Research on the middle-low temperature denitration and anti-poisoning properties of plate V2O5-MoO3/TiO2 SCR catalysts
-
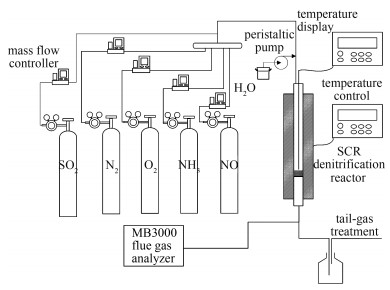
摘要: 针对中低温锅炉烟气脱硝技术需求的特点,采用等体积浸渍法,以V2O5为活性组分、MoO3为助剂,制备了高钒高钼含量的V2O5-MoO3/TiO2型粉末和平板式SCR脱硝催化剂,考察了活性组分和助剂含量对催化剂活性以及抗SO2和H2O中毒性能的影响,对反应前后的催化剂进行了微观表征,并针对最优催化剂研究了其在不同烟气工况下催化剂的脱硝性能。结果表明,提升V2O5负载量可以有效提高催化剂的脱硝活性;MoO3助剂的添加也可以提高催化剂的脱硝活性。XPS、XRF、FT-IR等表征结果表明,MoO3的含量会影响催化剂中V4+/V5+的比值,其相对含量的增加有利于催化剂中非化学计量钒物种的形成以及化学吸附氧比例的增加,钼与钒物种间的交互作用是抑制SO2和H2O对催化剂的毒化作用的关键。3V2O5-10MoO3/TiO2平板式催化剂在温度为200℃、空速为3 500 h-1含SO2和H2O烟气条件下,经30 d连续反应,脱硝效率稳定维持在82%左右,该催化剂在中低温下具有优异的抗SO2和H2O中毒性能以及稳定性。Abstract: Considering the technical requirements for middle-low temperature denitration of flue gas, a series of powder and plate type V2O5-MoO3/TiO2 SCR catalysts were prepared using incipient wetness impregnation method with V2O5 as the active component and with MoO3 as the promoter. Experiments were performed to investigate the effects of active component and promoter contents of the catalysts on the activities and the resistance to deactivation by SO2 and H2O. The characterization of the fresh and used catalysts was conducted, and the optimal catalyst was further studied to reveal the denitration performance under different flue gas conditions. The results indicate that the activities of the catalysts are enhanced with the increase of V2O5 loadings. Also, the addition of MoO3 can promote the catalytic activity. The characterization results from XRF, XPS, FT-IR and other analysis suggest that the MoO3 content could affect the V4+/V5+ ratio in the catalyst. The increase in relative MoO3 content is favorable for the formation of non-stoichiometry vanadium species as well as the rise of chemical adsorption oxygen. Therefore, the interactions between molybdenum and vanadium species might be an essential reason for the resistance to the deactivation by SO2 and H2O. The denitrification efficiency of 3V2O5-10MoO3/TiO2 plate catalyst keeps steady around 82% after 30 days test in the presence of SO2 and H2O at temperature of 200℃ and space velocity of 3 500 h-1. The catalyst is identified to have an excellent resistance to the deactivation by SO2 and H2O under middle-low temperatures.
-
Key words:
- middle-low temperature denitration /
- plate catalyst /
- V2O5 /
- MoO3 /
- resistance to deactivation
-
表 1 催化剂的比表面积和孔结构
Table 1 Specific surface area and pore structure of the catalysts
Sample Catalyst Specific surface area
A/(m2·g-1)Pore volume
v/(cm3·g-1)Average pore diameter
d/nm1 F-3V2O5-10MoO3/TiO2(fresh) 71.43 0.32 17.8 2 F-5V2O5-10MoO3/TiO2(fresh) 50.64 0.25 19.6 3 B-3V2O5-10MoO3/TiO2(fresh) 54.32 0.23 16.7 4 B-5V2O5-10MoO3/TiO2(fresh) 49.04 0.22 18.0 5 F-3V2O5-10MoO3/TiO2(after SCR) 62.09 0.28 18.0 6 F-5V2O5-10MoO3/TiO2(after SCR) 35.78 0.17 19.9 7 B-3V2O5-10MoO3/TiO2(after SCR)* 26.64 0.16 24.4 8 B-5V2O5-10MoO3/TiO2(after SCR) 38.14 0.18 19.3 *duration time of the SO2 and H2O deactivation experiment: NO.5 and NO.6 catalysts were 167 h, NO.7 catalyst was 120 d and NO.8 catalyst was 17 d 表 2 催化剂中各组分含量
Table 2 Each component content of the catalysts
Sample Content w/% V2O5 MoO3 SiO2 Al2O3 SO3 TiO2 1 2.86 8.66 - - - 88.27 2 4.98 8.62 - - - 86.22 3 2.70 8.04 6.59 5.37 - 75.82 4 4.80 7.96 6.38 5.18 - 74.39 5 2.80 8.46 - - 1.64 86.19 6 4.74 8.25 - - 5.43 80.70 7 2.51 7.33 6.21 4.49 8.47 69.06 8 4.34 7.54 6.25 4.93 4.52 71.45 *: annotation: duration time of the SO2 and H2O deactivation experiment: NO.5 and NO.6 catalysts were 167 h, NO.7 catalyst was 120 d and NO.8 catalyst was 17 d -
[1] 丁健, 刘清才, 孔明, 林凡, 杨剑, 任山.燃煤烟气中砷对V2O5-WO3/TiO2 SCR脱硝催化剂性能的影响[J].燃料化学学报, 2016, 44(4):495-499. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18819.shtmlDING Jian, LIU Qing-cai, KONG Ming, LIN Fan, YANG Jian, REN Shan. Influence of arsenic in flue gas on the performance of V2O5-WO3/TiO2 catalyst in selective catalytic reduction of NOx[J]. J Fuel Chem Technol, 2016, 44(4):495-499. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18819.shtml [2] 江博琼. Mn/TiO2系列低温SCR脱硝催化剂制备及其反应机理研究[D].杭州:浙江大学, 2008.JIANG Bo-qiong. The preparation of Mn/TiO2 series low-temperature SCR de NOx catalysts and its reaction mechanism[D]. Hangzhou:Zhejiang University, 2008. [3] 唐志雄, 岑超平, 陈雄波, 陈定盛, 曾文豪.平板玻璃工业窑炉烟气中低温SCR脱硝中试研究[J].环境工程学报, 2015, 9(2):817-822. http://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201502051.htmTANG Zhi-xiong, CEN Chao-ping, CHEN Xiong-bo, CHEN Ding-sheng, ZENG Wen-hao. Pilot-scale study on SCR technology applied in flue gas de NOx of flat glass furnaces at low & middle temperatures[J]. Chin J Environ Eng, 2015, 9(2):817-822. http://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201502051.htm [4] 李哲, 汪莉, 贠丽, 王振南, 李梦霞, 李鑫. Cr-MnOx/TiO2-ZrO2低温选择催化还原NO的活性及抗毒性能[J].工程科学学报, 2015, (8):1049-1056.LI Zhe, WANG Li, YUN Li, WANG Zhen-nan, LI Meng-xia, LI Xin. Activity and antitoxic properties of Cr-MnOx/TiO2-ZrO2 for low-temperature selective catalytic reduction of NO[J]. Chin J Eng, 2015, (8):1049-1056. [5] GAO X, JIANG Y, LUO Z, FU Y, ZHONG Y, CHEN K. Preparation and characterization of CeO2/TiO2 catalysts for selective catalytic reduction of NO with NH3[J]. Catal Commun, 2010, 11(5):465-469. doi: 10.1016/j.catcom.2009.11.024 [6] XU W, HE H, YU Y. Deactivation of a Ce/TiO2 catalyst by SO2 in the selective catalytic reduction of NO by NH3[J]. J Phys Chem C, 2009, 113(11):4426-4432. doi: 10.1021/jp8088148 [7] 彭悦.选择性催化还原氮氧化物铈基催化剂的研究[D].长春:吉林大学, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10183-1012365434.htmPENG Yue. Selective catalytic reduction of NOx on cerium-based catalysts[D]. Changchun:Jilin University, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10183-1012365434.htm [8] 刘亚明, 束航, 徐齐胜, 张玉华, 杨林军. SCR脱硝过程中SO2催化氧化的原位红外研究[J].燃料化学学报, 2015, 43(8):1018-1024. doi: 10.1016/S1872-5813(15)30030-XLIU Ya-ming, SHU Hang, XU Qi-sheng, ZHANG Yu-hua, YANG Lin-jun. FT-IR study of the catalytic oxidation of SO2 during the process of selective catalytic reduction of NO with NH3 over commercial catalysts[J]. J Fuel Chem Technol, 2015, 43(8):1018-1024. doi: 10.1016/S1872-5813(15)30030-X [9] KOBAYASHI M, KUMA R, MORITA A. Low temperature selective catalytic reduction of NO by NH3 over V2O5 supported on TiO2-SiO2-MoO3[J]. Catal Lett, 2006, 112(1/2):37-44. http://www.academia.edu/18733938/Selective_catalytic_reduction_of_NO_with_NH3_over_HZSM-5-supported_fe-cu_nanocomposite_catalysts_The_fe-cu_bimetallic_effect [10] 朱繁, 何洪, 李坚, 吴林彦, 杨品, 邓志鹏. V2O5-MoO3/TiO2催化剂的NOx选择性催化还原及SO2氧化活性[J].工业催化, 2012, 20(9):71-76. http://d.wanfangdata.com.cn/Periodical/dlgc200702029ZHU Fan, HE Hong, LI Jian, WU Lin-yan, YANG Pin, DENG Zhi-peng. Activities of NO selective catalytic reduction and SO2 oxidation over V2O5-MoO3/TiO2 catalysts[J].Ind Catal, 2012, 20(9):71-76. http://d.wanfangdata.com.cn/Periodical/dlgc200702029 [11] PHIL H H, REDDY M P, KUMAR P A, JU L K, HYO J S. SO2 resistant antimony promoted V2O5/TiO2 catalyst for NH3-SCR of NOx at low temperatures[J]. Appl Catal B:Environ, 2008, 78(3/4):301-308. [12] 闫东杰, 玉亚, 黄学敏, 刘树军, 刘颖慧. SO2对Mn-Ce/TiO2低温SCR催化剂的毒化作用研究[J].燃料化学学报, 2016, 44(2):232-238. doi: 10.1016/S1872-5813(16)30011-1YAN Dong-jie, YU Ya, HUANG Xue-min, LIU Shu-jun, LIU Ying-hui. Poisoning effect of SO2 on Mn-Ce/TiO2 catalysts for NO reduction by NH3 at low temperature[J]. J Fuel Chem Technol, 2016, 44(2):232-238. doi: 10.1016/S1872-5813(16)30011-1 [13] SHI Y J, SHU H, ZHANG Y H, FAN F M, ZHANG Y P, YANG L J. Formation and decomposition of NH4HSO4 during selective catalytic reduction of NO with NH3 over V2O5-WO3/TiO2 catalysts[J]. Fuel Process Technol, 2016, 150:141-147. doi: 10.1016/j.fuproc.2016.05.016 [14] KOBAYASHI M, KUMA R, MASAKI S, SUGISHIMA N. TiO2-SiO2 and V2O5/TiO2-SiO2 catalyst:Physico-chemical characteristics and catalytic behavior in selective catalytic reduction of NO by NH3[J]. Appl Catal B:Environ, 2005, 60(s3/4):173-179. [15] GAN L, GUO F, YU J, XU G. Improved low-temperature activity of V2O5-WO3/TiO2 for denitration using different vanadium precursors[J]. Catal, 2016, 6(2). [16] ARNARSON L, RASMUSSEN S B, FALSIG H, LAURITSEN J V, MOSES P G. Coexistence of square pyramidal structures of oxo Vanadium (+5) and (+4) species over low-coverage VOx/TiO2 (101) and (001) anatase catalysts[J]. J Phys Chem C, 2015, 119(41):23445-23452. doi: 10.1021/acs.jpcc.5b06132 [17] LÁZARO M J, BOYANO A, HERRERA C, LARRUBIA M A, ALEMANY L J, MOLINER R. Vanadium loaded carbon-based monoliths for the on-board NO reduction:Influence of vanadia and tungsten loadings[J]. Chem Eng J, 2009, 155(1/2):68-75. [18] SANG H C, CHO S P, LEE J Y, HONG S H, HONG S C, HONG S I. The influence of non-stoichiometric species of V/TiO2 catalysts on selective catalytic reduction at low temperature[J]. J Mol Catal A:Chem, 2009, 304(1):166-173. https://www.researchgate.net/publication/244279029_The_influence_of_non-stoichiometric_species_of_VTiO_2_catalysts_on_selective_catalytic_reduction_at_low_temperature [19] YAN W, SHEN Y, ZHU S, JIN Q, LIU Y, LI X. Promotional effect of Molybdenum additives on catalytic performance of CeO2/Al2O3, for selective catalytic reduction of NOx[J]. Catal Lett, 2016, 146(7):1221-1230. doi: 10.1007/s10562-016-1739-0 [20] KORNELAK P, SU D S, THOMAS C, CAMRA J, WESELUCHA-BIRCZYNSKAET A, TOBA M. Surface species structure and activity in NO decomposition of an anatase-supported V-O-Mo catalyst[J]. Catal Today, 2008, 137(2/4):273-277. https://www.researchgate.net/publication/244322741_Surface_species_structure_and_activity_in_NO_decomposition_of_an_anatase-supported_VOMo_catalyst [21] AL-KANDARI H, AL-KHARAFI F, AL-AWADI N, EI-DUSOUQUI O M, ALI S A, KATRIB A. The catalytic active sites in partially reduced MoO3 for the hydroisomerization of 1-pentene and n-pentane[J]. Appl Catal A:Gen, 2005, 295(1):1-10. doi: 10.1016/j.apcata.2005.07.023 [22] LIU J, LI X, ZHAO Q, KE J, XIAO H, LV X. Mechanistic investigation of the enhanced NH3-SCR on cobalt-decorated Ce-Ti mixed oxide:In situ, FTIR analysis for structure-activity correlation[J]. Appl Catal B:Environ, 2016, 200:297-308. https://www.researchgate.net/publication/305396631_Mechanistic_investigation_of_the_enhanced_NH3-SCR_on_cobalt-decorated_Ce-Ti_mixed_oxide_in_situ_FTIR_analysis_for_structure-activity_correlation [23] FANG J, BI X, SI D, JIANG Z, HUANG W. Spectroscopic studies of interfacial structures of CeO2-TiO2 mixed oxides[J]. Appl Surf Sci, 2007, 253(22):8952-8961. doi: 10.1016/j.apsusc.2007.05.013 [24] GU T, LIU Y, WENG X, WANG H, WU Z. The enhanced performance of ceria with surface sulfation for selective catalytic reduction of NO by NH3[J]. Catal Commun, 2010, 12(4):310-313. doi: 10.1016/j.catcom.2010.10.003 [25] KWON D W, PARK K H, HONG S C. Enhancement of SCR activity and SO2 resistance on VOx/TiO2 catalyst by addition of molybdenum[J]. Chem Eng J, 2016, 284:315-324. doi: 10.1016/j.cej.2015.08.152 [26] AUDI A A, SHERWOOD P M A. X-ray photoelectron spectroscopic studies of sulfates and bisulfates interpreted by Xα and band structure calculations[J]. Surf Interface Anal, 2000, 29(4):265-275. doi: 10.1002/(ISSN)1096-9918 [27] 曹政, 黄妍, 彭莉莉, 李建光. V2O5-Sb2O3-TiO2催化剂低温NH3还原NO及其抗H2O和SO2毒化性能[J].燃料化学学报, 2012, 40(4):456-462. http://rlhxxb.sxicc.ac.cn/CN/article/showSupportInfo.do?id=17927CAO Zheng, HUANG Yan, PENG Li-li, LI Jian-guang. Selective catalytic reduction of NO with ammonia over V2O5-Sb2O3-TiO2 at low temperature and resistance to H2O and SO2 poisoning[J]. J Fuel Chem Technol, 2012, 40(4):456-462. http://rlhxxb.sxicc.ac.cn/CN/article/showSupportInfo.do?id=17927 [28] 束航, 张玉华, 范红梅, 张亚平, 杨林军. SCR脱硝中催化剂表面NH4HSO4生成及分解的原位红外研究[J].化工学报, 2015, 66(11):4460-4468. http://www.hgxb.com.cn/CN/abstract/abstract17531.shtmlSHU Hang, ZHANG Yu-hua, FAN Hong-mei, ZHANG Ya-ping, YANG Lin-jun. FT-IR study of the formation and decomposition of ammonium bisulfates on the surface of SCR catalyst for nitrogen removal[J]. J Chem Ind Eng, 2015, 66(11):4460-4468. http://www.hgxb.com.cn/CN/abstract/abstract17531.shtml [29] 黄继辉, 童华, 童志权, 张俊丰, 黄妍. H2O和SO2对Mn-Fe/MPS催化剂用于NH3低温还原NO的影响[J].过程工程学报, 2008, 8(3):517-522. http://www.cqvip.com/Main/Detail.aspx?id=27465987HUANG Ji-hui, TONG Hua, TONG Zhi-quan, ZHANG Jun-feng, HUANG Yan. Effects of H2O and SO2 on Mn-Fe/MPS catalyst for NO reduction by NH3 at lower temperatures[J]. Chin J Process Eng, 2008, 8(3):517-522. http://www.cqvip.com/Main/Detail.aspx?id=27465987 -





 下载:
下载: