Preparation of Rh/N-GMCs nanocatalyst and its catalytic performance for the hydrolytic dehydrogenation of ammonia borane
-
摘要:
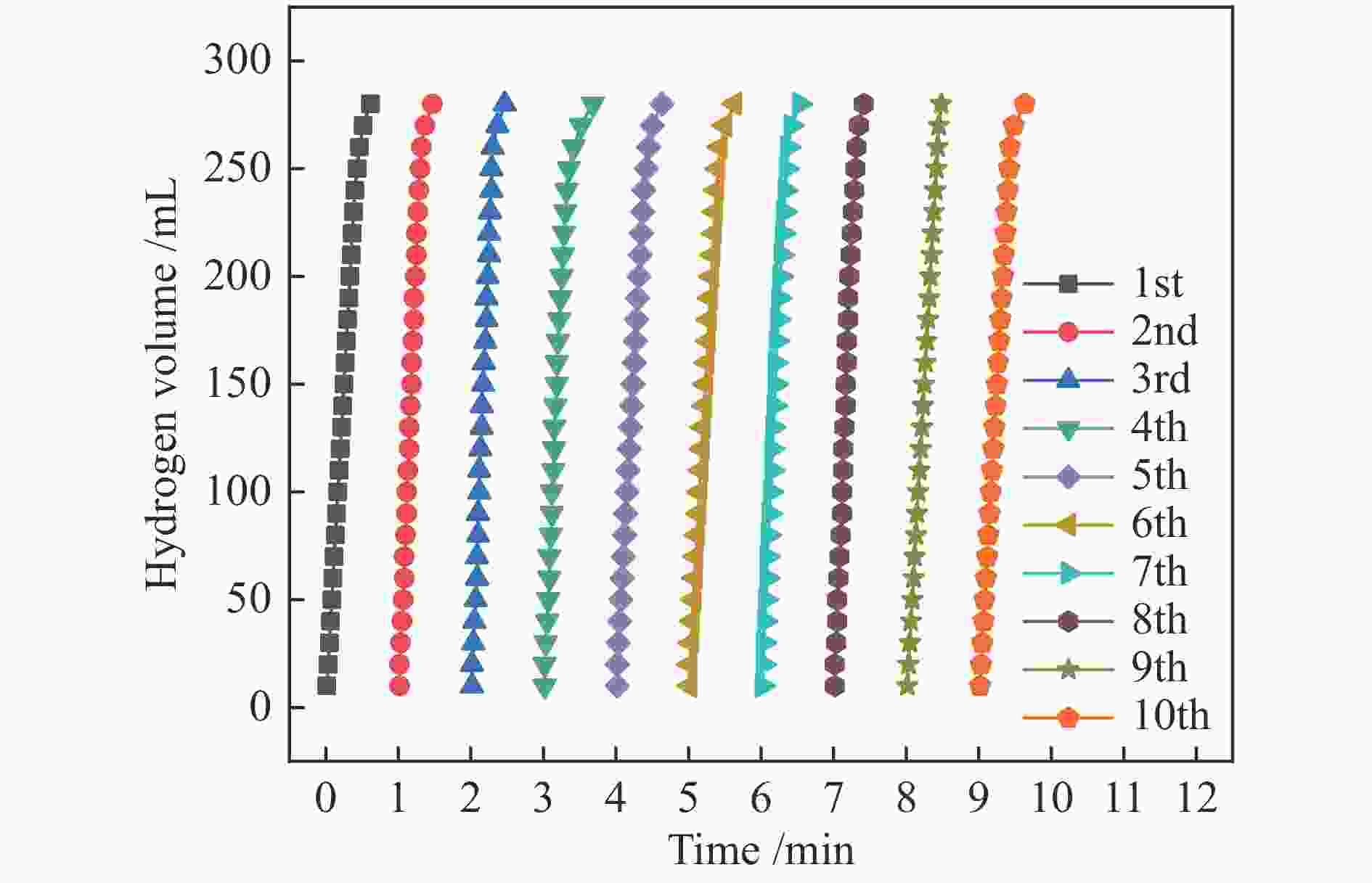
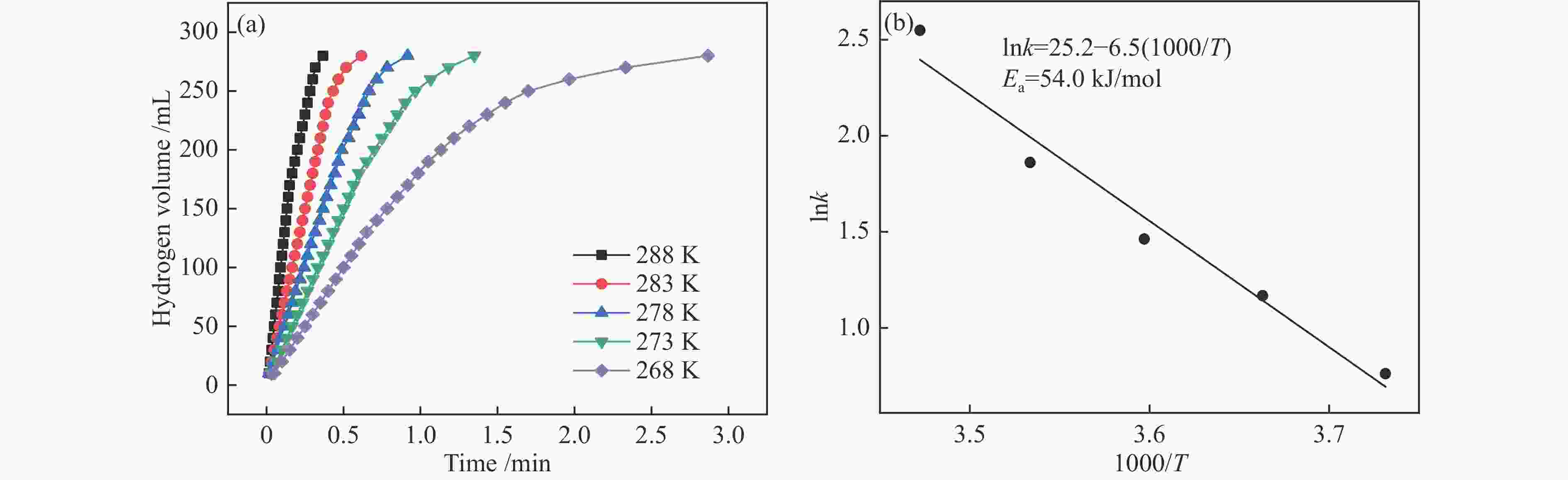
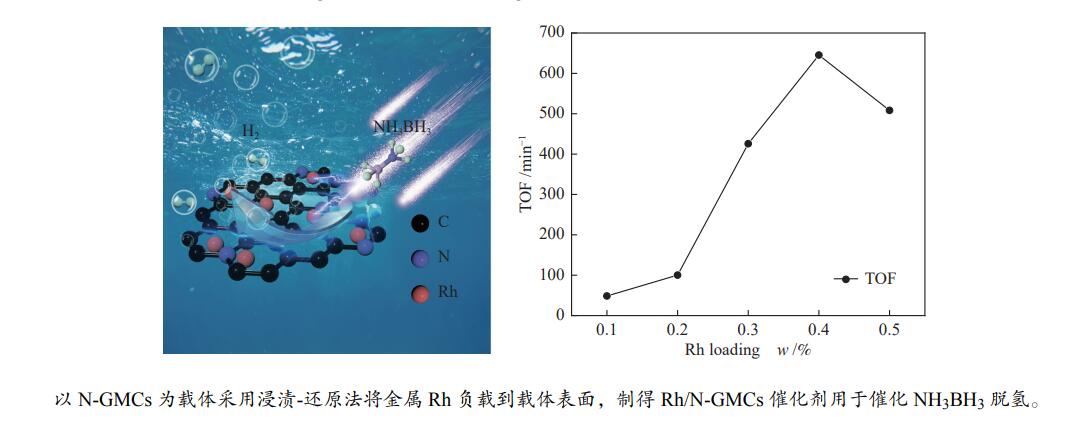
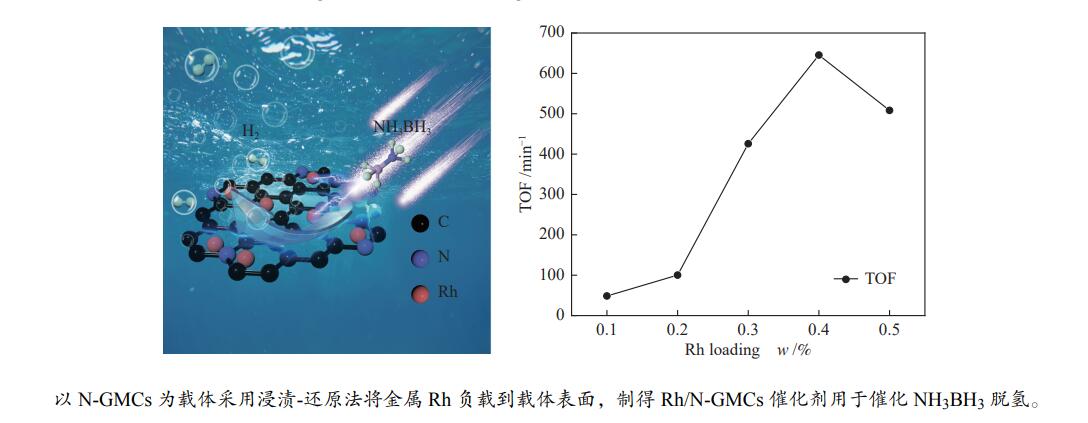
本实验通过高温焙烧三聚氰胺和葡萄糖混合物制备出具有独特层状结构的氮掺杂石墨相炭材料(N-GMCs)。再以N-GMCs为载体采用浸渍-还原法将金属Rh负载到载体表面,最终制得Rh/N-GMCs催化剂。结果表明,Rh与N-GMCs之间存在强的金属-载体相互作用,Rh的负载量为0.4%时,反应转化频率(TOF)值达到峰值,此时0.4%Rh/N-GMCs催化剂的TOF为645.3 min–1,该催化剂上氨硼烷水解的活化能(Ea)为54.0 kJ/mol,氨硼烷脱氢速率随氨硼烷浓度和催化剂浓度呈现正相关,该催化剂循环10次之后,催化活性未明显下降,表明该催化剂具有较好的循环稳定性。
Abstract:In this paper, nitrogen-doped graphitic carbon materials (N-GMCs) with unique layered structure were prepared by calcining a mixture of melamine and glucose at high temperature. The Rh/N-GMCs catalyst was finally prepared by impregnation-reduction method using N-GMCs as the carrier to support the metal Rh on the surface of the carrier. The results showed that there was a strong metal-support interaction between Rh and N-GMCs, and the reaction transition frequency (TOF) value reached a peak when the loading amount of Rh was 0.4%. At this time, the TOF value of AB dehydrogenation reaction catalyzed by 0.4%Rh/N-GMCs catalyst was 645.3 min–1, and the activation energy (Ea) of ammonia borane hydrolysis on this catalyst was 54.0 kJ/mol. The rate was positively correlated with the ammonia borane concentration and the catalyst concentration. After 10 cycles of the catalyst, the catalytic activity did not decrease significantly, indicating that the catalyst had excellent cyclic stability.
-
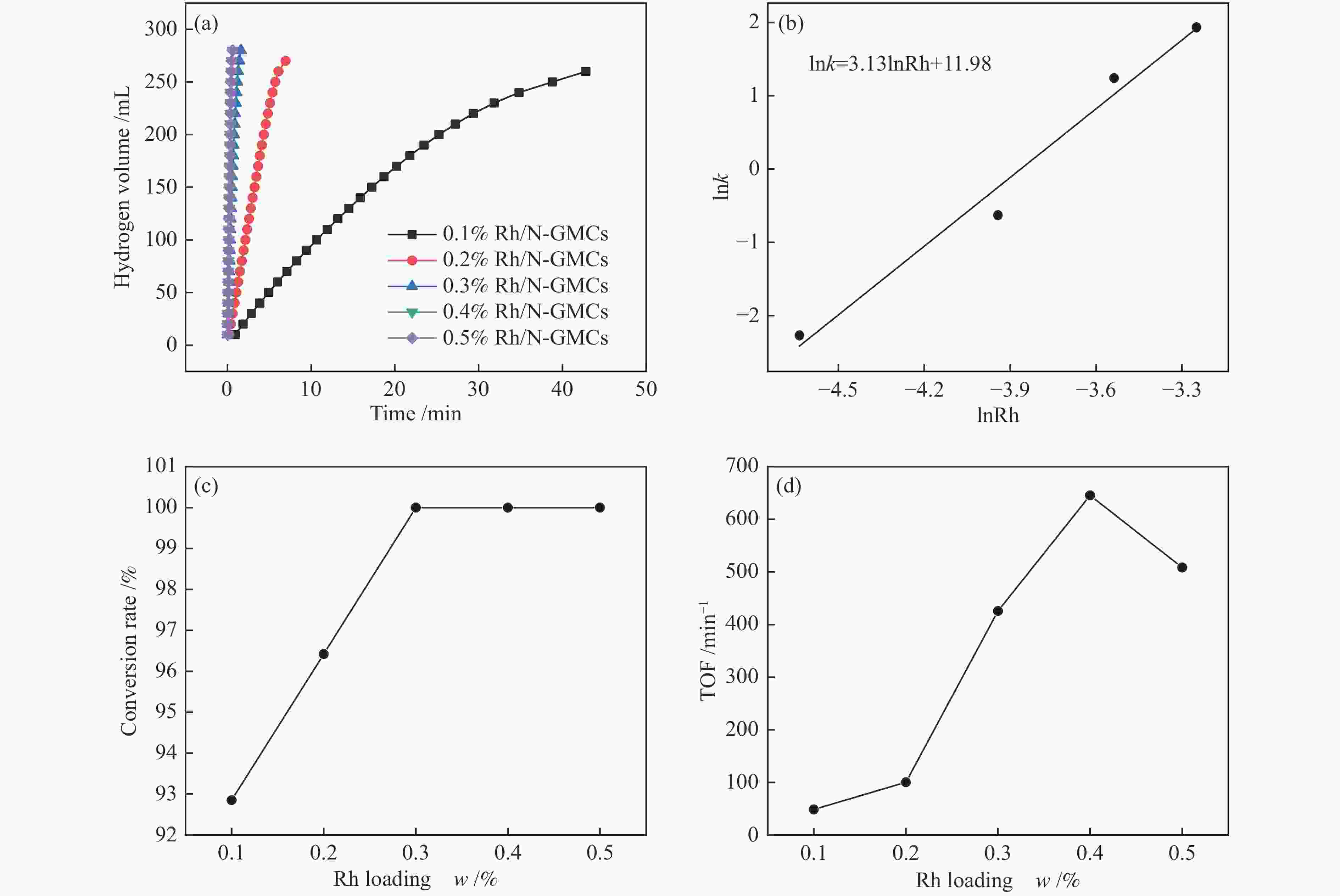
图 6 (a) 不同Rh负载量的Rh/N-GMCs催化剂在283 K催化AB水解反应中的脱氢速率;(b) 脱氢速率与Rh负载量的对数值;(c) AB的转化率随Rh负载量的变化;(d) 不同Rh负载量的Rh/N-GMCs催化剂在283 K脱氢AB所对应的TOF值
Figure 6 (a) Plots of hydrogen evolution rate for the hydrolysis of AB with different rhodium concentration at 283 K; (b) logarithmic plot of hydrogen evolution rate versus Rh loading; (c) conversion rate of AB for the dehydrogenation of AB at different rhodium concentration; (d) turnover frequency (TOF) values for the dehydrogenation of AB at 283 K when Rh/N-GMCs with different rhodium loading
表 1 室温下水溶液中AB水解制氢各种贵金属催化剂的催化活性
Table 1 Catalytic activities of various noble metal catalysts for AB hydrolysis in aqueous solution at room temperature
-
[1] ASCHLAPBACH L, ZUTTEL A. Hydrogen-storage materials for mobile applications[J]. Nature,2001,414(6861):353−358. doi: 10.1038/35104634 [2] ZHAO Y Z, MENG B, CHEN L X, WANG G, ZHENG J Y, GU C H, ZHANG X, ZHANG J F. Utilization status of hydrogen energy[J]. Chem Ind Eng Prog,2015,34(9):3248−3255. [3] IMAMURA H, MASANARI K, KUSUHARA M, KATSUMOTO H, SUMI T, SAKATA Y. High hydrogen storage capacity of nanosized magnesium synthesized by high energy ball-milling[J]. J Alloy Compd, 2005, 386(1/2): 211–216. [4] SRINIVASAN S, ESCOBAR D, GOSWAMI Y, STEFANAKOS E. Effects of catalysts doping on the thermal decomposition behavior of Zn(BH)[J]. Int J Hydrogen Energy,2008,33(9):2268−2272. doi: 10.1016/j.ijhydene.2008.02.062 [5] AU M, JURGENSEN A R, SPENCER W A, ANTON D L, PINKERTON F E, HWANG S J, KIM C, BOWMAN R C. Stability and reversibility of lithium borohydrides doped by metal halides and hydrides[J]. J Phys Chem C,2008,112(47):18661−18671. doi: 10.1021/jp8024304 [6] BOWMAN R C, BRENT F. Metallic hydrides I: Hydrogen storage and other gas-phase applications[J]. MRS Bull,2002,27(9):6. [7] WAN C, ZHU M Y, DU L, XU L X, YE M F, AN Y. Highly efficient aerobic oxidation of tetralin to α-tetralone over MnOx-CoOy/γ-Al2O3 catalysts[J]. Catal Commun,2019,125:87−92. doi: 10.1016/j.catcom.2019.04.003 [8] HU M G, GEANANGEL R A, WENDLANDT W W. The thermal decomposition of ammonia borane[J]. Thermochim Acta,1978,23(2):249−255. doi: 10.1016/0040-6031(78)85066-7 [9] WANG P, KANG X D. Hydrogen-rich boron-containing materials for hydrogen storage[J]. Dalton Trans,2008,40(40):5400−5413. [10] 陈健民. 镍基金属纳米复合材料的合成及其催化水合肼分解产氢[D]. 南昌: 江西师范大学, 2017.CHEN Jian-ming. Synthesis of Ni-based metal nanocomposites for hydrogen production from hydrous hydrazine[D]. Nanchang: Jiangxi Normal University, 2017. [11] XU Q, CHANDRA M. Catalytic activities of non-noble metals for hydrogen generation from aqueous ammonia-borane at room temperature[J]. J Power Sources,2006,163(1):364−370. doi: 10.1016/j.jpowsour.2006.09.043 [12] ÖZHAVA D, ÖZKAR S. Nanoalumina-supported rhodium(0) nanoparticles as catalyst in hydrogen generation from the methanolysis of ammonia borane[J]. Mol Catal,2017,439:50−69. doi: 10.1016/j.mcat.2017.06.016 [13] ÖZHAVA D, ÖZKAR S. Rhodium(0) nanoparticles supported on nanosilica: Highly active and long lived catalyst in hydrogen generation from the methanolysis of ammonia borane[J]. Appl Catal B: Environ,2016,181:716−726. doi: 10.1016/j.apcatb.2015.08.038 [14] YAN J M, ZHANG X B, SHIOYAMA H, XU Q. Room temperature hydrolytic dehydrogenation of ammonia borane catalyzed by Co nanoparticles[J]. J Power Sources,2010,195(4):1091−1094. doi: 10.1016/j.jpowsour.2009.08.067 [15] YAN J M, ZHANG X B, HAN S, SHIOYAMA H, XU Q. Iron-nanoparticle-catalyzed hydrolytic dehydrogenation of ammonia borane for chemical hydrogen storage[J]. J Power Sources,2010,39(23):2287−2289. [16] 邹爱华, 徐晓梅, 周浪, 林路贺, 康志兵. 石墨烯负载Co-CeOx纳米复合物的制备及其对氨硼烷水解产氢的催化性能[J]. 燃料化学学报,2021,49(9):1371−1378. doi: 10.1016/S1872-5813(21)60085-3ZOU Ai-hua, XU Xiao-mei, ZHOU Lang, LIN Lu-he, KANG Zhi-bing. Preparation of graphene-supported Co-CeOx nanocomposites as a catalyst for the hydrolytic dehydrogenation of ammonia borane[J]. J Fuel Chem Technol,2021,49(9):1371−1378. doi: 10.1016/S1872-5813(21)60085-3 [17] STAUBITZ A, ROBERTSON A P, MANNERS I. Ammonia-borane and related compounds as dihydrogen sources[J]. Chem Rev,2010,110(7):4079−4124. doi: 10.1021/cr100088b [18] MARDER T B. Will we soon be fueling our automobiles with ammonia-borane[J]. Angew Chem Int Ed,2007,46(43):8116−8118. doi: 10.1002/anie.200703150 [19] 姜伟丽, 何利梅, 黄斌, 陈雅琪, 周广林, 周红军. Rh-BIPHEPHOS催化剂作用下的丁烯异构与氢甲酰化反应[J]. 燃料化学学报,2021,49(8):1173−1180. doi: 10.1016/S1872-5813(21)60126-3JIANG Wei-li, HE Li-mei, HUANG Bin, CHEN Ya-qi, ZHOU Guang-in, ZHOU Hong-jun. Isomerization and hydroformylation of butenes under the catalysis of Rh-BIPHEPHOS[J]. J Fuel Chem Technol,2021,49(8):1173−1180. doi: 10.1016/S1872-5813(21)60126-3 [20] 石张平, 祁晓岚, 李旭光, 李华英, 李经球, 孔德金, 俞俊. La2O3助剂对Rh/SiO2催化CO加氢反应性能的影响[J]. 燃料化学学报,2020,48(4):483−489. doi: 10.3969/j.issn.0253-2409.2020.04.012SHI Zhang-ping, QI Xiao-lan, LI Xu-guang, LI Hua-ying, LI Jing-qiu, KONG De-jin, YU Jun. Effect of La2O3 addition on the catalytic performance of Rh/SiO2 for CO hydrogenation[J]. J Fuel Chem Technol,2020,48(4):483−489. doi: 10.3969/j.issn.0253-2409.2020.04.012 [21] WAN C, SUN L, XU L X, CHENG D G, CHEN F Q, ZHAN X L, YANG Y R. Novel NiPt alloy nanoparticle decorated 2D layered g-C3N4 nanosheets: a highly efficient catalyst for hydrogen generation from hydrous hydrazine[J]. J Mater Chem A,2019,7(15):8798−8804. doi: 10.1039/C9TA01535C [22] WAN C, ZHOU L, SUN L, XU L X, CHENG D G, CHEN F Q, ZHAN X L, YANG Y R. Boosting visible-light-driven hydrogen evolution from formic acid over AgPd/2D g-C3N4 nanosheets Mott-Schottky photocatalyst[J]. Chem Eng J,2020,396:125229. doi: 10.1016/j.cej.2020.125229 [23] LIU M M, ZHOU L, WAN C, YE M F, XU L X. Achieving complete hydrogen evolution from N2H4BH3 over mesoporous TiO2 immobilized NiPt alloy nanoparticles[J]. ChemSelect,2021,6:9185−9190. doi: 10.1002/slct.202102592 [24] YAO Q L, LU Z H, JIA Y S, CHEN X S, LIU X. In situ facile synthesis of Rh nanoparticles supported on carbon nanotubes as highly active catalysts for H2 generation from NH3BH3 hydrolysis[J]. Int J Hydrogen Energy,2014,40(5):2207−2215. [25] SHEN J, YANG L, HU K, LUO W, CHENG G Z. Rh nanoparticles supported on graphene as efficient catalyst for hydrolytic dehydrogenation of amine boranes for chemical hydrogen storage[J]. Int J Hydrogen Energy,2015,40(2):1062−1070. doi: 10.1016/j.ijhydene.2014.11.031 [26] CHANDRA M, XU Q. Room temperature hydrogen generation from aqueous ammonia-borane using noble metal nano-clusters as highly active catalysts[J]. J Power Sources,2007,168(1):135−142. doi: 10.1016/j.jpowsour.2007.03.015 [27] LU R, HU M, XU C, WANG Y, ZHANG Y, XU B, GAO D J, BI J, FAN G Y. Hydrogen evolution from hydrolysis of ammonia borane catalyzed by Rh/g-C3N4 under mild conditions[J]. Int J Hydrogen Energy,2018,43(14):7038−7045. doi: 10.1016/j.ijhydene.2018.02.148 [28] ALSAWAT M, ALTALHI T, SANTOS A, LOSIC D. Facile and controllable route for nitrogen doping of carbon nanotubes composite membranes by catalyst-free chemical vapour deposition[J]. Carbon,2016,106:295−305. doi: 10.1016/j.carbon.2016.05.043 [29] SOARES O S G P, ROCHA R P, GONCALVES A G, FIGUEIREDO J L, ÓRFÃO J J M, PEREIRA M F R. Highly active N-doped carbon nanotubes prepared by an easy ball milling method for advanced oxidation processes[J]. Appl Catal B: Environ,2016,192:296−303. doi: 10.1016/j.apcatb.2016.03.069 [30] SUN T, LU R, LONG Y, LI Q G, WU J, FAN G Y. Bamboo fungus-derived magnetic porous carbon encapsulated nickel stabilized Rh nanoparticles as efficient catalysts for hydrolytic dehydrogenation of ammonia borane[J]. Int J Hydrogen Energy,2021,46(69):34229−34238. doi: 10.1016/j.ijhydene.2021.07.222 [31] XU H, YU W L, ZHANG J K, ZHOU Z, ZHANG H X, GE H B, WANG G J, QIN Y. Rhodium nanoparticles confined in titania nanotubes for efficient hydrogen evolution from ammonia borane[J]. J Colloid Interface Sci,2022,609:755−763. doi: 10.1016/j.jcis.2021.11.078 [32] AKBAYRAK S, ÖZÇIFÇI Z, TABAKA A. Noble metal nanoparticles supported on activated carbon: Highly recyclable catalysts in hydrogen generation from the hydrolysis of ammonia borane[J]. J Colloid Interface Sci, 2019, 546: 324–332. [33] CHEN J Q, HU M, MING M, XU C L, WANG Y, ZHANG Y, WU J T, GAO D J, SI J, FAN G Y. Carbon-supported small Rh nanoparticles prepared with sodium citrate: Toward high catalytic activity for hydrogen evolution from ammonia borane hydrolysis[J]. Int J Hydrogen Energy,2018,43(5):2718−2725. doi: 10.1016/j.ijhydene.2017.12.079 [34] AKBAYRAK S, TONBUL Y, ÖZKAR S. Magnetically separable Rh0/Co3O4 nanocatalyst provides over a million turnovers in hydrogen release from ammonia borane[J]. ACS Sustainable Chem Eng,2020,8(10):4216−4224. doi: 10.1021/acssuschemeng.9b07402 [35] SUN Q M, WANG N, ZHANG T J, BAI R S, MAYORAL A, ZHANG P, ZHANG Q H, TERASAKI O, YU J H. Zeolite-encaged single-atom rhodium catalysts: Highly-efficient hydrogen generation and shape-selective tandem hydrogenation of nitroarenes[J]. Angew Chem Int Ed,2019,58:18570. doi: 10.1002/anie.201912367 [36] TONBUL Y, AKBAYRAK S, OZKAR S. Group 4 oxides supported Rhodium(0) catalysts in hydrolytic dehydrogenation of ammonia borane[J]. Int J Hydrogen Energy,2019,44(27):14164−14174. doi: 10.1016/j.ijhydene.2018.11.162 [37] ZHAO H, YU G, YUAN M, YANG J, XU D, DONG Z P. Ultrafine and highly dispersed platinum nanoparticles confined in a triazinyl-containing porous organic polymer for catalytic applications[J]. Nanoscale,2018,10(45):21466. doi: 10.1039/C8NR05756G [38] ZHANG H, HUANG M L, WEN J, LI Y P, LI A, ZHANG L H, ALI A M, LI Y D. Sub-3 nm Rh nanoclusters confined within a metal-organic framework for enhanced hydrogen generation[J]. Chem Commun,2019,55(32):4699. doi: 10.1039/C9CC00003H -




 下载:
下载: