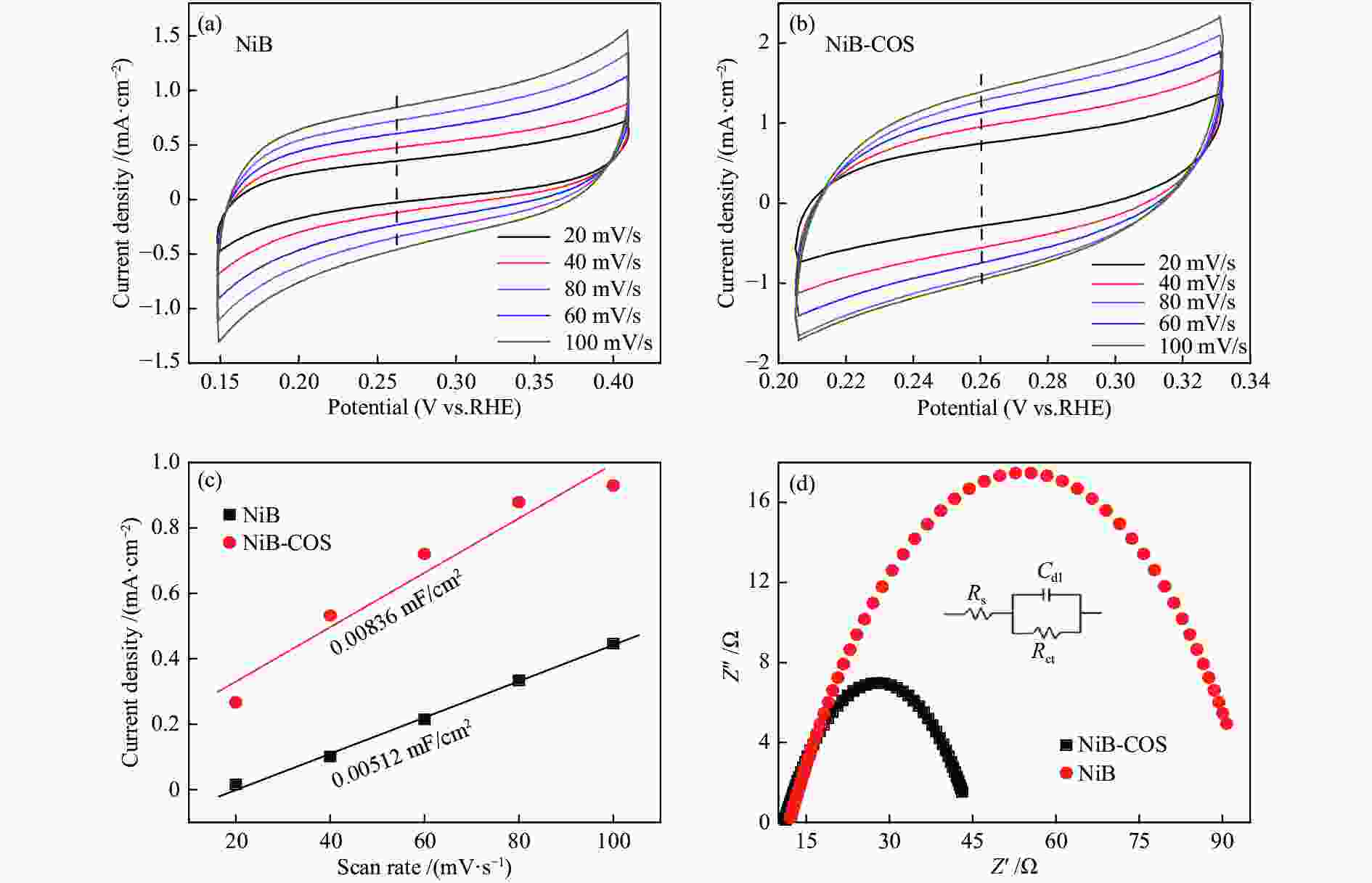
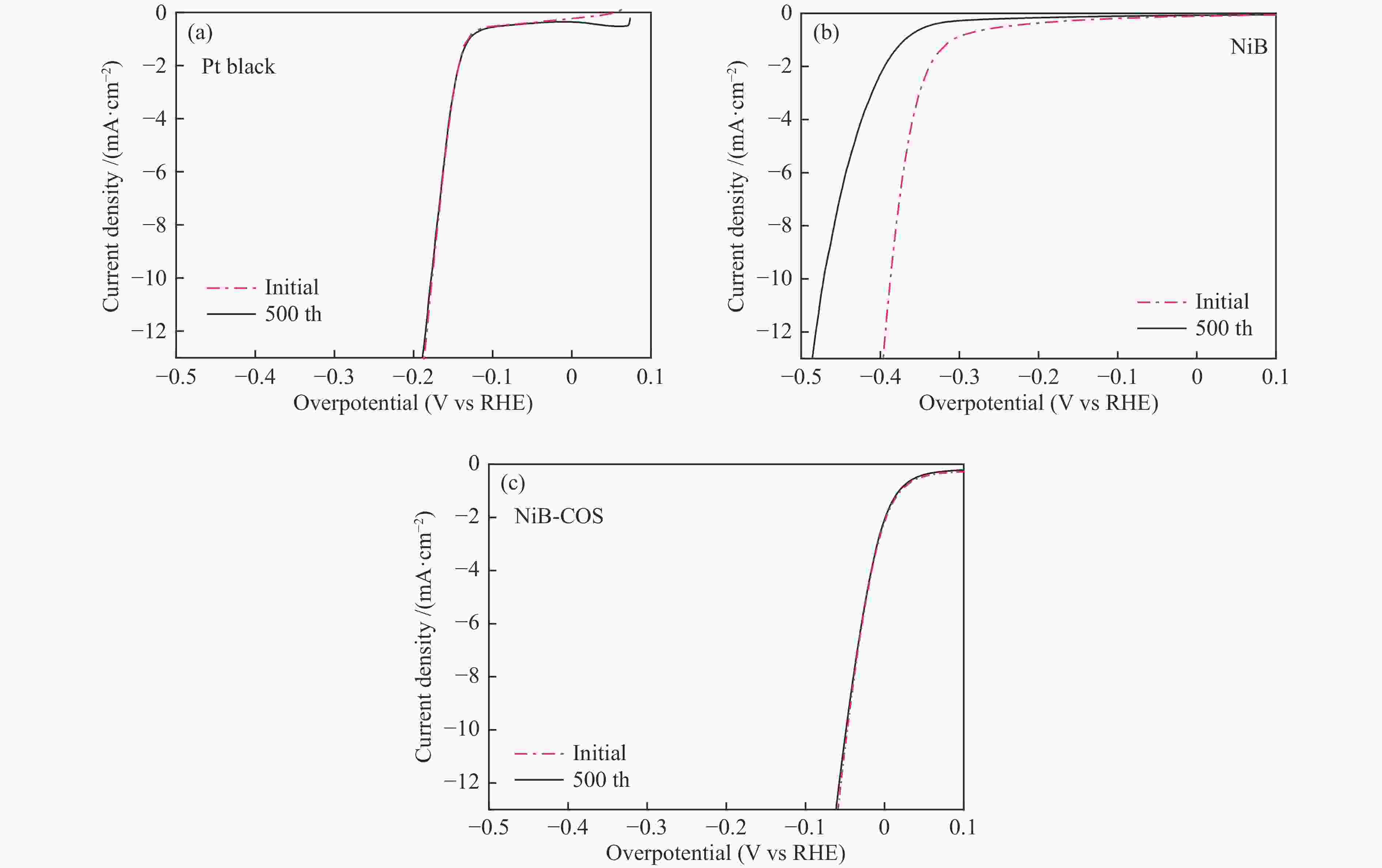
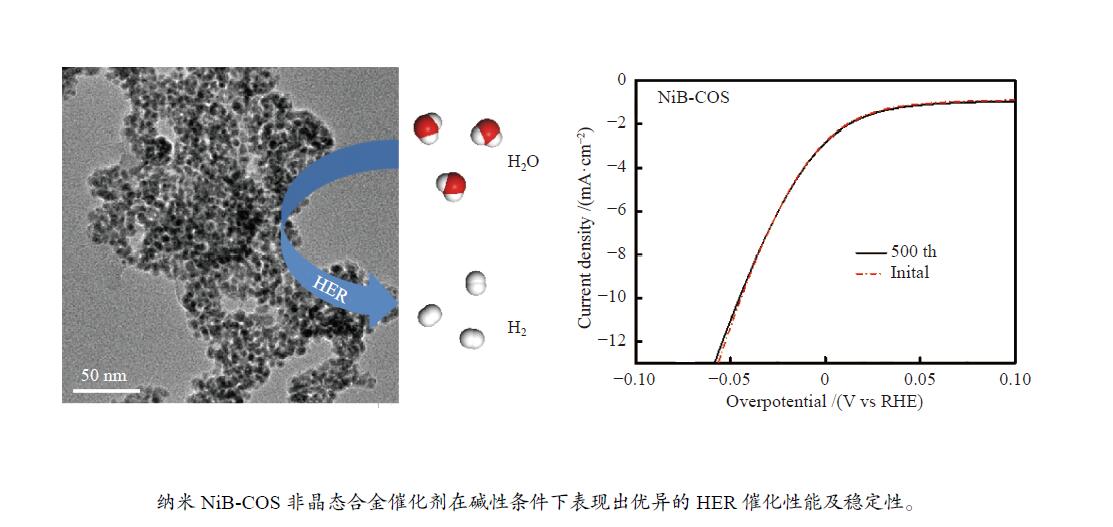
摘要: 可持续能源电解水制氢是实现零碳排放氢经济的有效途径。碱性环境下的电催化析氢反应(HER)是电解水技术主要的能量转换过程之一。开发高活性、低成本的非贵金属催化剂是碱性电解水析氢反应的关键所在。本研究以壳寡糖为保护剂,采用简单易行的化学还原法制备了纳米NiB非晶合金电催化剂并用于碱性析氢反应。采用X射线衍射(XRD)、透射电子显微镜(TEM)、电感耦合等离子体分析(ICP)和X射线光电子能谱(XPS)等多种表征方法研究了不同条件下获得的催化剂结构组成及特征物性参数。结果表明,壳寡糖的加入可以有效调控纳米粒子的平均粒径为4 nm左右,提升活性比表面积,增加活性位点,从而提高其电催化活性。所制备的NiB-COS在1.0 mol/L NaOH中表现出优异的HER性能,析氢反应起始过电位仅为15.1 mV,在电流密度为10 mA/cm2时HER过电位为49.4 mV,Tafel斜率为86.1 mV/dec,为制备高活性、低成本、简单易得的HER电催化剂提供了重要策略。Abstract: Hydrogen production from electrolyzed water driven by sustainable energy is an effective way to achieve the hydrogen economy with zero carbon emission. Alkaline electrocatalytic hydrogen evolution reaction (HER) is one of the main energy transforming processes in the field of electrolytic water technology. The development of active and cost-effective nonprecious catalytic electrodes is of great importance to alkaline hydrogen evolution reaction. Amorphous nanosized nickel-boron alloy particles (NiB-COS) have been obtained by using chitosan oligosaccharides (COS) as a stabilizer via chemical reduction method. The as-prepared electrocatalysts have been used for the hydrogen evolution reaction (HER). The electrocatalysts have been characterized by using X-ray diffraction (XRD), transmission electron microscopy (TEM), inductively coupled plasma analysis (ICP) and X-ray photoelectron spectroscopy (XPS). NiB-COS displays a significant increase in hydrogen evolution reaction properties in alkaline media, affording low overpotentials of 49.4 mV at 10 mA/cm2 and 15.1 mV onset overpotential for the hydrogen evolution reaction. Tafel slope of NiB-COS is 86.1 mV/dec. Remarkably, the formation of a nickel-boron alloyed phase and the decrease of particle size are helpful to improve HER activity of NiB-COS. The experimental data indicated that the NiB-COS showed excellent long-term stability as a very active electrocatalyst.
-
Key words:
- hydrogen evolution reaction /
- NiB-COS /
- overpotential /
- Tafel slope /
- alkaline water electrolysis
-
Table 1 Surface areas and bulk composition of the NiB and NiB-COS catalysts
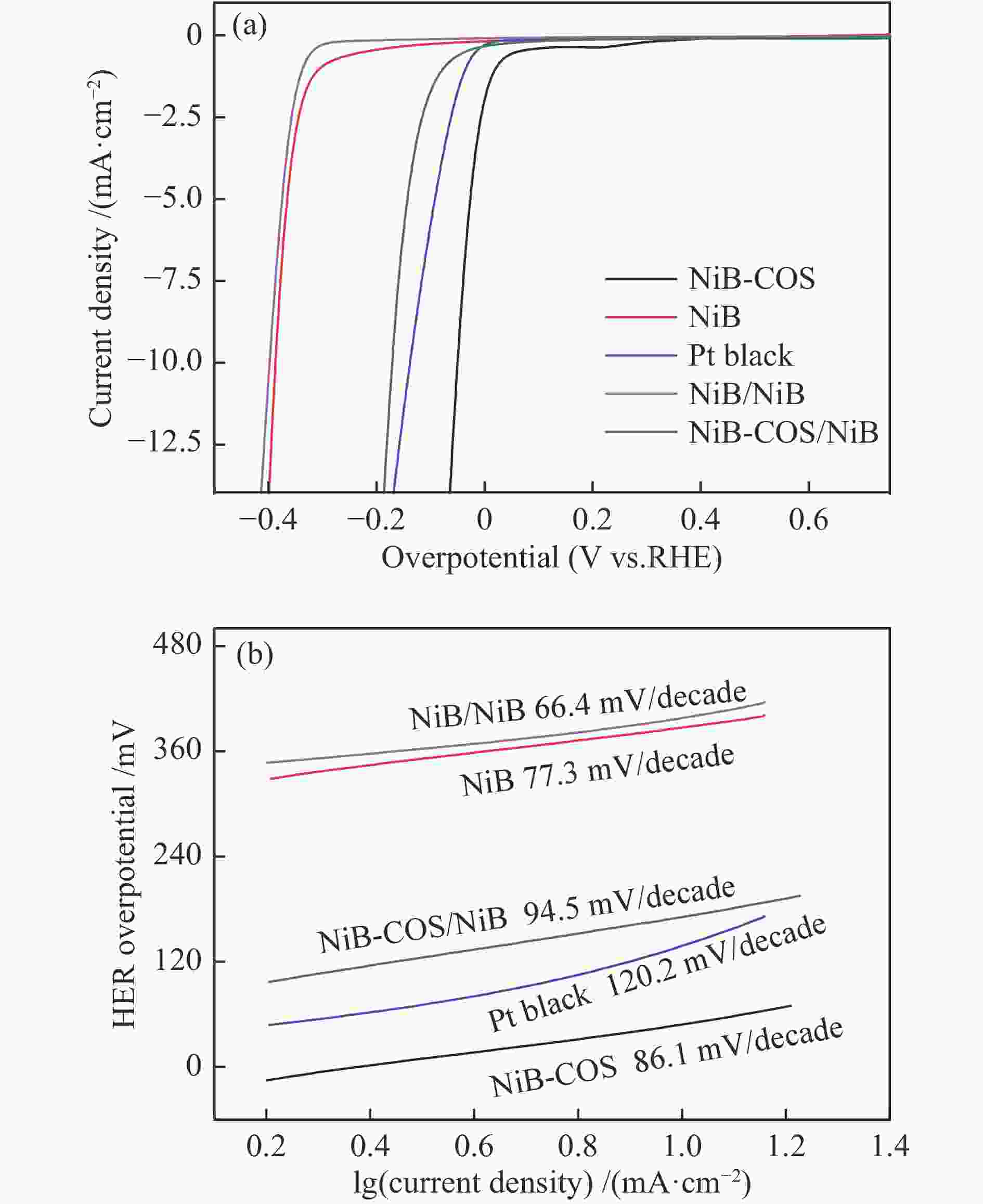
Catalyst Composition BET surface area /(m2·g−1 ) NiB Ni69.5B30.5 29.5 NiB-COS Ni74.5B25.5 68.6 Table 2 HER performance of NiB-based electrocatalysts in basic solution
Electrolyte Overpotential NiB-COS Pt black NiB-COS/ NiB NiB/ NiB NiB onset overpotential /mV 15.1 56.5 122.9 349.7 355.8 Basic condition overpotential
/mV @(10 mA/cm2)49.4 137.0 170.9 387.1 398.5 Tafel slope /(mV·dec−1) 86.1 120.2 94.5 66.4 77.3 -
[1] WANG H Y, WENG C C, REN J T, YUAN Z Y. An overview and recent advances in electrocatalysts for direct seawater splitting[J]. Front Chem Sci Eng,2021,15(6):1408−1426. doi: 10.1007/s11705-021-2102-6 [2] ZHENG Y, JIAO Y, JARONIEC M, QIAO S Z. Advancing the electrochemistry of the hydrogen evolution reaction through combining experiment and theory[J]. Angew Chem Int Ed,2015,54:52−65. doi: 10.1002/anie.201407031 [3] DEBE M K. Electrocatalyst approaches and challenges for automotive fuel cells[J]. Nature,2012,486:43−51. doi: 10.1038/nature11115 [4] SHARMA S, GHOSHAL S K. Hydrogen the future transportation fuel from production to applications[J]. Renewable Sustainable Energy Rev,2015,43:1151−1158. doi: 10.1016/j.rser.2014.11.093 [5] NIKOLAIDIS P, POULLIKKAS A. A comparative overview of hydrogen production processes[J]. Renewable Sustainable Energy Rev,2017,67:597−611. doi: 10.1016/j.rser.2016.09.044 [6] TRIPATHY R K, SAMANTARA A K, BEHERA J N. Metal-organic framework (MOF)-derived amorphous nickel boride: an electroactive material for electrochemical energy conversion and storage application[J]. Sustainable Energy Fuels,2021,5(4):1184−1193. doi: 10.1039/D0SE01831G [7] SCHMIDT T J, ROSS JR P N, MARKOVIC N M. Temperature dependent surface electrochemistry on Pt single crystals in alkaline electrolytes Part 2. The hydrogen evolution/oxidation reaction[J]. J Electro Chem,2002,524−525:252−260. [8] MARKOVIC N M, GRGUR B N, LUCAS C A, ROSS P N. Electrooxidation of CO and H2/CO mixtures on Pt(111) in acid Solutions[J]. J Phys Chem B,1999,103:487−495. doi: 10.1021/jp983177c [9] YANG Y, XU X, WANG X. Synthesis of Mo-based nanostructures from organic-inorganic hybrid with enhanced electrochemical for water splitting[J]. Sci China Mater,2015,58:775−784. doi: 10.1007/s40843-015-0088-4 [10] DOU S, TAO L, HUO J, WANG S Y, DAI L M. Etched and doped Co9S8/graphene hybrid for oxygen electrocatalysis[J]. Energy Environ Sci,2016,9:l320−l326. [11] CABÁN-ACEVEDO M, STONE M L, SCHMIDT J R, THOMAS J G, DING Q, CHANG H C, TSAI M L, He J H, JIN S. Efficient hydrogen evolution catalysis using ternary pyrite-type cobalt phosulphide[J]. Nat Mater,2015,14:1245−1251. doi: 10.1038/nmat4410 [12] MORALES-GUIO C G, HU X L. Amorphous molybdenum sulfides as hydrogen evolution catalysts[J]. Acc Chem Res,2014,47:2671−2681. [13] WU Z, GUO, W JIE, RONG L, WANG D. Hierarchically porous electrocatalyst with vertically aligned defect-rich CoMoS nanosheets for HER in alkaline medium[J]. ACS Appl Mater Interfaces,2017,9:5288−5294. doi: 10.1021/acsami.6b15244 [14] MA T Y, DAI S, JARONIEC M, SHI Z Q. Metal-organic framework derived hybrid Co3O4-carbon porous nanowire arrays as reversible oxygen evolution electrodes[J]. J Am Chem Soc,2014,l36:13925−l3931. [15] LIU Q, TIAN J Q, CUI W, JIANG P. Carbon nanotubes decorated with CoP nanocrystals a highly active non-noble-metal nanohybrid electrocatalyst for hydrogen evolution[J]. Angew Chem Int Ed,2014,53:6710−6714. doi: 10.1002/anie.201404161 [16] BAO J, ZHANG X D, FAN B, ZHANG J J, ZHOU M, YANG W L, HU X, WANG H, PAN B C, XIE Y. Ultrathin spinel-structured nanosheets rich in oxygen deficiencies for enhanced electrocatalytic water oxidation[J]. Angew Chem Int Ed,2015,54:7399−7404. doi: 10.1002/anie.201502226 [17] ZHU Y P, LIU Y P, REN T Z, YUAN Z Y. Self-supported cobalt phosphide mesoporous nanorod arrays a flexible and bifunctional electrode for highly active electrocatalytic water reduction and oxidation[J]. Adv Funct Mater,2015,25:7337−7347. doi: 10.1002/adfm.201503666 [18] YAN Y, XIA B Y, GE X M, LIU Z L, FISHER A, WANG X. A flexible electrode based on iron phosphide nanotubes for overall water splitting[J]. Chem Eur J,2015,21:18062−18067. doi: 10.1002/chem.201503777 [19] WONTERGHEM J V, MORUP S, KOCH C J W, CHARLES S W, WELLS S. Formation of ultra-fine amorphous alloy particles by reduction in aqueous solution[J]. Nature,1986,322:622−62. doi: 10.1038/322622a0 [20] WANG Y D, AI X P, YANG H X. Electrochemical hydrogen storage behaviors of ultrasmall amorphous Co-B alloy particles[J]. Chem Mater,2004,16:5194−5197. doi: 10.1021/cm049252f [21] LIU Z, LI Z L, WANG F, LIU J J, JI J, PARK K C, ENDO M. Electroless preparation and characterization of Ni-B nanoparticles supported on multi-walled carbon nanotubes and their catalytic activity towards hydrogenation of styrene[J]. Mater Res Bull,2012,47:338−343. doi: 10.1016/j.materresbull.2011.11.010 [22] WU Z J, GE S H, ZHANG M H, LI W, MU S C, TAO K Y. Controlled synthesis of supported nickel boride catalyst using electroless plating[J]. J Phys Chem C,2007,111:8587−8593. doi: 10.1021/jp070096k [23] PARKS G L, PEASE M L, BURNS A W, LAYMAN K A, BUSSELL M E, WANG X, HANSON J, RODRIGUEZ J A. Characterization and hydrodesulfurization properties of catalysts derived from amorphous metal-boron materials[J]. J Catal,2007,246:277−292. doi: 10.1016/j.jcat.2006.12.009 [24] SKRABALAK S E, SUSLICK K S. On the possibility of metal borides for hydrodesulfurization[J]. Chem Mater,2006,18:3103−3107. doi: 10.1021/cm060341x [25] LEWANDOWSKI M. Hydrotreating activity of bulk NiB alloy in model reaction of hydrodenitrogenation of carbazole[J]. Appl Catal B: Environ,2015,168:322−332. [26] ZENG M, WANG H, ZHAO C, WEI J K, QI K, WANG W L, BAI X D. Nanostructured amorphous nickel boride for high efficiency electrocatalytic hydrogen evolution over a broad pH range[J]. ChemCatChem,2016,8:708−712. [27] LI Z P, SHANG J P, SU C N, ZHANG S B, WU M X, GUOY. Preparation of amorphous NiP-based catalysts for hydrogen evolution reactions[J]. J Fuel Chem Technol,2018,46(4):473−478. doi: 10.1016/S1872-5813(18)30021-5 [28] DENG K, REN T, XU Y, DENG K, REN T L, XU Y, LIU S L, DAI Z C, WANG Z Q, LI X N, LIANG WANG, WANG H J. Crystalline core-amorphous shell heterostructures: Epitaxial assembly of NiB nanosheets onto PtPd mesoporous hollow nanopolyhedra for enhanced hydrogen evolution electrocatalysis[J]. J Mater Chem A,2020,8(18):8927−8933. doi: 10.1039/D0TA02537B [29] LEGRAND J, TALEB A, GOTA S, GUITTET M J, PETIT C. Synthesis and XPS characterization of nickel boride nanoparticles[J]. Langmuir,2002,18:4131−4137. doi: 10.1021/la0117247 [30] HUANG T, SHEN T, GONG M, DENG S F, LAI C L, LIU X P, ZHAO T H, TENG L, WANG D L. Ultrafine Ni-B nanoparticles for efficient hydrogen evolution reaction[J]. Chin J Catal,2019,40(12):1867−1873. doi: 10.1016/S1872-2067(19)63331-0 [31] ZHANG R, LIU H, WANG C, WANG L C, YANG Y J, GUO Y H. Electroless plating of transition metal boride with high boron content as superior HER electrocatalyst[J]. ChemCatChem,2020,12(11):3068−3075. [32] BOCKRIS J O M, POTTER E C. The mechanism of the cathodic hydrogen evolution reaction[J]. J Electrochem Soc,1952,99:169−186. doi: 10.1149/1.2779692 [33] POPCZUN E J, MCKONE J R, READ C G, BIACCHI A J, WILTROUT A M, LEWIS N S, SCHAAK R E. Nanostructured nickel phosphide as an electrocatalyst for the hydrogen evolution reaction[J]. J Am Chem Soc,2013,135(25):9267−9270. doi: 10.1021/ja403440e [34] PAN Y, HU W, LIU D, LIU Y, LIU C. Carbon nanotubes decorated with nickel phosphide nanoparticles as efficient nanohybrid electrocatalysts for the hydrogen evolution reaction[J]. J Mater Chem A,2015,3(24):13087−13094. doi: 10.1039/C5TA02128F [35] PAN Y, LIU Y, LIU C. Nanostructured nickel phosphide supported on carbon nanospheres: Synthesis and application as an efficient electrocatalyst for hydrogen evolution[J]. J Power Sources,2015,285:169−177. doi: 10.1016/j.jpowsour.2015.03.097 [36] LIN Y, ZHANG J, PAN Y, LIU Y. Nickel phosphide nanoparticles decorated nitrogen and phosphorus co-doped porous carbon as efficient hybrid catalyst for hydrogen evolution[J]. Appl Surf Sci,2017,422:828−837. doi: 10.1016/j.apsusc.2017.06.102 [37] PAN Y, YANG N, CHEN Y, LIN Y, LI Y, LIU Y, LIU C. Nickel phosphide nanoparticles nitrogen-doped graphene hybrid as an efficient catalyst for enhanced hydrogen evolution activity[J]. J Power Sources,2015,297:45−52. doi: 10.1016/j.jpowsour.2015.07.077 [38] WANG P, PU Z, LI Y, WU L, TU Z, JIANG M, KOU Z, SAANA AMIINU I, MU S. Iron-doped nickel phosphide nanosheet arrays: An efficient bifunctional electrocatalyst for water splitting[J]. ACS Appl Mater Interfaces,2017,9(31):26001−26007. doi: 10.1021/acsami.7b06305 -





 下载:
下载: