Role of interfacial effects in the oxidation of toluene by MnOx-modified CeO2 nanocubes
-
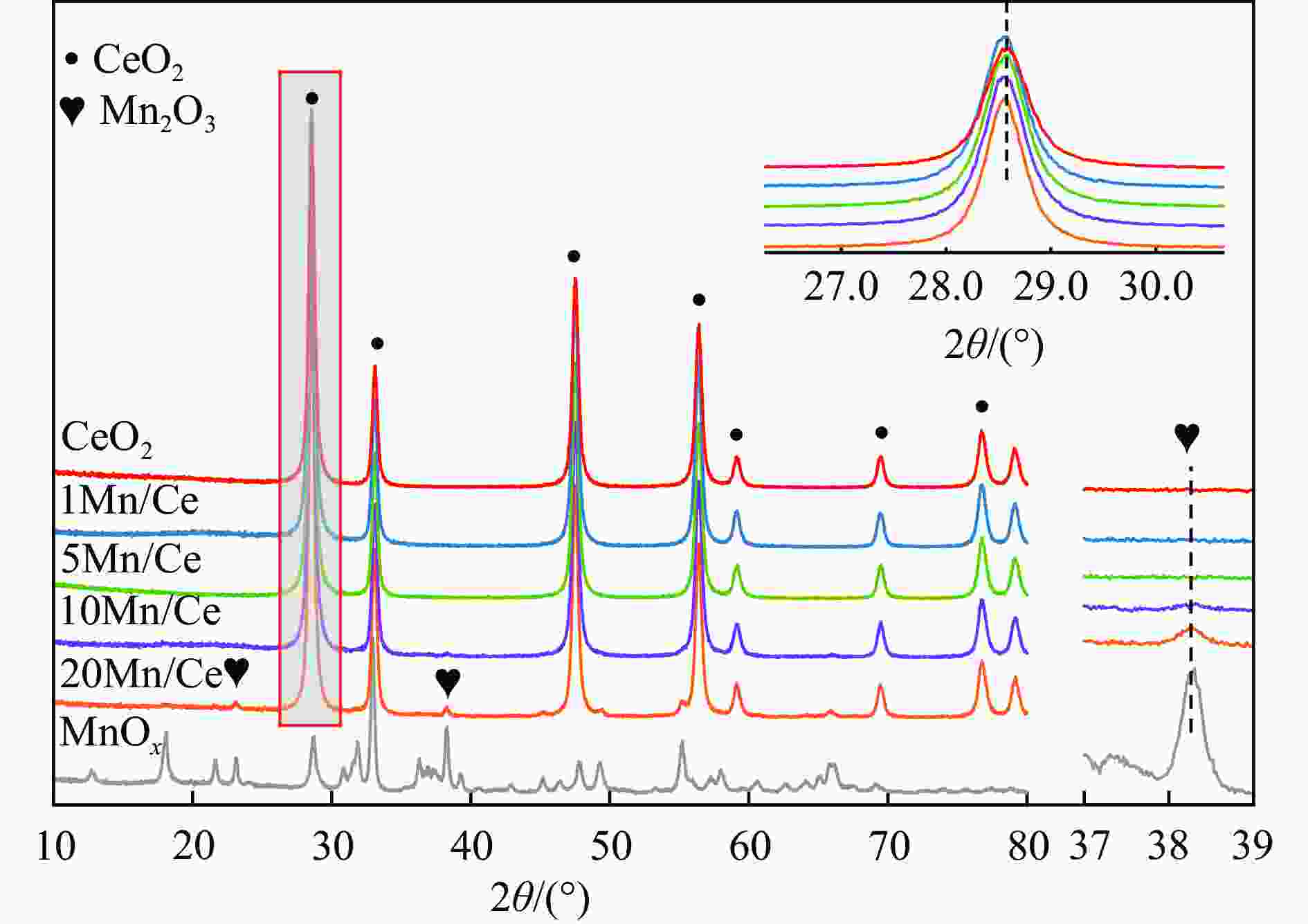
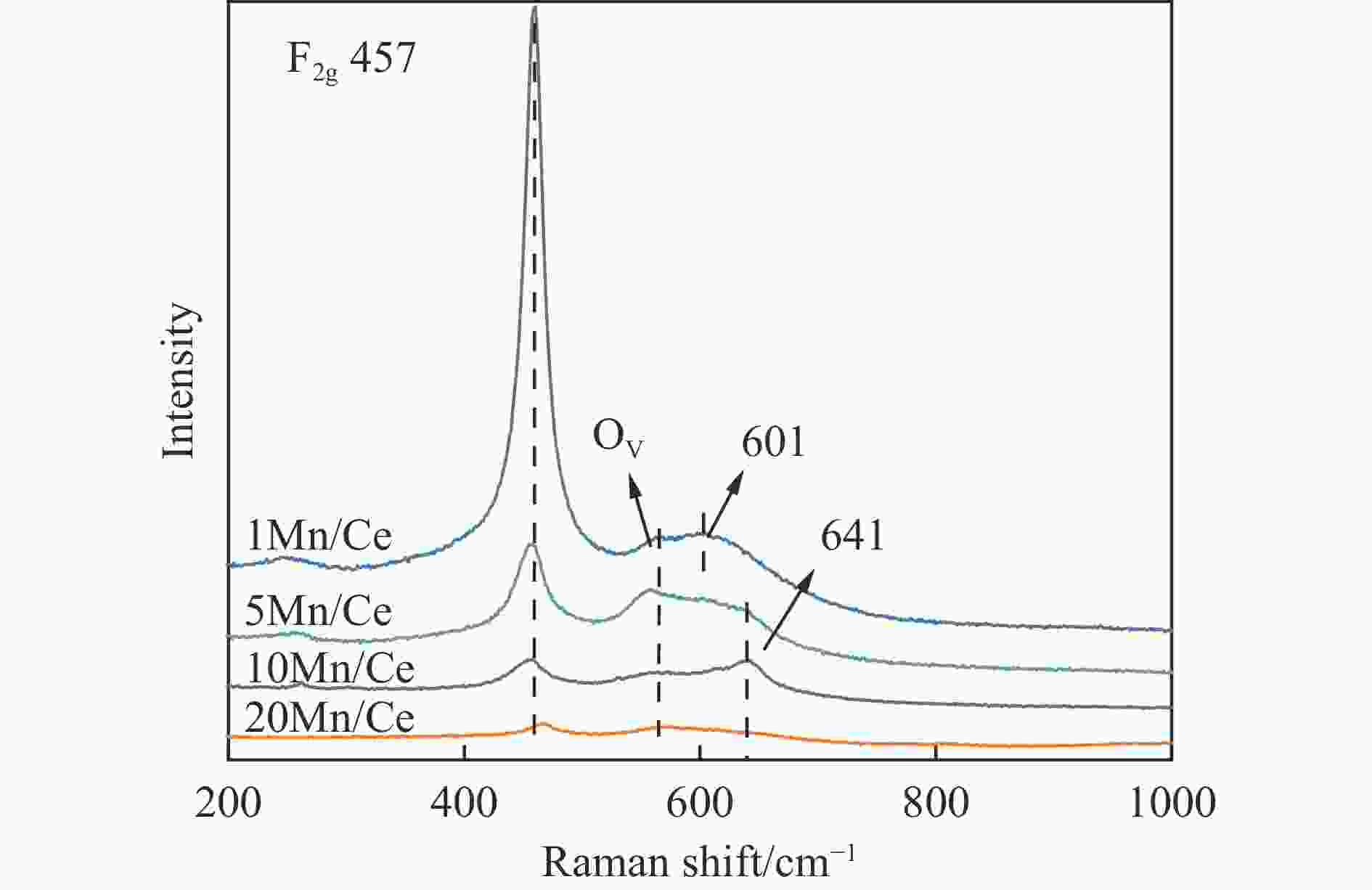
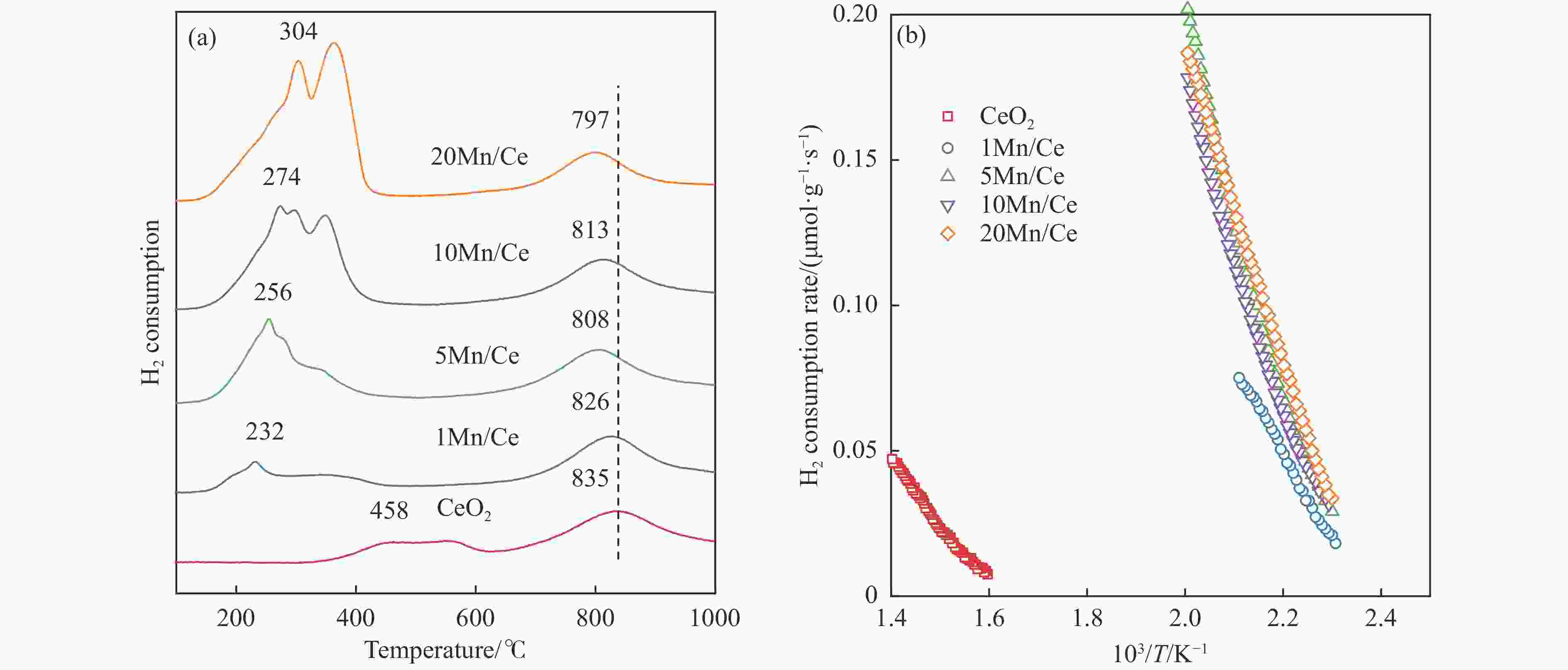
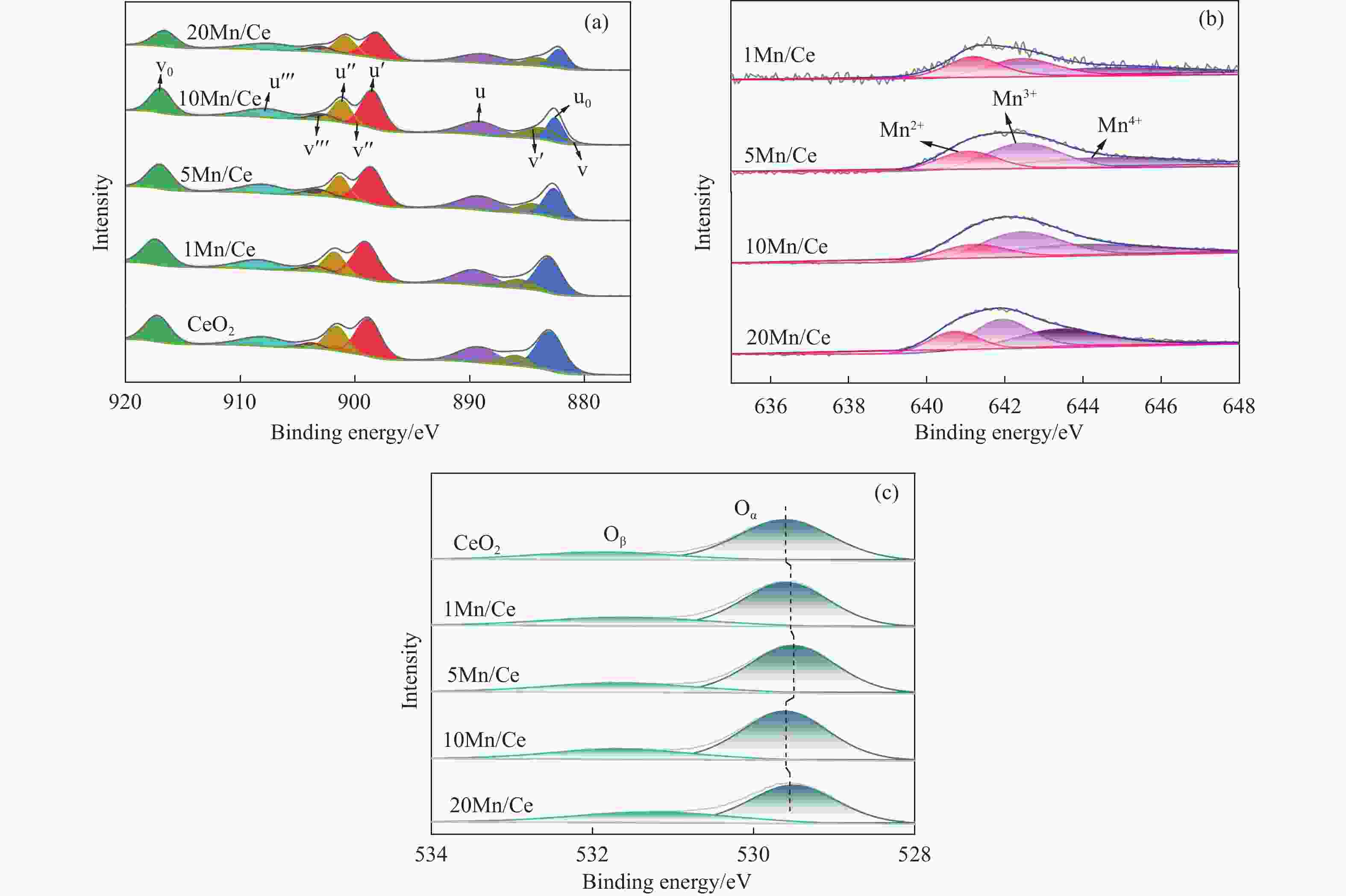
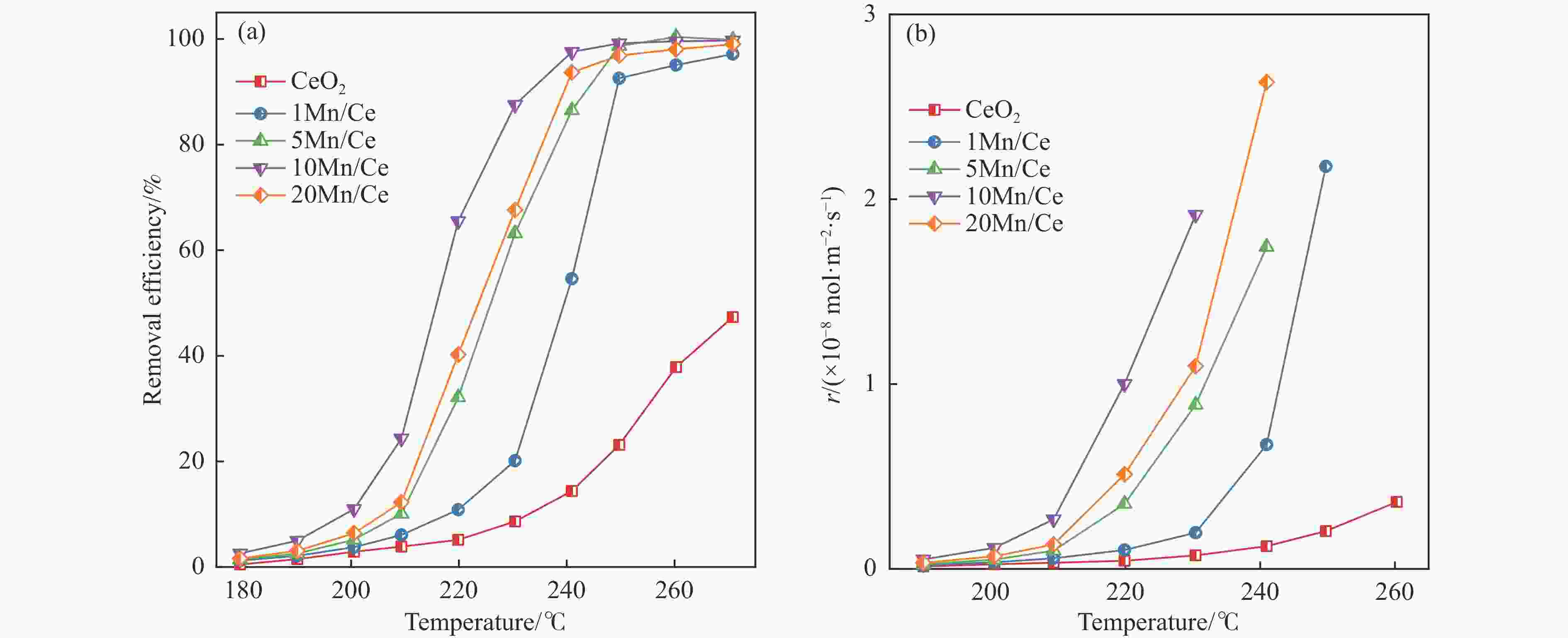
摘要: 本研究,通过水热-浸渍两步法成功制备了不同Mn负载量的二元xMn/Ce(xMnOx/CeO2)催化剂,并评估了这些催化剂在甲苯催化氧化反应中的性能。研究结果表明,引入MnOx能显著提高催化剂的甲苯氧化活性。特别是当Mn负载量为10%(10Mn/Ce)时,在气体空速为60000 mL/(g·h)的条件下,t90(甲苯转化率达到90%时的温度)仅为233 ℃,显示出最优的甲苯催化氧化活性。这一结果说明,适量的MnOx加入能够显著提高催化剂的催化性能。通过X射线衍射(XRD)、拉曼光谱(Raman)、透射电子显微镜(TEM)、程序升温还原(H2-TPR)和X射线光电子能谱(XPS)等表征手段,发现MnOx的加入在MnOx与CeO2之间形成了界面效应,这显著改变了Mn/Ce催化剂的物理化学性质。由于界面效应的作用,不仅提高了10Mn/Ce催化剂中Ce3+、Mn3+离子的浓度以及氧空位的浓度,而且还降低了催化剂表面Ce−O键强度,使得表面晶格氧更易于参与甲苯的催化氧化,提升了催化剂的氧化还原性能,从而促进了甲苯的催化氧化。本研究不仅成功制备了具有优异甲苯氧化活性的Mn/Ce催化剂,而且揭示了其背后的界面效应机制,为VOCs高效氧化催化剂设计与制备提供了简单有效的方法与思路。Abstract: Toluene, a common volatile organic compounds (VOCs), can have adverse effects on the natural environment as well as on human health. Catalytic oxidation technology can remove toluene economically and efficiently, and the key to this technology is the development of efficient catalysts. In order to improve the oxidation efficiency of toluene, it is of great significance to explore and study new efficient catalysts. In this study, binary xMn/Ce (xMnOx/CeO2) catalysts with different Mn loadings were successfully prepared by a two-step hydrothermal-impregnation method, and the performance of these catalysts was evaluated in the catalytic oxidation reaction of toluene. The results showed that the introduction of MnOx significantly increased the toluene oxidation activity of the catalysts. In particular, when the Mn loading was 10% (10Mn/Ce), the t90 (temperature at which toluene conversion reached 90%) was only 233 ℃ at gas hourly space velocity of 60000 mL/(g·h), showing optimal toluene catalytic oxidation activity. This result suggests that the addition of moderate amount of MnOx can significantly improve the catalytic performance of the catalysts. By characterization means such as X-ray diffraction (XRD), Raman, transmission electron microscopy (TEM), programmed temperature-raising reduction (H2-TPR), and X-ray photoelectron spectroscopy (XPS), we found that the incorporation of MnOx creates an interfacial effect between MnOx and CeO2, which significantly alters the physicochemical properties of the Mn/Ce catalysts. Due to the interfacial effect, the concentration of Ce3+ and Mn3+ ions and the oxygen vacancy in the 10Mn/Ce catalyst were not only increased, but also the strength of Ce−O bond on the catalyst surface was reduced, which made it easier for surface lattice oxygen to participate in the catalytic oxidation of toluene, and enhanced the redox performance of the catalyst, thus promoting the catalytic oxidation of toluene. In this study, we not only successfully prepared Mn/Ce catalysts with excellent toluene oxidation activity, but also revealed the mechanism of interfacial effect behind it, which provides a simple and effective method and idea for the design and preparation of efficient oxidation catalysts for VOCs.
-
表 1 Mn/Ce催化剂与同类型催化剂活性对比
Table 1 Comparison of the activity of Mn/Ce catalysts with the same type of catalysts
Catalysts Preparation method Reaction condition t90/℃ Reference K0.1-Mn-Ce sol-gel 1.0×10−3 toluene 20% O2/N2 space velocity: 60000 h−1 229 [20] MnCe/ZrO impregnation 0.5×10−3 toluene 20% O2/N2 space velocity: 50000 h−1 290 [21] MC-TPAOH sol-gel 1.0×10−3 toluene 20% O2/N2 space velocity: 60000 h−1 221 [22] MnCe-OH impregnation 1.0×10−3 toluene 20% O2/N2 space velocity: 36000 h−1 237 [23] 10Mn/Ce Hydrothermal-impregnation 1.0×10−3 toluene 20% O2/N2 space velocity: 60000 h−1 233 this work 表 2 CeO2和xMn/Ce催化剂的表面化学组成
Table 2 Surface chemical composition of CeO2 and xMn/Ce catalysts
Catalyst AD/
AF2gaH2
consumptionCe3+/
(Ce3++Ce4+)bMn3+/Mn4+b Oα/Oβb CeO2 − 2.9 0.09 − 0.29 1Mn/Ce 0.13 3.5 0.11 0.46 0.25 5Mn/Ce 0.85 4.2 0.13 0.51 0.38 10Mn/Ce 1.81 4.9 0.16 1.61 0.35 20Mn/Ce 0.92 6.0 0.13 1.22 0.64 a: 通过Raman图谱计算得到;b: 通过XPS图谱定量分析得到。 表 3 CeO2和xMn/Ce催化剂的孔结构
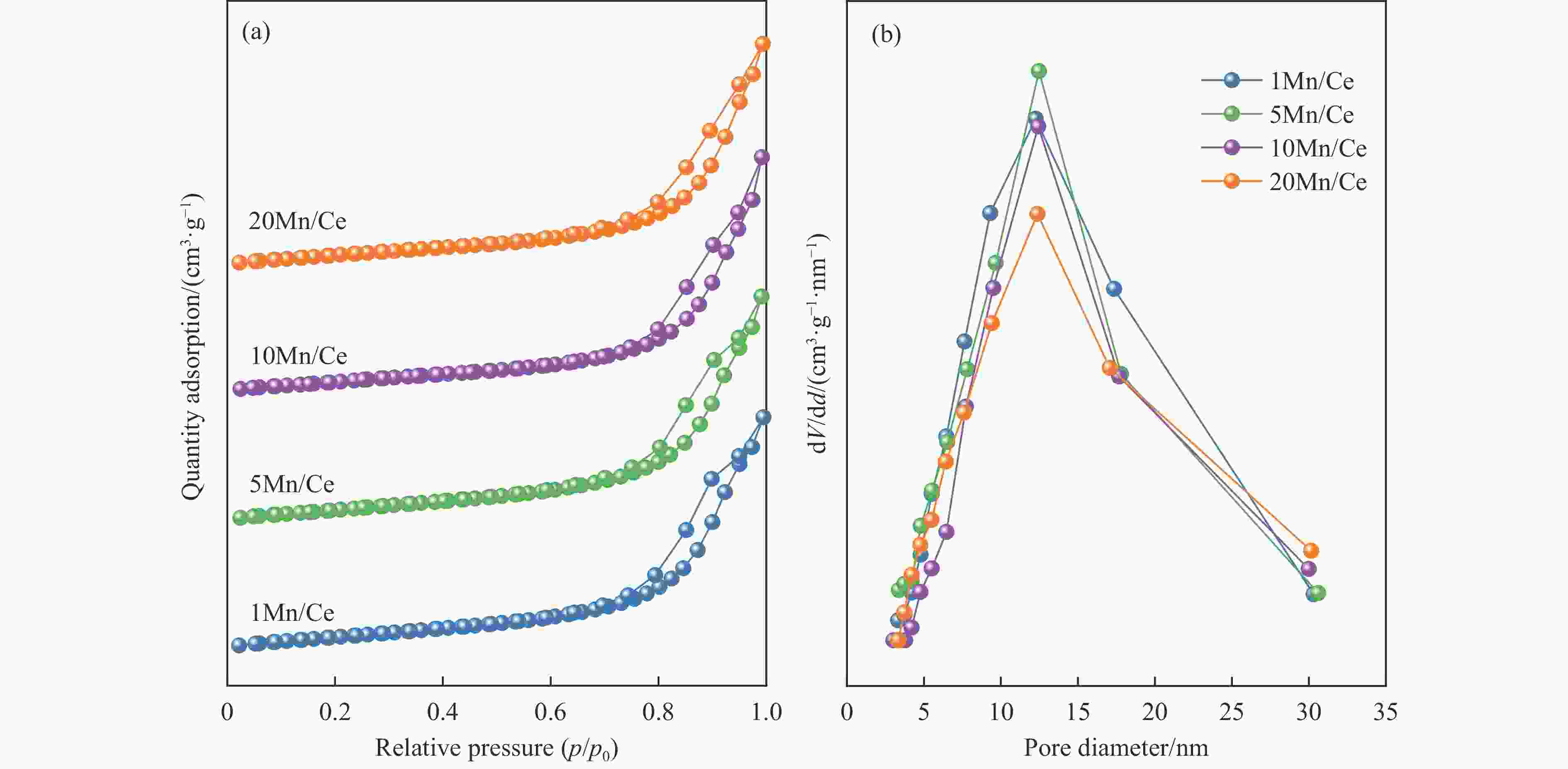
Table 3 Pore structure of CeO2 and xMn/Ce catalysts
Catalyst BET surface area/(m2·g−1) Pore volume/(cm3·g−1) Pore size/nm CeO2 35 − − 1Mn/Ce 46 0.18 12.26 5Mn/Ce 45 0.17 12.47 10Mn/Ce 44 0.18 12.42 20Mn/Ce 41 0.17 12.38 -
[1] 刘旭, 黄妍, 赵令葵, 等. 负载型CuMn2O4催化剂同时去除甲苯与NO x性能及机理研究[J]. 燃料化学学报(中英文),2023,51(12):1856−65.LIU Xu, HUANG Yan, ZHAO Lingkui, et al. Study on performance and mechanism of CuMn2O4 supported catalyst for simultaneous removal of toluene and NO x[J]. J Fuel Chem Technol,2023,51(12):1856−65. [2] HU F, CHEN J, ZHAO S, et al. Toluene catalytic combustion over copper modified Mn0.5Ce0.5O x solid solution sponge-like structures[J]. Appl Catal A-Gen,2017,540:57−67. doi: 10.1016/j.apcata.2017.04.010 [3] 刘宗耀, 曾永辉, 刘俊伟, 等. 挥发性有机物末端治理技术研究进展[J]. 现代化工,2022,42(3):74−8+84.LIU Zongyao, ZENG Yonghui, LIU Junwei, et al. Research progress of terminal treatment technology of volatile organic compounds[J]. Modern Chemical Industry,2022,42(3):74−8+84. [4] 李长英, 陈明功, 盛楠, 等. 挥发性有机物处理技术的特点与发展[J]. 化工进展,2016,35(3):917−25.LI Changyin, CHEN Minggong, SHEN Nan, et al. Characteristics and development of volatile organic compounds treatment technology[J]. Chemical Industry and Engineering Progress,2016,35(3):917−25. [5] 权燕红, 苗超, 李涛, 等. 不同制备方法对氧化铈结构及甲苯催化燃烧性能的影响燃料化学学报[J]. 燃料化学学报,2021,49(2):211−9. doi: 10.1016/S1872-5813(21)60014-2QUAN Yanhong, MIAO Chao, LI Tao, et al. Effects of different preparation methods on the structure of cerium oxide and catalytic combustion performance of toluene[J]. J Fuel Chem Technol,2021,49(2):211−9. doi: 10.1016/S1872-5813(21)60014-2 [6] ZHU D, HUANG Y, LI R, et al. Constructing active Cu2+-O-Fe3+ sites at the CuO-Fe3O4 interface to promote activation of surface lattice oxygen[J]. Environ. Sci. Technol,2023,57(45):17598−609. doi: 10.1021/acs.est.3c05431 [7] YANG C, MIAO G, PI Y, et al. Abatement of various types of VOCs by adsorption/catalytic oxidation: A review[J]. Chem. Eng. J,2019,370:1128−53. doi: 10.1016/j.cej.2019.03.232 [8] ZHANG K, DING H, PAN W, et al. Research progress of a composite metal oxide oatalyst for VOC degradation[J]. Environ. Sci. Technol,2022,56(13):9220−36. doi: 10.1021/acs.est.2c02772 [9] WANG Q, YEUNG K L, BAñARES M A. Ceria and its related materials for VOC catalytic combustion: A review[J]. Catal. Today,2020,356:141−54. doi: 10.1016/j.cattod.2019.05.016 [10] HU F, CHEN J, PENG Y, et al. Novel nanowire self-assembled hierarchical CeO2 microspheres for low temperature toluene catalytic combustion[J]. Chem. Eng. J,2018,331:425−34. doi: 10.1016/j.cej.2017.08.110 [11] HAO Z-R, FENG S, XING Y-Y, et al. Experimental study of Fe modified Mn/CeO2 catalyst for simultaneous removal of NO and toluene at low temperature[J]. J. Fuel Chem. Technol,2023,51(12):1866−78. doi: 10.1016/S1872-5813(23)60358-5 [12] ZHOU S, FANG J, CHAO K, et al. Construction of Pt decorated CeO2 nanocomposite for efficient VOCs catalytic oxidation and atmospheric total organic carbon dictation[J]. Catal Commun,2023,177:106663. doi: 10.1016/j.catcom.2023.106663 [13] CHEN W, YANG S, LIU H, et al. Single-atom Ce-modified alpha-Fe2O3 for selective catalytic reduction of NO with NH3[J]. Environ. Sci. Technol,2022,56(14):10442−53. doi: 10.1021/acs.est.2c02916 [14] ARENA F. Multipurpose composite MnCeO xcatalysts for environmental applications[J]. Catal. Sci. Technol,2014,4(7):1890−8. doi: 10.1039/C4CY00022F [15] 李安明, 卫广程, 郝乔慧, 等. Mn含量对CeO2-ZrO2-MnO x催化剂甲苯氧化净化性能的影响燃料化学学报[J]. 燃料化学学报,2020,48(2):231−9.LI Anming, WEI Guangchen, HAO Qiaohui, et al. Effect of Mn content on toluene oxidation purification performance of CeO2-ZrO2-MnO x catalyst[J]. J Fuel Chem Technol,2020,48(2):231−9. [16] PUTLA S, AMIN M H, REDDY B M, et al. MnO x nanoparticle-dispersed CeO2 nanocubes: A remarkable heteronanostructured system with unusual structural characteristics and superior catalytic performance[J]. ACS Appl. Mater. Interfaces,2015,7(30):16525−35. doi: 10.1021/acsami.5b03988 [17] LI B, HUANG Q, YAN X K, et al. Low-temperature catalytic combustion of benzene over Ni–Mn/CeO2/cordierite catalysts[J]. J Ind Eng Chem,2014,20(4):2359−63. doi: 10.1016/j.jiec.2013.10.013 [18] HU F, PENG Y, CHEN J, et al. Low content of CoO x supported on nanocrystalline CeO2 for toluene combustion: The importance of interfaces between active sites and supports[J]. Appl. Catal. B,2019,240:329−36. doi: 10.1016/j.apcatb.2018.06.024 [19] WU P, JIN X, QIU Y, et al. Recent progress of thermocatalytic and photo/thermocatalytic oxidation for VOCs purification over manganese-based oxide catalysts[J]. Environ. Sci. Technol,2021,55(8):4268−86. doi: 10.1021/acs.est.0c08179 [20] YANG B, ZENG Y, ZHANG M, et al. Highly efficient K-doped Mn–Ce catalysts with strong K–Mn–Ce interaction for toluene oxidation[J]. Journal of Rare Earths,2023,41(3):374−80. doi: 10.1016/j.jre.2022.03.007 [21] LI L, SONG L, FEI Z, et al. Effect of different supports on activity of Mn–Ce binary oxides catalysts for toluene combustion[J]. Journal of Rare Earths,2022,40(4):645−51. doi: 10.1016/j.jre.2021.02.004 [22] CHEN Z, ZHOU J, ZHUGE X, et al. Catalytic oxidation of toluene using layer-modified Mn-Ce solid solution with high specific surface area[J]. Journal of Environmental Chemical Engineering,2023,11(6):111427. doi: 10.1016/j.jece.2023.111427 [23] LI L, ZHANG C, YAN J, et al. Distinctive bimetallic oxides for enhanced catalytic toluene combustion: Insights into the tunable fabrication of Mn−Ce hollow structure [J]. 2020, 12(10): 2872-9. [24] LUO Y, DENG Y Q, MAO W, et al. Probing the surface structure of α-Mn2O3 nanocrystals during CO oxidation by operando Raman spectroscopy[J]. J. Phys. Chem. C,2012,116(39):20975−81. doi: 10.1021/jp307637w [25] MARROCCHELLI D, BISHOP S R, KILNER J. Chemical expansion and its dependence on the host cation radius[J]. J. Mater,2013,1(26):7673−80. [26] VECCHIETTI J, BONIVARDI A, XU W Q, et al. Understanding the role of oxygen vacancies in the water gas shift reaction on ceria-supported platinum catalysts[J]. ACS Catal,2014,4(6):2088−96. doi: 10.1021/cs500323u [27] MARROCCHELLI D, BISHOP S R, KILNER J. Chemical expansion and its dependence on the host cation radius [J]. J. Mater, 2013, 1(26). [28] ARTIGLIA L, AGNOLI S, PAGANINI M C, et al. TiO2@CeO x core-shell nanoparticles as artificial enzymes with peroxidase-like activity[J]. ACS Appl. Mater,2014,6(22):20130−6. doi: 10.1021/am5057129 [29] DU X J, ZHANG D S, SHI L Y, et al. Morphology dependence of catalytic properties of Ni/CeO2 nanostructures for carbon dioxide reforming of methane[J]. J. Phys. Chem. C,2012,116(18):10009−16. doi: 10.1021/jp300543r [30] CAO H Q, WU X M, WANG G H, et al. Biomineralization strategy to α-Mn2O3 hierarchical nanostructures[J]. J. Phys. Chem. C,2012,116(39):21109−15. doi: 10.1021/jp306984c [31] PUTLA S, AMIN M H, REDDY B M, et al. MnO x nanoparticle-dispersed CeO2 nanocubes: A remarkable heteronanostructured system with unusual structural characteristics and superior catalytic performance[J]. ACS Appl. Mater,2015,7(30):16525−35. doi: 10.1021/acsami.5b03988 [32] SHEN Y, DENG J, HU X, et al. Expediting toluene combustion by harmonizing the Ce-O strength over Co-doped CeZr oxide catalysts[J]. Environ. Sci. Technol,2023,57(4):1797−806. doi: 10.1021/acs.est.2c07853 [33] GAO W, ZHANG Z Y, LI J, et al. Surface engineering on CeO2 nanorods by chemical redox etching and their enhanced catalytic activity for CO oxidation[J]. Nanoscale,2015,7(27):11686−91. doi: 10.1039/C5NR01846C [34] WU Z L, LI M J, HOWE J, et al. Probing defect Sites on CeO2 nanocrystals with well-defined surface planes by Raman spectroscopy and O2 adsorption[J]. Langmuir,2010,26(21):16595−606. doi: 10.1021/la101723w [35] SUDARSANAM P, MALLESHAM B, REDDY P S, et al. Nano-Au/CeO2 catalysts for CO oxidation: Influence of dopants (Fe, La and Zr) on the physicochemical properties and catalytic activity[J]. Appl. Catal. B,2014,144:900−8. doi: 10.1016/j.apcatb.2013.08.035 [36] SAYLE T X T, PARKER S C, CATLOW C R A. The role of oxygen vacancies on ceria surfaces in the oxidation of carbon monoxide[J]. Surf Sci (Netherlands),1994,316(3):329−36. doi: 10.1016/0039-6028(94)91225-4 [37] LóPEZ J M, GILBANK A L, GARCíA T, et al. The prevalence of surface oxygen vacancies over the mobility of bulk oxygen in nanostructured ceria for the total toluene oxidation[J]. Appl. Catal. B,2015,174:403−12. [38] XU J H, HARMER J, LI G Q, et al. Size dependent oxygen buffering capacity of ceria nanocrystals[J]. Chem. Commun,2010,46(11):1887−9. doi: 10.1039/B923780A [39] BORCHERT H, FROLOVA Y V, KAICHEV V V, et al. Electronic and chemical properties of nanostructured cerium dioxide doped with praseodymium[J]. J. Phys. Chem. B,2005,109(12):5728−38. doi: 10.1021/jp045828c [40] PFAU A, SCHIERBAUM K D. The electronic structure of stoichiometric and reduced CeO2 surfaces: an XPS, UPS and HREELS study[J]. Surf Sci (Netherlands),1994,321(1-2):71−80. doi: 10.1016/0039-6028(94)90027-2 [41] LIU Z M, ZHU J Z, LI J H, et al. Novel Mn-Ce-Ti mixed-oxide catalyst for the selective catalytic reduction of NO x with NH3[J]. ACS Appl. Mater,2014,6(16):14500−8. doi: 10.1021/am5038164 [42] MAITARAD P, HAN J, ZHANG D S, et al. Structure-activity relationships of NiO on CeO2 nanorods for the selective catalytic reduction of NO with NH3: Experimental and DFT studies[J]. J. Phys. Chem. C,2014,118(18):9612−20. doi: 10.1021/jp5024845 [43] 王辰, 史秀锋, 武鲜凤, 等. 氧化还原法制备Mn3O4催化剂及其甲苯催化氧化性能与机理研究[J]. 化工学报,2023,74(6):2447−57.WANG Chen, SHI Xiufeng, WU Xianfeng, et al. Preparation of Mn3O4 catalyst by redox method and its catalytic oxidation performance and mechanism of toluene[J]. CIESC Journal,2023,74(6):2447−57. [44] LIU X, MI J, SHI L, et al. In situ modulation of A-site vacancies in LaMnO3.15 perovskite for surface lattice oxygen activation and boosted redox reactions[J]. Angew. Chem. Int. Ed.,2021,60(51):26747−54. doi: 10.1002/anie.202111610 -




 下载:
下载: