Improved coking resistance of direct methane solid oxide fuel cell with Ni-BZCYYb anode
-
摘要:
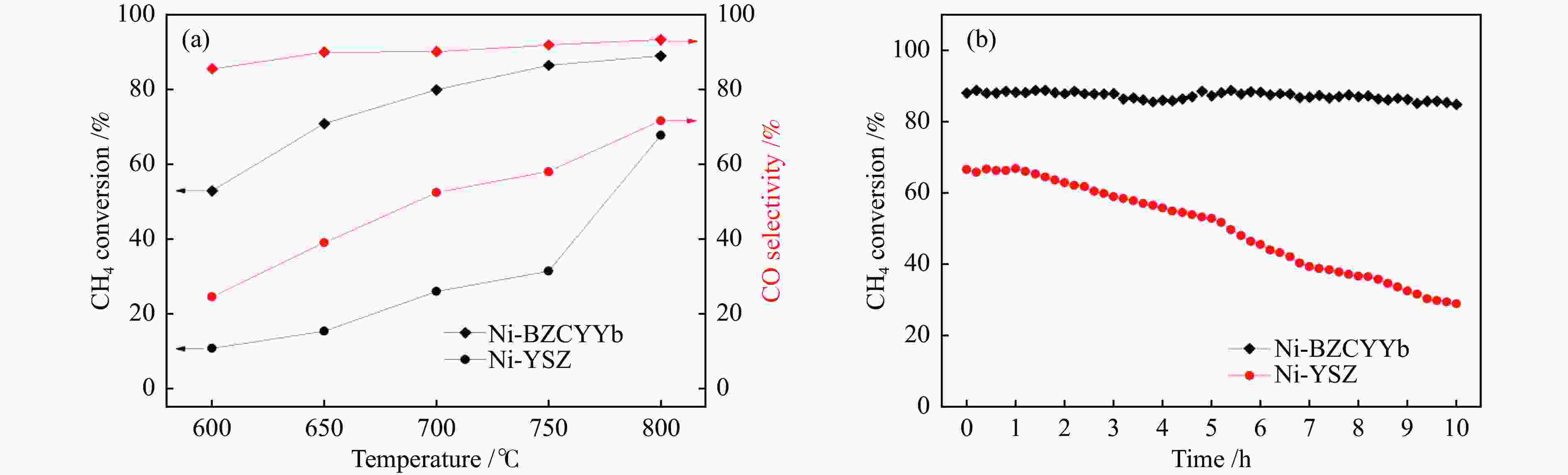
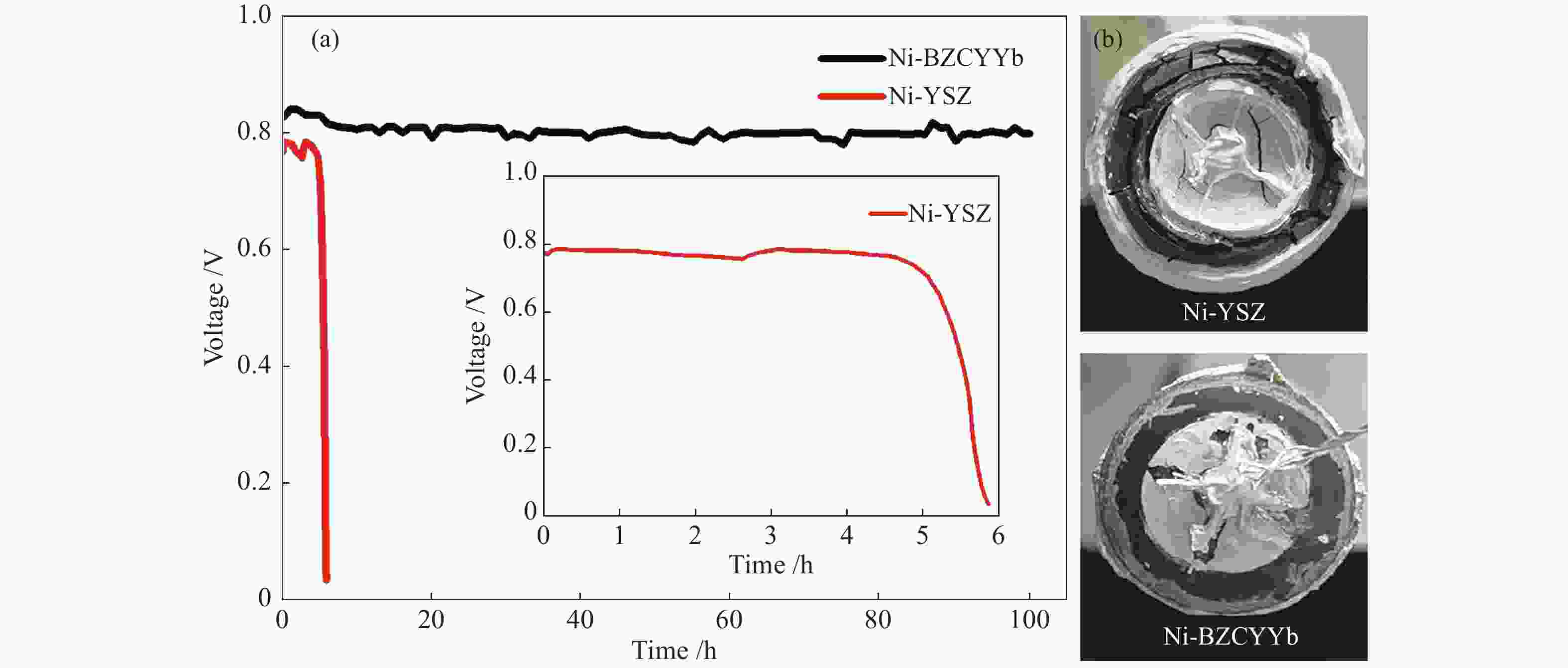
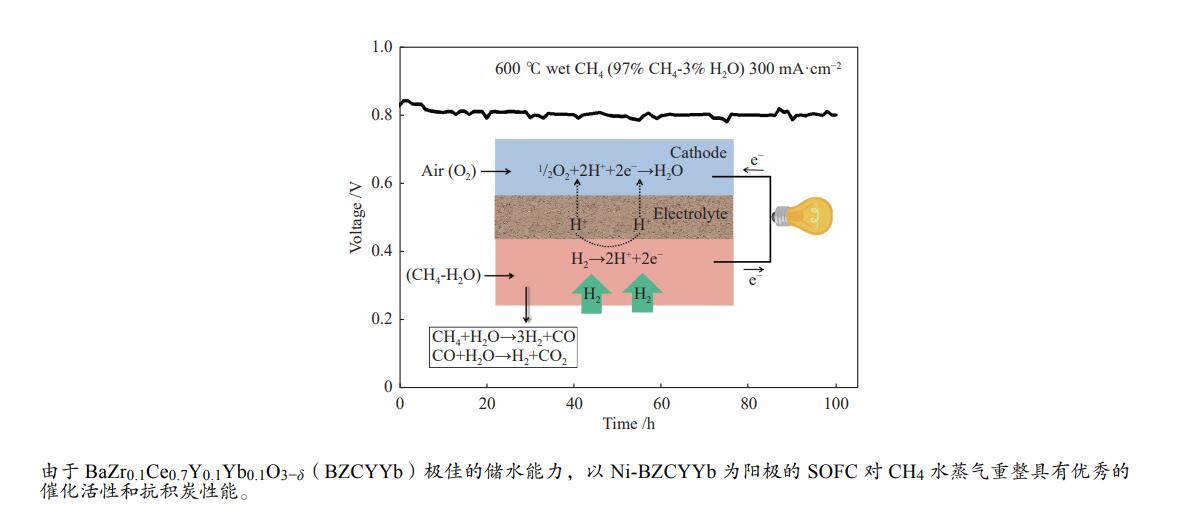
直接以甲烷(CH4)为燃料的固体氧化物燃料电池(Solid Oxide Fuel Cell,SOFC)具有操作系统简单、发电效率高、环境友好等优点,但传统镍基阳极使用CH4时极易产生积炭,导致电池性能下降甚至破裂。因此,如何有效抑制积炭的产生是目前镍基阳极面临的重要挑战。本文针对传统Ni - Y0.08Zr0.92O2−δ(Ni-YSZ)阳极使用CH4时存在严重的积炭行为,采用质子导体BaZr0.1Ce0.7Y0.1Yb0.1O3−δ(BZCYYb)代替氧离子导体YSZ,考查了Ni-BZCYYb对CH4水蒸气重整反应的催化活性和抗积炭性能,同时与Ni-YSZ进行比较。以Ni-BZCYYb为阳极的SOFC,在700–600 ℃、湿CH4(97% CH4-3% H2O)为燃料时取得了较好的电化学性能,同时,该电池在600 ℃、恒电流密度下稳定运行100 h电压没有明显降低。但以Ni-YSZ为阳极的SOFC在相同条件运行不到6 h电压降为零。通过对比表明,BZCYYb提高了阳极的抗积炭能力,Ni-BZCYYb可以应用于以CH4为燃料的SOFC中,是优异的抗积炭阳极材料。
-
关键词:
- SOFC /
- 甲烷重整 /
- Ni-BZCYYb阳极 /
- 抗积炭
Abstract:Solid oxide fuel cell (SOFC) is a promising power-generation device. Direct operation of SOFC on methane has several important advantages, such as simple system, high efficiency and low emissions. The challenge of the state-of-the-art nickel cermet anode is prone to coke formation when operating on methane, which may cause rapidly deteriorate of the performance and durability on SOFC. In this work, the anode Ni-BaZr0.1Ce0.7Y0.1Yb0.1O3−δ (BZCYYb) was investigated for wet methane (97% CH4-3% H2O) conversion in the temperature range of 700 to 600 ℃. The Ni-BZCYYb anode showed a good electrochemical performance for the steam reforming of methane. Furthermore, under a constant current density, a good operational stability was achieved at 600 ℃ for 100 h operating. For comparison, a conventional Ni-YSZ anode was also prepared, the voltage of cell dropped to zero after feeding wet CH4 for ~ 6 h. These results indicate that the Ni-BCZYYb is a good candidate as the anode in SOFC on methane fuel.
-
Key words:
- SOFC /
- methane reforming /
- Ni-BZCYYb anode /
- coking resistance
-
表 1 分别在650和700 ℃下、湿CH4中处理后的电池与新鲜电池之间的质量差
Table 1 Weight difference between the fresh cell and the treated cell in wet CH4 at 650 and 700 ℃
Temperature t/℃ m0 /g mt /g 100(mt−m0)/m0 /% Ni-BZCYYb Ni-YSZ Ni-BZCYYb Ni-YSZ Ni-BZCYYb Ni-YSZ 650 0.344 0.341 0.361 0.376 4.94 10.26 700 0.343 0.345 0.365 0.396 6.41 14.78 m0: the weight of fresh cells; mt: the weight of treated cells -
[1] ZHOU S, TONG Q, PAN X Z, CAO M, WANG H L, GAO J, QU X M. Research on low-carbon energy transformation of China necessary to achieve the Paris agreement goals: A global perspective[J]. Energy Econ,2021,95:105137. doi: 10.1016/j.eneco.2021.105137 [2] FAN L Y, LI C E, ARAVIND P V, CAI W W, HAN M F, BRABDON N. Methane reforming in solid oxide fuel cells: challenges and strategies[J]. J Power Sources,2022,538:231573. doi: 10.1016/j.jpowsour.2022.231573 [3] ORMEROD R M. Solid oxide fuel cells[J]. Chem Soc Rev,2003,32:17−28. doi: 10.1039/b105764m [4] TURGUT M G. Comprehensive review of methane conversion in solid oxide fuel cells: Prospects for efficient electricity generation from natural gas[J]. Prog Energy Combust Sci,2016,54:1−64. doi: 10.1016/j.pecs.2015.10.004 [5] RAMADHANI F, HUSSAIN M A, MOKHLIS H, HAJIMOLANA S. Optimization strategies for solid oxide fuel cell (SOFC) application: a literature survey[J]. Renewable Sustainable Energy Rev,2017,76:460−484. doi: 10.1016/j.rser.2017.03.052 [6] SINGH M, ZAPPA D, COMINI E. Solid oxide fuel cell: Decade of progress, future perspectives and challenges[J]. Int J Hydrogen Energy,2021,46:27643−27674. doi: 10.1016/j.ijhydene.2021.06.020 [7] LI Q H, WANG X, LIU C Z, YANG X W, LI C, JIA L C, LI J. A direct-methane solid oxide fuel cell with a functionally engineered Ni-Fe metal support[J]. J Power Sources,2022,537:231533. doi: 10.1016/j.jpowsour.2022.231533 [8] SENGODANA S, LAN R, HUMPHREYSA J, DU D, XU W, WANG H T, TAO S W. Advances in reforming and partial oxidation of hydrocarbons for hydrogen production and fuel cell applications[J]. Renewable Sustainable Energy Rev,2018,82:761−780. doi: 10.1016/j.rser.2017.09.071 [9] WEI T, LIU B, JIA L C, LI R H. Perovskite materials for highly efficient catalytic CH4 fuel reforming in solid oxide fuel cell[J]. Int J Hydrogen Energy,2021,46:24441−24460. doi: 10.1016/j.ijhydene.2021.05.021 [10] SAADABAD S A, THATTAI A T, FAN L Y, LINDEBOOM R E F, SPANJERS H, ARAVIND P V. Solid oxide fuel cells fuelled with biogas: Potential and constraints[J]. Renewable Energy,2019,134:194−214. doi: 10.1016/j.renene.2018.11.028 [11] SHAO Z P, ZHANG C M, WANG W, SU C, ZHOU W, ZHU Z H, PARK H J, KWAK C. Electric power and synthesis gas co-generation from methane with zero waste gas emission[J]. Angew Chem Int Ed,2011,50:1792−1797. doi: 10.1002/anie.201006855 [12] BOLDRIN P, ENRIQUE R, UIZ-TREJO E, MERMELSTEIN J, BARNDON N P. Strategies for carbon and sulfur tolerant solid oxide fuel cell materials, Incorporating lessons from heterogeneous catalysis[J]. Chem Rev,2016,116:13633−13684. doi: 10.1021/acs.chemrev.6b00284 [13] ABDELKAREEM M A, TANVEER W H, SAYED E T, ASSAD M E H, ALLAGUI A CHA S W. On the technical challenges affecting the performance of direct internal reforming biogas solid oxide fuel cells[J]. Renewable Sustainable Energy Rev,2019,101:361−375. doi: 10.1016/j.rser.2018.10.025 [14] WANG X X, WEI K W, YAN S L, WU Y J, KANG J H, FENG P Z, WANG S R, ZHOU F B, LING Y H. Efficient and stable conversion of oxygen-bearing low-concentration coal mine methane by the electrochemical catalysis of SOFC anode: from pollutant to clean energy[J]. Appl Catal B: Environ,2020,268:118413. doi: 10.1016/j.apcatb.2019.118413 [15] WEI T, QIU P, YANG J, JIA L C, CHI B, PU J, LI J. High-performance direct carbon dioxide-methane solid oxide fuel cell with a structure-engineered double-layer anode[J]. J Power Sources,2021,484:229199. doi: 10.1016/j.jpowsour.2020.229199 [16] WANG W, SU C, WU Y Z, RAN R, SHAO Z P. Progress in solid oxide fuel cells with nickel-based anodes operating on methane and related fuels[J]. Chem Rev,2013,113:8104−8151. doi: 10.1021/cr300491e [17] HANNA J, LEE W Y, SHI Y, GHONIEM A F. Fundamentals of electro-and thermochemistry in the anode of solid-oxide fuel cells with hydrocarbon and syngas fuels[J]. Prog Energy Combust Sci,2014,40:74−111. doi: 10.1016/j.pecs.2013.10.001 [18] RAFIQUE M, NAWAZ H, RAFIQUE M S, TAHIR M B, NABI G, KHALID N R. Material and method selection for efficient solid oxide fuel cell anode: recent advancements and reviews[J]. Int J Hydrogen Energy,2019,43:2423−2446. [19] LV X Q, CHEN H L, ZHOU W, CHEN F Q, LI S D, SHAO Z P. Direct-methane solid oxide fuel cells with an in situ formed Ni-Fe alloy composite catalyst layer over Ni-YSZ anodes[J]. Renewable Energy,2020,150:334−341. doi: 10.1016/j.renene.2019.12.126 [20] ZHU W Z, DEEVI S C. A review on the status of anode materials for solid oxide fuel cells[J]. Mater Sci Eng A,2003,362:228−239. doi: 10.1016/S0921-5093(03)00620-8 [21] TAILLADES G, PERS P, MAO V, TAILLADES M. High performance anode-supported proton ceramic fuel cell elaborated by wet powder spraying[J]. Int J Hydrogen Energy,2016,41:12330−12336. doi: 10.1016/j.ijhydene.2016.05.094 [22] YANG L, WANG S Z, BLINN K, LIU M F, LIU Z, CHEN Z, LIU M L. Enhanced sulfur and coking tolerance of a mixed ion conductor for SOFCs: BaZr0.1Ce0.7Y0.1Yb0.1O3−δ[J]. Science,2009,326:126−129. doi: 10.1126/science.1174811 [23] HUA B, LI M, PU J, CHI B, LI J. BaZr0.1Ce0.7Y0.1Yb0.1O3-δ enhanced coking-free on-cell reforming for direct-methane solid oxide fuel cells[J]. J Mater Chem A,2014,2:12576−12582. doi: 10.1039/C4TA01989J [24] MA J J, JIANG C R, CONNOR P A, CASSIDY M, IRVINE T S. Highly efficient, coking-resistant SOFCs for energy conversion using biogas fuels[J]. J Mater Chem A,2015,3:19068−19076. doi: 10.1039/C5TA06421J [25] ARPORNWICHANOPA A, PATCHARAVORACHO Y. Investigation of a proton-conducting SOFC with internal autothermal reforming of methane[J]. Chem Eng Res Des,2013,9:1508−1516. [26] KAN H, LEE H. Sn-doped Ni/YSZ anode catalysts with enhanced carbon deposition resistance for an intermediate temperature SOFC[J]. Appl Catal B: Environ,2010,97:108−114. doi: 10.1016/j.apcatb.2010.03.029 [27] WANG W, SU C, RAN R, ZHAO B, SHAO Z P, TADE M O, LIU S M. Nickel-based anode with water storage capability to mitigate carbon deposition for direct ethanol solid oxide fuel cells[J]. ChemSusChem,2014,7:1719−1728. doi: 10.1002/cssc.201301341 [28] YANG L, CHOI Y M, QIN W T, CHEN H Y, BLIIN K, LIU M F, LIU P, BAI J M, TYSON T A, LIU M L. Promotion of water-mediated carbon removal by nanostructured barium oxide/nickel interfaces in solid oxide fuel cells[J]. Nat Commun,2011,2:357. doi: 10.1038/ncomms1359 [29] GUO Y M, RAN R, SHAO Z P. Zirconium doping effect on the performance of proton-conducting BaZryCe0.8−yY0.2O3−δ (0.0≤y≤0.8) for fuel cell applications[J]. J Power Sources,2009,193:400−407. doi: 10.1016/j.jpowsour.2009.03.044 [30] CHANG H, CHEN H L, SHAO Z P, SHI J, BAI J P, LI S D. In situ fabrication of (Sr, La)FeO4 with CoFe alloy nanoparticles as an independent catalyst layer for direct methane-based solid oxide fuel cells with a nickel cermet anode[J]. J Mater Chem A,2016,4:13997−14007. doi: 10.1039/C6TA04639H [31] QIU P, SUN S C, YANG X, CHEN F L, XIONG C Y, JIA L C, LI J. A review on anode on-cell catalyst reforming layer for direct methane solid oxide fuel cells[J]. Int J Hydrogen Energy,2021,46:25208−25224. doi: 10.1016/j.ijhydene.2021.05.040 [32] 姚瑶, 蔡佩君, 王绍荣. 沼气燃料固体氧化物燃料电池阳极相关问题综述[J]. 陶瓷学报,2021,42(4):560−568. doi: 10.13957/j.cnki.tcxb.2021.04.004YAO Yao, CAI Pei-jun, WANG Shao-rong. Overview in anode related problems of solid oxide fuel cells fed with biogas[J]. J Ceram,2021,42(4):560−568. doi: 10.13957/j.cnki.tcxb.2021.04.004 [33] MAMONTOV E, EGAMI T, BREZNY R, KORANNE M, TYAGI S. Lattice defects and oxygen storage capacity of nanocrystalline ceria and ceria-zirconia[J]. J Phys Chem B,2000,104:11110−11116. doi: 10.1021/jp0023011 [34] ZHANG Y, XIE D, CHI B, PU J, LI J, YAN D. Basic properties of proton conductor BaZr0.1Ce0.7Y0.1Yb0.1O3−δ (BZCYYb) material[J]. Asia-Pac J Chem Eng,2019,14:e2322. doi: 10.1002/asia.201900550 -




 下载:
下载: